- Department of Neurosurgery, Karolinska University Hospital, Stockholm, Sweden
- Department of Neuroradiology, Karolinska University Hospital, Stockholm, Sweden
- Department of Medical Radiation Physics and Nuclear Medicine, Karolinska University Hospital, Stockholm, Sweden
Correspondence Address:
Amir Samadi
Department of Neurosurgery, Karolinska University Hospital, Stockholm, Sweden
DOI:10.4103/sni.sni_391_16
Copyright: © 2017 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Georges Sinclair, Heather Martin, Michael Fagerlund, Amir Samadi, Hamza Benmakhlouf, Ernest Doodo. Adaptive hypofractionated gamma knife radiosurgery in the acute management of large thymic carcinoma brain metastases. 26-May-2017;8:95
How to cite this URL: Georges Sinclair, Heather Martin, Michael Fagerlund, Amir Samadi, Hamza Benmakhlouf, Ernest Doodo. Adaptive hypofractionated gamma knife radiosurgery in the acute management of large thymic carcinoma brain metastases. 26-May-2017;8:95. Available from: http://surgicalneurologyint.com/surgicalint-articles/adaptive-hypofractionated-gamma-knife-radiosurgery-in-the-acute-management-of-large-thymic-carcinoma-brain-metastases/
Abstract
Background:Brain metastases often lead to serious neurological impairment and life threatening states. Their acute management remains complex, particularly in the case of rare malignancies with aggressive evolution. In large single lesions, open surgery followed by radiation to the surgical cavity is widely regarded as the best approach; yet in many cases, microsurgery is not feasible due to the lesion's critical location and/or the number of brain metastases present. We report the effects of adaptive hypofractionated gamma knife radiosurgery in the acute management of critically located thymic carcinoma metastases.
Case Description:A 50-year-old male with metastatic thymic carcinoma was treated with radiosurgery for two large supratentorial brain metastases (M3 and M4) adjacent to eloquent areas and one smaller cerebellar metastasis (M2). M3 and M4 were treated with adaptive hypofractionated gamma knife radiosurgery, showing a dramatic volume reduction 4 weeks after treatment completion without radiation-induced side effects. Thirteen months later, two new small, threatening supratentorial lesions (M5-M6) were treated with the same technique. Interestingly, M2 (treated with standard single fraction) and M5-M6 developed local adverse radiation events. The patient's general and neurological status remained next to normal by the time of paper submission.
Conclusion:The application of adaptive hypofractionated radiosurgery in this acute setting proved effective in terms of rapid tumor ablation, with salvage of neurological functionality and limited toxicity. We have called the overall procedure rapid rescue radiosurgery (RRR). A systematic study of past and ongoing RRR-treatments is warranted and in progress.
Keywords: Adaptive hypofractionation, adverse radiation event, critical areas, gamma knife radiosurgery, recursive partitioning analysis, whole brain radiation induced cognitive impairment
INTRODUCTION
Brain metastases often lead to serious neurological impairment and life threatening states. Their acute management remains complex, particularly in the case of rare malignancies such as thymic carcinoma. In large single lesions, open surgery followed by radiation to the surgical cavity is widely regarded as the best approach; yet in many cases, microsurgery is not feasible due to the lesion's critical location and/or the number of brain metastases present. Our case report describes the effects of adaptive hypofractionated gamma knife radiosurgery in the acute management of critically located brain metastases.
CASE PRESENTATION
We present the case of a 50-year-old male patient reporting intermittent upper abdominal pain throughout 2012 and 2013. In February 2014, the patient experienced breathing difficulties and exacerbation of pain. Fludeoxyglucose positron emission tomography/computed tomography (18F-FDG PET/CT) revealed high FDG uptake in a large, heterogeneous intrathoracic mass in the middle mediastinum abutting the right atrium and the right hemidiaphragm, without evidence of distant metastases. The patient underwent subtotal primary tumor resection in April 2014. The histopathology demonstrated a poorly differentiated thymic carcinoma though without primary malignant manifestation in the gland itself. The patient received three adjuvant chemotherapy sessions (Carboplatine/Vepesid) followed by fractionated radiotherapy (2 Gy/day, 30 sessions = 2 Gy × 30 = 60 Gy) to the right thoracic region (July to September 2014). The patient developed paroxysmal atrial fibrillation after thoracic surgery as well as a series of respiratory infections during and after his radiochemotherapy, though without serious sequelae. His general condition remained otherwise stable. By February 2015, the patient developed progressive expressive dysphasia. A brain magnetic resonance imaging (MRI) in May 2015 showed a 3.5 cm ring-enhancing lesion in the left temporal lobe with extensive edema (metastasis # 1 = M1). Speech impairment resolved shortly after gross total resection (GTR). The histopathology proved the lesion to be a thymic carcinoma metastasis with MIB 1 proliferation index of up to 50%. Postsurgical hypofractionated LINAC-radiotherapy (6 Gy/day, 5 sessions = 30 Gy) was delivered to the surgical cavity in June 2015. CT of the neck, thorax, and abdomen in September 2015 showed no signs of distant metastases. However, a follow-up brain MRI in October 2015 demonstrated three new intracranial lesions – the first a 1.9 cm lesion in the right cerebellar hemisphere (metastasis # 2 = M2), the second, a larger extra-axial left temporal mass measuring 3.9 cm with adjacent edema and mass effect threatening the hippocampus, uncus, and language areas (metastasis # 3 = M3), and the third, a 3.0 cm dural-based left parietal lesion exerting mass effect on the sensory cortex (metastasis # 4 = M4). There was no evidence of recurrence in the surgical cavity (M1). Although M2 was relatively small, M3 and M4 were quite large (≥3 cm) and both threatened eloquent areas of the brain. By this time, the patient once again developed expressive dysphasia and mental fatigue [Karnofsky Performance Score (KPS) 90–100], though without evidence of extracranial tumour proliferation (RPA 1) on prior screening CT scan September 2015; the patient remained free from systemic treatment at this stage and was referred to our unit for further management (October 2015).
Radiosurgical account
The radiosurgical plan was designed based on a series of factors – past/present radiological evolution, tumor localization, identification of organs at risk (OAR), tumor volume, histopathological traits of prior resected metastasis (M1), general and neurological health status (KPS/RPA), absence of extracranial tumor activity at the time of radiosurgery, positive outcome after microsurgery, and good response to prior anti-tumoral treatments (including chemotherapy and extra/intracranial radiation). Our strategy aimed to rapidly relieve/salvage the language areas, the hippocampus, and the post-central gyrus from the larger left-sided lesions (M3 and M4) within a time frame of 7 days (treatment time between RS1 and RS3) to 4 weeks (MRI at 1 month). Because of the patient's previous positive response to extra and intracranial radiotherapy, we considered the tumor to be radiosensitive and proceeded to plot a peripheral prescription dose biologically isoeffective to the cranial hypofractionated treatment delivered to the surgical cavity earlier (6 Gy × 5). To achieve maximal probability of local tumor control while minimizing the risk of focal radiation-induced adverse reaction, treatment settings included an adaptive and MRI-guided hypofractionated radiosurgical approach; the procedure would allow tumor bed dose distribution readjustments in relation to local tumor volume variations during the course of treatment. The treatment was structured on three radiosurgeries (RS) delivered every third day i.e., day 1 (RS 1), day 4 (RS 2), and day 7 (RS 3). Cranial fixation was achieved by applying a Leksell Coordinate Frame G (Elekta AB, Stockholm) prior to each RS. A stereotactic MRI was performed before each RS session (s-MRI 1, 2, 3) for proper gross tumor volume (GTV) delineation; we set no margins to the GTV (GTV = CTV = PTV). As described in Tables
Table 1
LGP-based tumor volume estimates covering time of treatment (Day 1 to Day 7) and follow up period (1 to 7 months). *M3 showed signs of hemorrhage at RS 2 (Day 4) and RS 3 (Day 7); bleeding zones were included in the field of treatment (GTV); see
RESULTS
The radiological evolution during the course of treatment proved interesting for all concerned lesions. M2 developed increased central necrosis between s-MRI 1 (day 1) and s-MRI 2 (day 4). M3 developed local hemorrhage at s-MRI 2 (day 4) while its solid component suffered a slight volume reduction at s-MRI 3 (day 7) [Tables
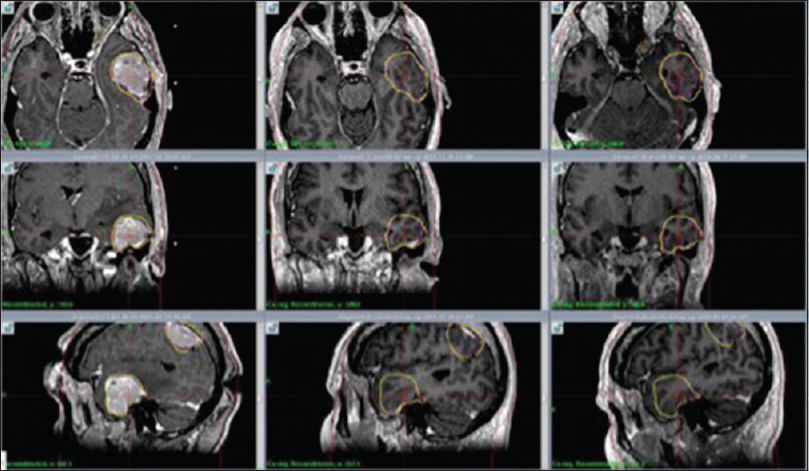
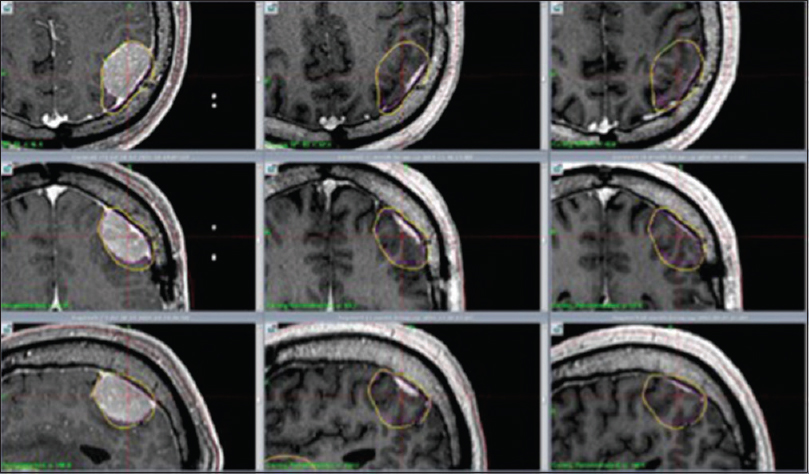
Figure 2
M4: Axial, coronal and sagittal contrast enhanced T1 weighted MR images show significant tumor reduction at 1 month (center column) and 7 months (right column) respectively after RRR. Left column shows tumor size at day 1 (RS 1). Significant tumor reduction at 1 month (center column) and 7 months (right column)
Figure 4
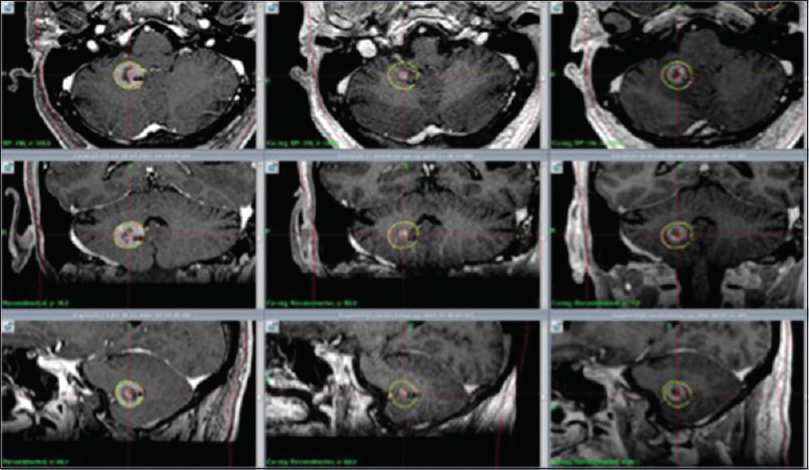
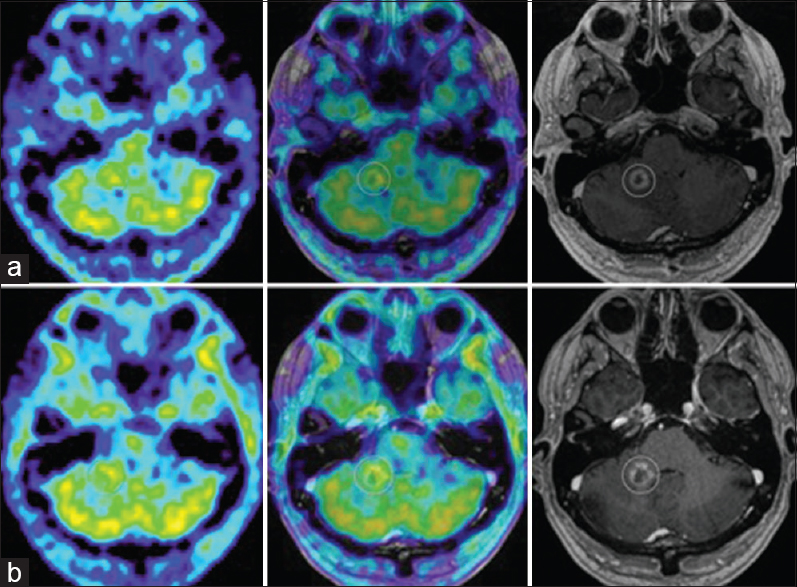
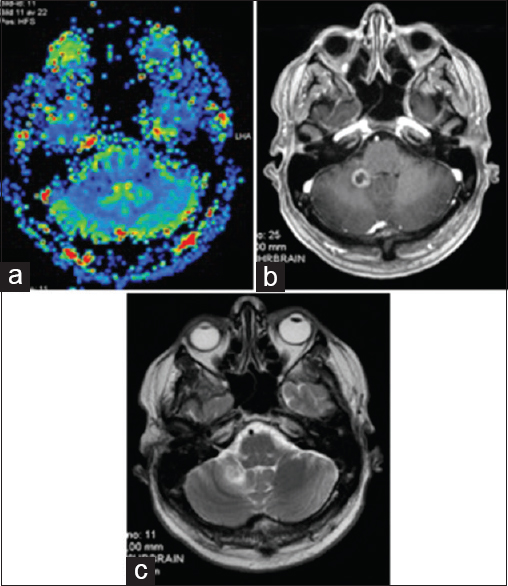
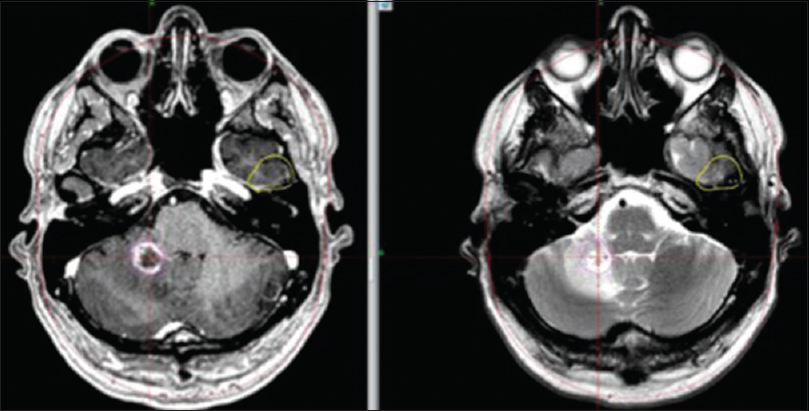
(a) M2's evolution on amino acid PET. 11C-Methionine PET (MET PET, left), MET PET fused with MRI CE T1 (middle) and MRI CE T1 weighted (right) axial images at 7 months demonstrating intermediate focal MET uptake in the medial aspect of the ring enhancing right cerebellar lesion (T/N ratio = 1.5 compared to contralateral mirrored tissue) and local edema. (b) MET PET (left) at 10 months now demonstrating T/N ratio = 1.3, diminished compared to MET PET 3 months prior, MET PET fused with MRI CE T1 (middle) and MRI CE T1 weighted (right): suspected ARE
Figure 7
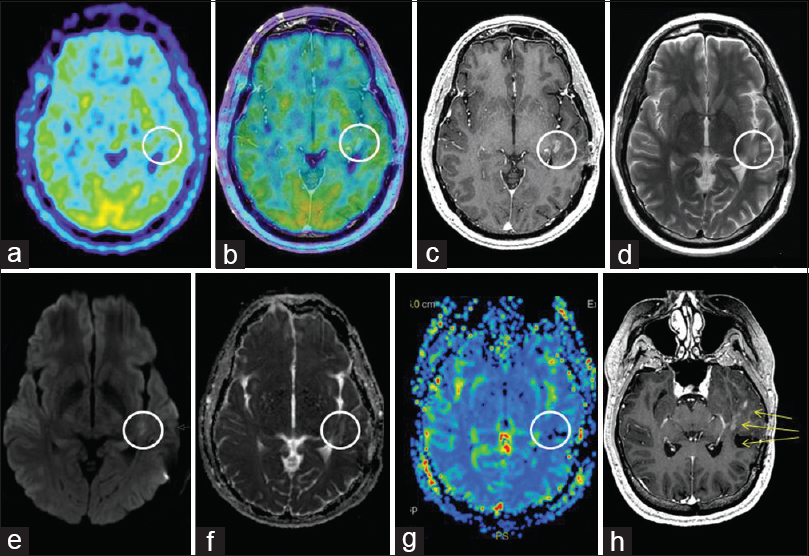
MET PET and MR imaging at 13 months: (a) Axial MET PET (b) axial MET PET + MRI CET1 (c) axial MRI CET1 (d) axial T2 (e) axial DWI (f) axial ADC (g) MR perfusion rCBV (h) reoriented CET1 demonstrating M5 and M6's proximity to the surgical area as well as new nodular rim enhancement of the surgical cavity
Extracranial tumor screening in November 2016 showed no recurrence. By the time of paper submission, the patient's clinical condition was assessed as good (KPS 90–100/RPA 1) without ongoing antitumoral therapy. The patient remains on low dose cortisone due to M5-M6's ARE. Further clinical and radiological follow up (including MRI and PET scans) are planned to carefully monitor M5-M6's evolution and identify further brain metastatic development.
DISCUSSION
The management of brain metastases remains complex and requires tailored treatment, often including microsurgery, different radiotherapeutic modalities, chemotherapy, as well as targeted therapy.[
Based on our institutional experience and previous reports,[
We have achieved similar results on a number of cases with large metastatic brain lesions with more common histopathological traits; a retrospective analysis covering the short and long-term outcome of RRR on these cases will be the subject of our next two papers.
CONCLUSION
In this particular case, Rapid Rescue Radiosurgery (RRR) proved highly effective in achieving next to comparable surgical decompression results on two large aggressive metastatic brain lesions. However, particular factors such as the tumor's histopathology/intrinsic radiosensitivity as well as RPA-surrogate factors could have played a substantial role in the outcome of the treatment. We believe this procedure has the potential to become an important surgical tool in the future management of large unresectable metastases. Retrospective analysis of all cases treated with RRR as well as further prospective studies are warranted and indeed ongoing.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Al-Barbarawi M, Smith SF, Sekhon LH. Haemorrhagic brain metastasis from a thymic carcinoma. J Clin Neurosci. 2004. 11: 190-4
2. Al-Omair A, Soliman H, Xu W, Karotki A, Mainprize T, Phan N. Hypofractionated stereotactic radiotherapy in five daily fractions for post-operative surgical cavities in brain metastases patients with and without prior whole brain radiation. Technol Cancer Res Treat. 2013. 12: 493-9
3. Asao C, Korogi Y, Kitajima M, Hirai T, Baba Y, Makino K. Diffusion-weighted imaging of radiation-induced brain injury for differentiation from tumor recurrence. AJNR Am J Neuroradiol. 2005. 26: 1455-60
4. Asao T, Fujiwara Y, Sunami K, Kitahara S, Goto Y, Kanda S. Medical treatment involving investigational drugs and genetic profile of thymic carcinoma. Lung Cancer. 2016. 93: 77-81
5. Barajas RF, Chang JS, Sneed PK, Segal MR, McDermott MW, Cha S. Distinguishing recurrent intra-axial metastatic tumor from radiation necrosis following gamma knife radiosurgery using dynamic susceptibility-weighted contrast-enhanced perfusion MR imaging. AJNR Am J Neuroradiol. 2009. 30: 367-72
6. D’Souza MM, Sharma R, Jaimini A, Panwar P, Saw S, Kaur P. 11C-MET PET/CT and advanced MRI in the evaluation of tumor recurrence in high-grade gliomas. Clin Nucl Med. 2014. 39: 791-8
7. Deng SM, Zhang B, Wu YW, Zhang W, Chen YY. Detection of glioma recurrence by (1)(1) C-methionine positron emission tomography and dynamic susceptibility contrast-enhanced magnetic resonance imaging: A meta-analysis. Nucl Med Commun. 2013. 34: 758-66
8. Eaton BR, Gebhardt B, Prabhu R, Shu HK, Curran WJ, Crocker I. Hypofractionated radiosurgery for intact or resected brain metastases: Defining the optimal dose and fractionation. Radiat Oncol. 2013. 8: 135-
9. Filosso PL, Guerrera F, Rendina AE, Bora G, Ruffini E, Novero D. Outcome of surgically resected thymic carcinoma: A multicenter experience. Lung Cancer. 2014. 83: 205-10
10. Fowler JF. 21 years of biologically effective dose. Br J Radiol. 2010. 83: 554-68
11. Iorio-Morin C, Masson-Cote L, Ezahr Y, Blanchard J, Ebacher A, Mathieu D. Early Gamma Knife stereotactic radiosurgery to the tumor bed of resected brain metastasis for improved local control. J Neurosurg. 2014. 121: 69-74
12. Ishihara T, Yamada K, Harada A, Isogai K, Tonosaki Y, Demizu Y. Hypofractionated stereotactic radiotherapy for brain metastases from lung cancer: Evaluation of indications and predictors of local control. Strahlenther Onkol. 2016. 192: 386-93
13. Kocher M, Wittig A, Piroth MD, Treuer H, Seegenschmiedt H, Ruge M. Stereotactic radiosurgery for treatment of brain metastases. A report of the DEGRO Working Group on Stereotactic Radiotherapy. Strahlenther Onkol. 2014. 190: 521-32
14. Le Rhun E, Dhermain F, Vogin G, Reyns N, Metellus P. Radionecrosis after stereotactic radiotherapy for brain metastases. Expert Rev Neurother. 2016. 16: 903-14
15. Martens B, Janssen S, Werner M, Fruhauf J, Christiansen H, Bremer M. Hypofractionated stereotactic radiotherapy of limited brain metastases: A single-centre individualized treatment approach. BMC Cancer. 2012. 12: 497-
16. Minamimoto R, Saginoya T, Kondo C, Tomura N, Ito K, Matsuo Y. Differentiation of Brain Tumor Recurrence from Post-Radiotherapy Necrosis with 11C-Methionine PET: Visual Assessment versus Quantitative Assessment. PLoS One. 2015. 10: e0132515-
17. Minniti G, Scaringi C, Paolini S, Lanzetta G, Romano A, Cicone F. Single-Fraction Versus Multifraction (3×9 Gy) Stereotactic Radiosurgery for Large (>2 cm) Brain Metastases: A Comparative Analysis of Local Control and Risk of Radiation-Induced Brain Necrosis. Int J Radiat Oncol Biol Phys. 2016. 95: 1142-8
18. Mohammadi AM, Recinos PF, Barnett GH, Weil RJ, Vogelbaum MA, Chao ST. Role of Gamma Knife surgery in patients with 5 or more brain metastases. J Neurosurg. 2012. 117: 5-12
19. Mohammadi AM, Schroeder JL, Angelov L, Chao ST, Murphy ES, Yu JS. Impact of the radiosurgery prescription dose on the local control of small (2 cm or smaller) brain metastases. J Neurosurg. 2017. 126: 735-43
20. Momose T, Nariai T, Kawabe T, Inaji M, Tanaka Y, Watanabe S. Clinical benefit of 11C methionine PET imaging as a planning modality for radiosurgery of previously irradiated recurrent brain metastases. Clin Nucl Med. 2014. 39: 939-43
21. Navarria P, Pessina F, Cozzi L, Ascolese AM, De Rose F, Fogliata A. Hypo-fractionated stereotactic radiotherapy alone using volumetric modulated arc therapy for patients with single, large brain metastases unsuitable for surgical resection. Radiat Oncol. 2016. 11: 76-
22. Nicolato A, Ferraresi P, Bontempini L, Tomazzoli L, Magarotto R, Gerosa M. Multiple brain metastases from “lymphoepithelioma-like” thymic carcinoma: A combined stereotactic-radiosurgical approach. Surg Neurol. 2001. 55: 232-4
23. Niranjan A, Lunsford LD. Gamma Knife Radiosurgery for 5 to 10 Brain Metastases: A Good Option for Upfront Treatment. Oncology. 2016. 30: 314-5, 7
24. Ojerholm E, Lee JY, Thawani JP, Miller D, O’Rourke DM, Dorsey JF. Stereotactic radiosurgery to the resection bed for intracranial metastases and risk of leptomeningeal carcinomatosis. J Neurosurg. 2014. 121: 75-83
25. Owen S, Souhami L. The management of brain metastases in non-small cell lung cancer. Front Oncol. 2014. 4: 248-
26. Powell JW, Chung CT, Shah HR, Canute GW, Hodge CJ, Bassano DA. Gamma Knife surgery in the management of radioresistant brain metastases in high-risk patients with melanoma, renal cell carcinoma, and sarcoma. J Neurosurg. 2008. 109: 122-8
27. Shuryak I, Carlson DJ, Brown JM, Brenner DJ. High-dose and fractionation effects in stereotactic radiation therapy: Analysis of tumor control data from 2965 patients. Radiother Oncol. 2015. 115: 327-34
28. Sinclair G, Bartek J, Martin H, Barsoum P, Dodoo E. Adaptive hypofractionated gamma knife radiosurgery for a large brainstem metastasis. Surg Neurol Int. 2016. 7: S130-8
29. Soliman H, Das S, Larson DA, Sahgal A. Stereotactic radiosurgery (SRS) in the modern management of patients with brain metastases. Oncotarget. 2016. 7: 12318-30
30. Tamura Y, Kuroiwa T, Doi A, Min KY. Thymic carcinoma presenting as cranial metastasis with intradural and extracranial extension: Case report. Neurosurgery. 2004. 54: 209-11
31. Terakawa Y, Tsuyuguchi N, Iwai Y, Yamanaka K, Higashiyama S, Takami T. Diagnostic accuracy of 11C-methionine PET for differentiation of recurrent brain tumors from radiation necrosis after radiotherapy. J Nucl Med. 2008. 49: 694-9
32. Thomas de Montpreville V, Ghigna MR, Lacroix L, Besse B, Broet P, Dartevelle P. Thymic carcinomas: Clinicopathologic study of 37 cases from a single institution. Virchows Arch. 2013. 462: 307-13
33. Thompson EM, Sather MD, Reyes CA, Long DJ. Intracranial leptomeningeal metastasis from thymic carcinoma: Case report and review. Surg Neurol. 2007. 68: 233-8
34. Tsuyuguchi N, Sunada I, Iwai Y, Yamanaka K, Tanaka K, Takami T. Methionine positron emission tomography of recurrent metastatic brain tumor and radiation necrosis after stereotactic radiosurgery: Is a differential diagnosis possible?. J Neurosurg. 2003. 98: 1056-64
35. Xu JL, Li YL, Lian JM, Dou SW, Yan FS, Wu H. Distinction between postoperative recurrent glioma and radiation injury using MR diffusion tensor imaging. Neuroradiology. 2010. 52: 1193-9
36. Yamamura K, Kubo O, Aoki N, Kagawa M. Falx metastasis of thymic carcinoma: A case report and review of literature. No Shinkei Geka. 1993. 21: 921-4
37. Zada G, Yu C, Pagnini PG, Khalessi AA, Zelman V, Apuzzo ML. Early decreased tumor volume following fractionated Gamma Knife Radiosurgery for metastatic melanoma and the role of “adaptive radiosurgery”: Case report. Neurosurgery. 2010. 67: E512-3