- Department of Neurosurgery, Sher-I-Kashmir Institute of Medical Sciences, Srinagar, Jammu and Kashmir, India
Correspondence Address:
Arif Hussain Sarmast
Department of Neurosurgery, Sher-I-Kashmir Institute of Medical Sciences, Srinagar, Jammu and Kashmir, India
DOI:10.4103/2152-7806.183498
Copyright: © 2016 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Kirmani AR, Sarmast AH, Bhat AR. A unique case of calvarial hemangioma. Surg Neurol Int 03-Jun-2016;7:
How to cite this URL: Kirmani AR, Sarmast AH, Bhat AR. A unique case of calvarial hemangioma. Surg Neurol Int 03-Jun-2016;7:. Available from: http://surgicalneurologyint.com/surgicalint_articles/a-unique-case-of-calvarial-hemangioma/
Abstract
Background:Calvarial hemangiomas are one of the rarest neoplasms affecting the skull, predominantly occurring in parietal and frontal bones.
Case Description:We report a parietal hemangioma in a middle-aged female which presented as a painless swelling that was progressively increasing in size and was treated surgically.
Conclusion:Although rare, calvarial hemangioma should be a part of the differential diagnosis of calvarial swellings.
Keywords: Calvarial, hemangioma, skull
INTRODUCTION
Primary intraosseous hemangiomas are benign slow-growing neoplasms. More than 50% of incidences occur in the vertebrae and skull.[
CASE REPORT
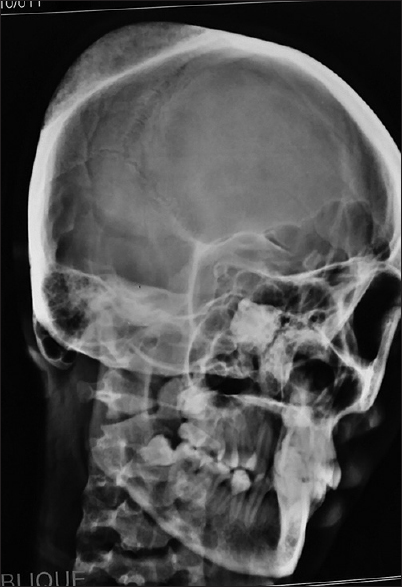
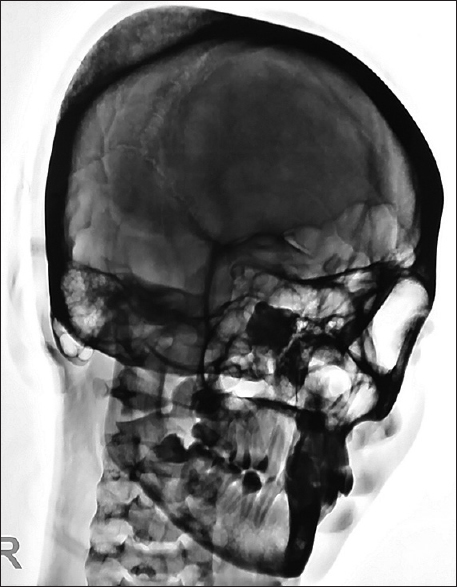
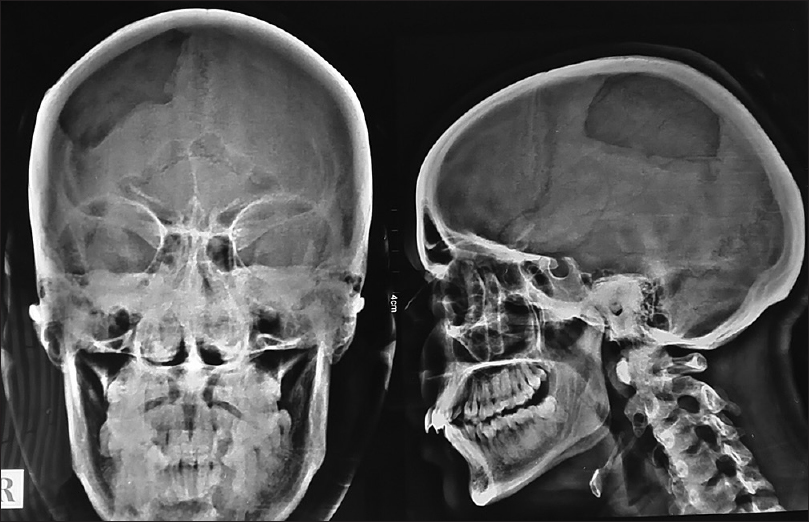
A 40-year-old female with no history of head trauma presented with painless swelling in the parietal area, progressively increasing in size, since last 2 years. The patient complained of local tenderness. Anteroposterior and lateral views of plain X-ray skull revealed classical honeycomb appearance lesion [Figures
DISCUSSION
Presentation
Calvarial hemangiomas are benign, malformed vascular lesions, are usually asymptomatic, and are usually discovered incidentally on imaging or postmortem examination. They are mostly encountered in the middle-aged persons, generally occurs more commonly in females than males with a ratio of 3:2. There is no documental racial variation in the frequency of hemangiomas of skull, worldwide. Though cranial hemangiomas are second only to vertebral hemangiomas in frequency, such lesions are rarely congenital and multiple. It is probable that the true incidence of congenital calvarial hemangiomas is higher than that reported in the literature, as they are unlikely to undergo imaging, most being asymptomatic and without a significant soft tissue component.[
Symptomatic patients present with headaches, often with focal scalp pain or swelling. A palpable bony sessile mass may or may not be present.[
Cases have been reported from the roof of the orbit when they present with proptosis, blindness, or extraocular palsies.[
Differential diagnosis
The differential diagnosis of a solitary circumscribed expansile intradiploic cranial lesion includes an osteoma, aneurysmal bone cyst, giant cell tumor, fibrous dysplasia, Langerhans’ cell histiocytosis, sarcoma, meningioma, metastatic disease, Paget's disease, lymphoma, and dermoid tumor.[
Pathology
According to the predominant type of vascular channel, four histological variants of hemangiomas are known; cavernous, capillary, arteriovenous, and venous. Bone hemangiomas are predominantly of the cavernous and capillary varieties. Cavernous hemangiomas most frequently occur in the skull whereas capillary hemangiomas predominate in the vertebral column; overall cavernous ones are commonly seen. Malignant degeneration is virtually unknown. Rarely, locally aggressive growth patterns are recognized, mimicking malignant lesions. Gross pathology reveals well-demarcated, unencapsulated lesions with cystic red cavities. Microscopic examination shows hamartomatous proliferations of vascular tissue within endothelial-lined spaces, but may also contain fat, smooth muscle, fibrous tissue, and thrombus.[
Radiographic features
Plain film
Evaluation of cranial vault hemangioma starts with a plain radiograph, which usually shows lytic lesion with a sclerotic rim (usually with a honeycomb or sunburst-like appearance). Outer table is usually involved with the preservation of the inner table. Prominent vascular grooves are usually present.
Computed tomography
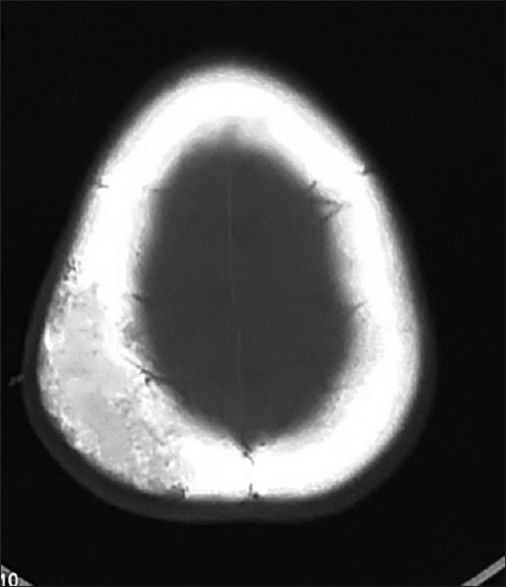
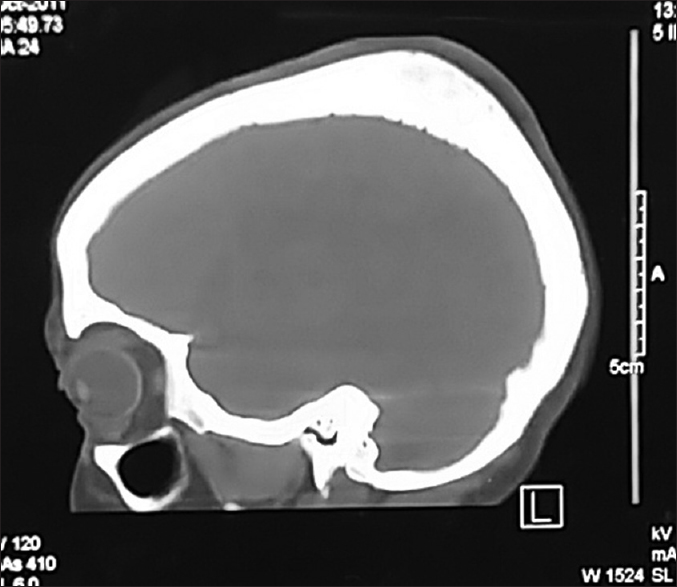
CT scan is required for further characterization of these lesions as it is useful for assessing the changes in bone trabeculae and supports the plain radiographic findings, in a greater detail. Usually, it presents as an expansive bone lesion with thin borders and characteristic sunburst pattern of trabecular thickening radiating from a common center. Erosions of both internal and external plates can occur and may be associated with internal or external tumor expansion. On contrast administration, homogenous enhancement is seen.[
Magnetic resonance imaging
MRI features largely depend on the proportion of fat and vascularity of the lesions. Scattered foci of hyper- and hypo-intense T1 signal with predominant intermediate T1 signal are seen on MRI imaging. Low signal intensity on T1-weighted images indicates decreased marrow fat or greater vascular component, may be correlated with more aggressive behavior. Thickened trabeculae demonstrate low signal intensity on MRIs obtained in all sequences.[
On T2-weighted images, hemangiomas generally appear as multiple high-signal intensity lobules that resemble a bunch of grapes. This appearance is due to cavernous or cystic vascular spaces containing stagnant blood. Fluid-fluid levels can also be noted within these spaces.[
Digital subtraction angiography
Angiography confirms the hypervascularity of the lesions showing enlargement of the external carotid artery branches.[
Skeletal scintigraphy
On 99mTc methylene diphosphonate scintigrams, calvarial hemangiomas may show a level of radionucleotide uptake that is lower than, equal to, or slightly higher than that in surrounding bone. Hence, in addition, use of adjunctive99m Tc-labeled red blood cell (RBC) scans is made. Likewise the dynamic scintigraphic imaging in the diagnosis of hepatic hemangiomas which is characterized by increased accumulation of a RBC tracer on delayed blood pool images, similar criterion has been used in the diagnosis of calvarial hemangiomas; however, detection is limited in lesions <10 mm.[
Treatment and prognosis
Treatment is not always necessary and indications include mass effects, hemorrhage control, and esthetic improvement. Whenever indicated, total surgical removal is the recommended treatment and is determinant of a definitive diagnosis. Radiotherapy is advisable in situations where excision is not feasible. Doses up to 30 Gy (3000 rads), in 3 weeks, may be required. In addition to these, embolization to reduce the intraoperative blood loss and intralesional steroid injection have also been tried.[
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Banerji D, Inao S, Sugita K, Kaur A, Chhabra DK. Primary intraosseous orbital hemangioma: A case report and review of the literature. Neurosurgery. 1994. 35: 1131-4
2. Davis DO, Rumbaugh CL, Petty J. Calvarial hemangioma: Tumor stain and meningeal artery blood supply. Case report. J Neurosurg. 1966. 25: 561-3
3. Dorfman HD, Steiner GC, Jaffe HL. Vascular tumors of bone. Hum Pathol. 1971. 2: 349-76
4. Gavant ML, Massie JD, Gerald B. Radionuclide blood pool imaging of calvarial hemangioma. Clin Nucl Med. 1983. 8: 495-6
5. Gottfried ON, Gluf WM, Schmidt MH. Cavernous hemangioma of the skull presenting with subdural hematoma. Case report. Neurosurg Focus. 2004. 17: ECP1-
6. Gourin CG, Millay DJ. Pathology forum: Quiz case 3. Diagnosis: Cavernous hemangioma of the nasal bones. Arch Otolaryngol Head Neck Surg. 2000. 126: 906-7
7. Honda M, Toda K, Baba H, Yonekura M. Congenital cavernous angioma of the temporal bone: Case report. Surg Neurol. 2003. 59: 120-3
8. Ki WW, Shin JW, Won KS, Ryu JS, Yang SO, Lee HK. Diagnosis of orbital cavernous hemangioma with Tc-99m RBC SPECT. Clin Nucl Med. 1997. 22: 546-9
9. Koulouris G, Rao P. Multiple congenital cranial hemangiomas. Skeletal Radiol. 2005. 34: 485-9
10. Kumar NA, Ranganadham P, Bhaskar G, Chowdhury AR. Multiple calvarial cavernous haemangiomas: Case report and review of the literature. Neuroradiology. 1996. 38: S83-5
11. Kuzeyli K, Usul H, Cakir E, Caylan R, Rei A, Baykal S. Multifocal intradiploic cavernous hemangioma of the skull associated with nasal osteoma. Acta Neurochir (Wien). 2003. 145: 323-6
12. Moss WT, Brand WN, Battifora H.editors. The heart and blood vessels. Radiation Oncology, Rationale, Technique, Results. St. Louis: CV Mosby Co; 1973. p. 248-56
13. Peterson DL, Murk SE, Story JL. Multifocal cavernous hemangioma of the skull: Report of a case and review of the literature. Neurosurgery. 1992. 30: 778-81
14. Politi M, Romeike BF, Papanagiotou P, Nabhan A, Struffert T, Feiden W. Intraosseous hemangioma of the skull with dural tail sign: Radiologic features with pathologic correlation. AJNR Am J Neuroradiol. 2005. 26: 2049-52
15. Ramamurthi B, Narasimhan ST, Pillai KM. Haemangioma of the skull. J Indian Med Assoc. 1959. 32: 201-3
16. Ramon F, De Schepper AM.editors. Tumors and tumor-like lesions of blood vessels. Imaging of Soft Tissue Tumors. Berlin, Germany: Springer-Verlag; 2001. p. 225-44
17. Reis BL, Carvalho GT, Sousa AA, Freitas WB, Brandão RA. Primary hemangioma of the skull. Arq Neuropsiquiatr. 2008. 66: 569-71
18. Rosenbaum AE, Rossi P, Schechter MM, Sheehan JP. Angiography of haemangiomata of the calvarium. Br J Radiol. 1969. 42: 682-7
19. Sayit E, Durak I, Capakaya G, Yilmaz M, Durak H. The role of Tc-99m RBC scintigraphy in the differential diagnosis of orbital cavernous hemangioma. Ann Nucl Med. 2001. 15: 149-51
20. Suzuki Y, Ikeda H, Matsumoto K. Neuroradiological features of intraosseous cavernous hemangioma – case report. Neurol Med Chir (Tokyo). 2001. 41: 279-82
21. van Rijswijk CS, van der Linden E, van der Woude HJ, van Baalen JM, Bloem JL. Value of dynamic contrast-enhanced MR imaging in diagnosing and classifying peripheral vascular malformations. AJR Am J Roentgenol. 2002. 178: 1181-7