- Professor of Clinical Neurosurgery, School of Medicine, State University of N.Y. at Stony Brook, Mineola, New York, USA
- Chief of Neurosurgical Spine and Education, NYU Winthrop Hospital, NYU Winthrop NeuroScience, Mineola, New York, USA
Correspondence Address:
Nancy E. Epstein
Professor of Clinical Neurosurgery, School of Medicine, State University of N.Y. at Stony Brook, Mineola, New York, USA
Chief of Neurosurgical Spine and Education, NYU Winthrop Hospital, NYU Winthrop NeuroScience, Mineola, New York, USA
DOI:10.4103/sni.sni_230_18
Copyright: © 2018 Surgical Neurology International This is an open access journal, and articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as appropriate credit is given and the new creations are licensed under the identical terms.How to cite this article: Nancy E. Epstein. A review of the disagreements in the prevalence and treatment of the tethered cord syndromes with chiari-1 malformations. 14-Aug-2018;9:161
How to cite this URL: Nancy E. Epstein. A review of the disagreements in the prevalence and treatment of the tethered cord syndromes with chiari-1 malformations. 14-Aug-2018;9:161. Available from: http://surgicalneurologyint.com/surgicalint-articles/a-review-of-the-disagreements-in-the-prevalence-and-treatment-of-the-tethered-cord-syndromes-with-chiari%e2%80%911-malformations/
Abstract
Background:The tethered cord syndrome (TCS) accompanying Chiari-1 (CM-1) malformations and the occult tethered cord syndrome (OTCS) syndrome accompanying the low lying cerebellar tonsil (LLCT) syndrome may be treated with sectioning of the filum terminale (SFT).
Methods:Utilizing PubMed, we reviewed the neurosurgical literature to determine how frequently spinal neurosurgeons diagnosed the TCS (e.g., conus terminating below the normal L1-L2 disc level) on lumbar magnetic resonance (MR) studies in patients with CM-1 malformations [e.g. tonsils >5–12 mm below the foramen magnum (FM) warranting SFT]. In addition, we assessed how frequently spinal neurosurgeons encountered the OTCS (e.g., conus normally located at L1-L2 on MR) accompanying the LLCT (e.g., tonsils herniated
Results:According to the neurosurgical literature, the incidence of TCS accompanying CM-1 requiring SFT ranged from 2.2% to
Conclusions:Given the differences in the literature, more studies are needed to assess the risks (complications) vs. benefits (improved neurological outcomes) of SFT surgery for TCS with CM-1 and SFT for OCTS with LLCT.
Keywords: Chiari-1 malformations, definitions, indications, low lying cerebellar tonsil syndrome, occult tethered cord syndrome, prevalence, section of filum terminale, tethered cord syndrome
INTRODUCTION
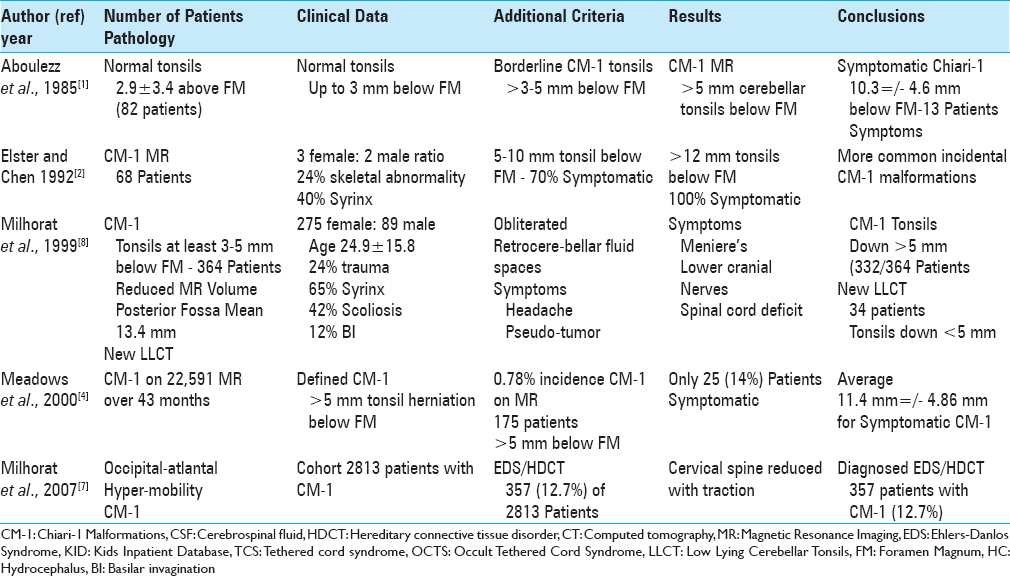
Utilizing PubMed, we reviewed the neurosurgical literature regarding the prevalence of the tethered cord syndrome (TCS) with Chiari-1 malformations (CM-1) requiring sectioning of the filum terminale (SFT). Additionally, we assessed how often the occult tethered cord syndrome (OCTS) was correlated with the low lying cerebellar syndrome (LLCT) also warranting SFT. Further, we asked how frequently the TCS, defined on magnetic resonance (MR) by the conus located below the L1-L2 level, correlated with CM-1 (tonsils >5 mm–12 mm below the foramen magnum FM), and with the LLCT [tonsils herniated < 5 mm below the FM on MR] [Tables
DEFINITIONS OF NORMAL TONSIL LOCATION VERSUS CHIARI-1 MALFORMATIONS
Normally, the cerebellar tonsils are located 2.9 mm ± 3.4 mm above or up to 3 mm below the FM [
FREQUENCIES OF CHIARI-1 MALFORMATIONS IN ADULTS AND CHILDREN
Different frequencies of CM-1 have been reported in the adult and pediatric age groups [Tables
SYMPTOMATIC CHIARI-1 MALFORMATIONS
According to the literature, the onset of symptoms correlated with differing degrees of CM-1 tonsillar herniation [
DEFINITION AND FREQUENCY OF THE LOW LYING CEREBELLAR TONSIL SYNDROME (LLCT)
The neurosurgical literature revealed few references to the LLCT [
ANOMALIES ASSOCIATED WITH ADULT/PEDIATRIC CHIARI-1/LOW LYING CEREBELLAR TONSIL SYNDROME (LLCT)
The neurosurgical literature demonstrated multiple anomalies accompanying the CM-1/LLCT syndromes in adult and pediatric populations [Tables
DIFFERENT FREQUENCIES OF TETHERED CORD SYNDROME WITH CHIARI-1
A review of the neurosurgical literature revealed a variable correlation between CM-1 and the TCS (e.g. conus below the L1-L2 disc level on MR) requiring SFT [
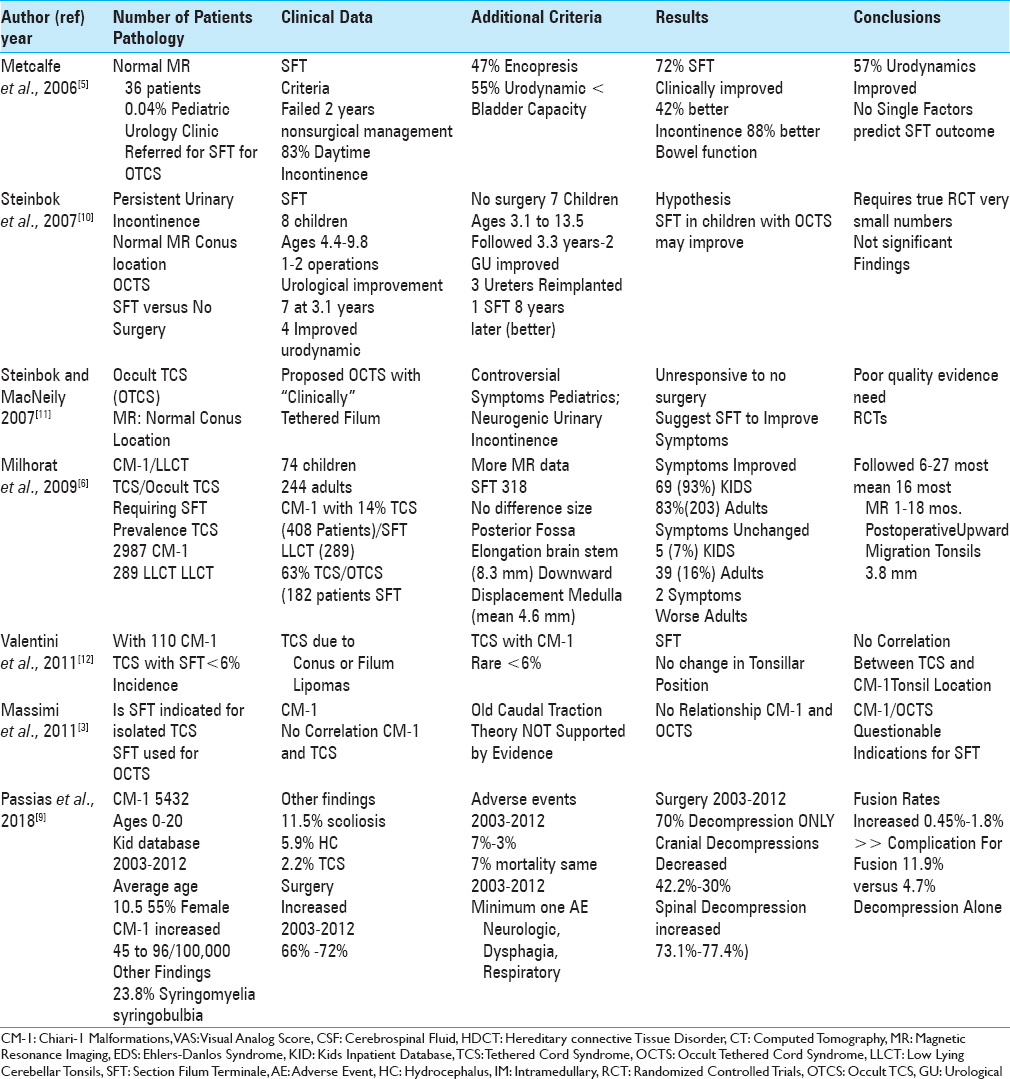
CRANIAL VS. SPINAL DECOMPRESSION VS. FUSION FOR CHIAIR-1 MALFORMATIONS IN THE PEDIATRIC AGE GROUP
In the pediatric neurosurgical literature, the study by Passias et al. (2018) was the most prominent one discussing the successive 2003–2013 frequencies of cranial vs. spinal decompression vs. fusion for CM-1 malformations [
DEFINITION OF THE OCCULT TETHERED CORD SYNDROME (OCTS)
A review of the neurosurgical literature showed that few spinal neurosurgeons highly correlated the OCTS (normal location of the conus at the L1-L2 level on MR) warranting SFT with the LLCT [
DIFFERENT FREQUENCIES OF SECTIONING OF THE FILUM TERMINALE FOR OCCULT TETHERED CORD SYNDROME IN PATIENTS WITH CHIARI-1/LOW LYING CEREBELLAR TONSIL SYNDROMES (LLCT)
Multiple studies in the neurosurgical literature summarized the results of SFT performed for OTCS in patients with CM-1/LLCT syndromes [
CONCLUSION
Reviewing the neurosurgical literature revealed a 2.2% to <6%, up to a higher 14% frequency of TCS requiring SFT in patients with CM-1 malformations [
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Aboulezz AO, Sartor K, Geyer CA, Gado MH. Position of cerebellar tonsils in the normal population and in patients with Chiari malformation: A quantitative approach with MR imaging. J Comput Assist Tomogr. 1985. 9: 1033-6
2. Elster AD, Chen MY. Chiari I malformations: Clinical and radiologic reappraisal. Radiology. 1992. 183: 347-53
3. Massimi L, Peraio S, Peppucci E, Tamburrini G, Di Rocco C. Section of the filum terminale: Is it worthwhile in Chiari type I malformation?. Neurol Sci. 2011. 32: S349-51
4. Meadows J, Kraut M, Guarnieri M, Haroun RI, Carson BS. Asymptomatic Chiari type I malformations identified on magnetic resonance imaging. J Neurosurg. 2000. 92: 920-6
5. Metcalfe PD, Luerssen TG, King SJ, Kaefer M, Meldrum KK, Cain MP. Treatment of the occult tethered spinal cord for neuropathic bladder: Results of sectioning the filum terminale. J Urol. 2006. 176: 1826-9
6. Milhorat TH, Bolognese PA, Nishikawa M, Francomano CA, McDonnell NB, Roonprapunt C. Association of Chiari malformation type I and tethered cord syndrome: Preliminary results of sectioning filum terminale. Surg Neurol. 2009. 72: 20-35
7. Milhorat TH, Bolognese PA, Nishikawa M, McDonnell NB, Francomano CA. Syndrome of occipitoatlantoaxial hypermobility, cranial settling, and Chiari malformation type I in patients with hereditary disorders of connective tissue. J Neurosurg Spine. 2007. 7: 601-9
8. Milhorat TH, Chou MW, Trinidad EM, Kula RW, Mandell M, Wolpert C. Chiari I malformation redefined: Clinical and radiographic findings for 364 symptomatic patients. Neurosurgery. 1999. 44: 1005-17
9. Passias PG, Pyne A, Horn SR, Poorman GW, Janjua MB, Vasquez-Montes D. Developments in the treatment of Chiari type 1 malformations over the past decade. J Spine Surg. 2018. 4: 45-54
10. Steinbok P, Kariyattil R, MacNeily AE. Comparison of section of filum terminale and non-neurosurgical management for urinary incontinence in patients with normal conus position and possible occult tethered cord syndrome. Neurosurgery. 2007. 61: 550-5
11. Steinbok P, MacNeily AE. Section of the terminal filum for occult tethered cord syndrome: Toward a scientific answer. Neurosurg Focus. 2007. 23: E5-
12. Valentini LG, Selvaggio G, Visintini S, Erbetta A, Scaioli V, Solero CL. Tethered cord: Natural history, surgical outcome and risk for Chiari malformation 1 (CM1): A review of 110 detethering. Neurol Sci. 2011. 32: S353-6