- Department of Neurosurgery, Riverside University Health Systems Medical Center, Moreno Valley, California, USA
- Department of Neurosurgery, Arrowhead Regional Medical Center, Colton, California, USA
Correspondence Address:
Tyler Carson
Department of Neurosurgery, Riverside University Health Systems Medical Center, Moreno Valley, California, USA
Department of Neurosurgery, Arrowhead Regional Medical Center, Colton, California, USA
DOI:10.4103/sni.sni_291_16
Copyright: © 2017 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Tyler Carson, Dan Miulli. Bedside twist drill aspiration of cerebral abscess less than 2.5 cm in size: A case series and discussion. 26-Sep-2017;8:230
How to cite this URL: Tyler Carson, Dan Miulli. Bedside twist drill aspiration of cerebral abscess less than 2.5 cm in size: A case series and discussion. 26-Sep-2017;8:230. Available from: http://surgicalneurologyint.com/surgicalint-articles/bedside-twist-drill-aspiration-of-cerebral-abscess-less-than-2-5-cm-in-size-a-case-series-and-discussion/
Abstract
Background:Intracranial abscess remains a potentially deadly condition despite development of newer antibiotics and improved surgical methods. Many studies have evaluated the surgical indications for abscess drainage, and it has been generally accepted that intracranial abscesses greater than 2.5 cm may best be treated with surgical intervention followed by antibiotic therapy. More recently, studies have shown good results with stereotactic aspiration of abscesses to 1 cm in size. Furthermore, a recent case series in 2014 of 103 cases of bedside twist drill aspiration of cerebral abscess >2.5 cm showed a good recovery in 83.4% of cases.
Case Description:This case series examines the benefits of bedside twist drill aspiration of peripherally located brain abscess less than 2.5 cm in size. In our series, all patients were placed on broad-spectrum antibiotics and had negative blood and cerebrospinal fluid cultures. Our bedside biopsy resulted in de-escalation of antibiotics in 2 of the 3 patients and decreased hospital length of stay.
Conclusion:In patients with peripherally located brain abscesses less than 2.5 cm in size, bedside twist drill drainage may be a safe and reasonable approach. Positive identification of infective pathogen allows for a decreased hospital length of stay and de-escalation of antibiotics regiment resulting in significant reduction of hospital costs and resources in 2 of the 3 patients treated. This is of benefit to the patient as well as the health system.
Keywords: Abscess, aspiration, bedside, brain, evacuation, surgical
BACKGROUND
Intracranial abscess remains a potentially deadly condition despite development of newer antibiotics and improved surgical methods. Cerebral abscess affects 1 in 10,000 hospital admissions or 1,500 to 2,500 people per year in the US.[
Presenting symptoms have classically been described as a triad of fever, headache, and nausea, however, this classic triad is present in on 20% of the patients.[
Treatment for cerebral abscess relies on both surgical and nonsurgical approaches. The classic study by Rosenblum et al. showed that patients may be successfully treated nonsurgically with small abscess.[
The two main surgical options for brain abscess are excision versus aspiration. A retrospective comparison between the two approaches between 1990 and 2008 performed by Ratnaike et al. showed a 6.6% mortality for patients who underwent aspiration compared to a 12.7% mortality for those undergoing excision of abscess.[
This articles examines the role for aspiration of cerebral abscess via twist drill craniotomy, specifically for patients with abscess less than 2.5 cm in size and in whom no source had been identified. A recent case series by Singh et al. details their experience with 103 cases of pyogenic brain abscess >2.5 cm in size that were treated with aspiration via twist drill craniotomy.[
CASE DESCRIPTION
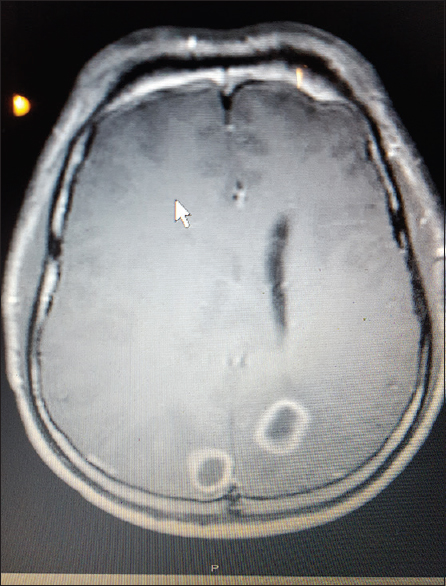
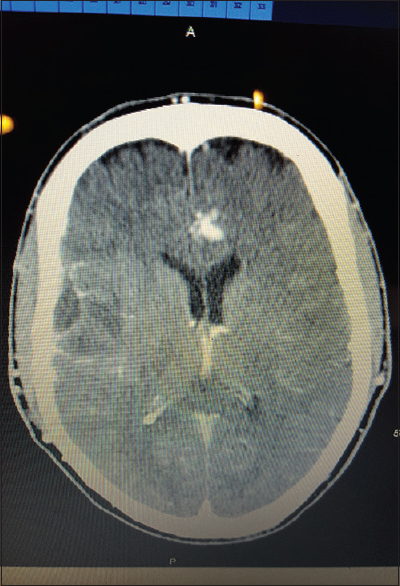
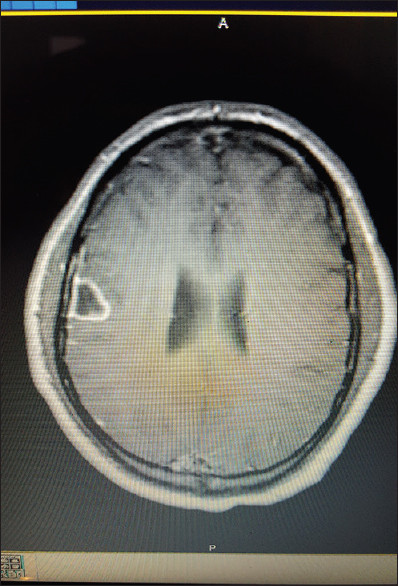
All patients received computed tomography (CT) of the head upon admission with a significant finding of lesion(s) concerning for intracranial abscess based on patient history. Using CT reconstructions and imaging software, the entry point for each biopsy site was calculated using fixed boney anatomy. Entry point was selected to be orthogonal to skull surface and traversing as little cortex as possible. Specifically, the entry point was calculated by measuring the distance above the external auditory meatus (EAM), the distance posterior from the Nasion corresponding to 90-degree angle to the entry site and the distance from the midline to the entry point. This site was then clipped of hair and marked with a 1 cm × 1 cm square metal marker and in one case an MR-Spot® marker. Follow-up imaging after marker placement confirmed entry point, adjustments to entry point were made as necessary.
Another option would be bedside stereotactic navigation, however, it was not used in these 3 cases. In our institution, we used AxiEM™ electromagnetic navigation for bedside localization of hematomas and lesions. The benefits include possible improved accuracy and real-time adjustment of trajectory with image feedback. The downside is its availability. There is also expense associated with the equipment use and possibly additional imaging studies needed.
After entry point was confirmed we proceeded with surgical aspiration of the abscess at bedside using a twist drill method. Patients received versed preoperatively and local anesthetic of 5–10 cc of 1% lidocaine with epinephrine. A 2-cm linear incision was made and the hand twist drill was utilized to access the intracranial space. Dura was palpated and opened with an 11 blade, and a brain needle (Dandy ventricular needle) was advanced orthogonal to the skull to a depth corresponding the abscess depth noted on CT/MRI imaging. Fluid from abscess was aspirated and sent for pathology and microbiology studies. The incision was then closed with suture or staple and patient was sent for a postoperative CT to confirm that there were no hemorrhage and correct site was accessed.
All patients presented with headache. 2/3 presented with fever. One presented with seizure. The results of each individual patient are detailed in
Of the 3 patients who received bedside twist drill aspiration of cerebral abscess, a positive identification of the infective agent was made in 2 cases. This was despite each having had blood cultures drawn prior to antibiotic therapy and both having had lumbar punctures performed with negative cultures. In addition, each had already been placed on vancomycin, flagyl, and ceftriaxone prior to biopsy and identification of infective agent.
Based on the final MIC/MBC results from cultures, both patients 2 and 3 were able to have their antibiotics de-escalated to a single agent (vancomycin in both cases) per infectious disease recommendations rather than broad-spectrum coverage of vancomycin flagyl and fortaz. Both patients were discharged with a PICC line and home antibiotic therapy. This resulted in decreased cost to the patient and health system in terms of fewer medications, fewer trips for home health to administer IV medications, and less inpatient hospital days. Home antibiotic regiment would not have been possible with more than 2 infusions per day which would have required the patient to be placed in a skilled nursing facility rather than discharging home. Upon follow-up, both patients had good outcomes without any residual neurologic deficit or return of abscess.
Patient 1 did not have a positive result from biopsy of suspected abscess and underwent open craniotomy for evacuation and tissue biopsy. Of note, this patient had multiple intracranial lesions and known cavitary lung lesion. The results from a lung biopsy as well as open biopsy of brain were negative for malignant tissue or infection. Patient was discharged to inpatient rehab and referred to outside hospital for rheumatology and infectious disease work-up.
CONCLUSION
We show in this small series of patients that peripherally located brain abscesses less than 2.5 cm in size and peripherally located may be safely treated with bedside twist drill aspiration. We did not have a lower size limit to treat though abscesses less than 0.5 cm in size may be difficult to access and/or obtain material from. Positive identification of infective pathogen allowed for a decreased hospital length of stay and de-escalation of antibiotics regiment in 2 of our 3 patients resulting in significant reduction of hospital costs and resources. The ability to perform this procedure at the bedside rather than utilizing valuable operating room time and expense as well as subjecting the patient to the risks of general anesthesia may also be of benefit to the patient as well as the health system.
We intend to use this method of bedside twist drill aspiration in certain situations at our institution. The ability to safely obtain a diagnosis in patients who may otherwise be treated solely with broad-spectrum antibiotic therapy for many weeks may be of benefit. Furthermore, at an extremely busy county medical center where resources are limited, the ability to perform this at bedside can be tremendously helpful for maintaining hospital resources.
Though no adverse outcomes were seen in this limited series of 3 patients, more data is needed before determining whether this approach is safe and effective. In addition, we realized a decreased hospital stay and antibiotic regiment in 2 patients, but again more patient outcomes are needed to determine whether this results in any significant benefit to the patient or health system.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Alvis Miranda H, Castellar-Leones SM, Elzain MA, Moscote-Salazar LR. Brain abscess: Current management. J Neurosci Rural Pract. 2013. 4: S67-S81
2. Brouwer MC, Coutinho JM, van de Beek D. Clinical characteristics and outcome of brain abscess: Systematic review and meta-analysis. Neurology. 2014. 82: 806-13
3. Brouwer MC, Tunkel AR, McKhann GM, van de Beek D. Brain Abscess. N Engl J Med. 2014. 371: 447-56
4. Duma CM, Kondziolka D, Lunsford LD. Image-guided stereotactic management of non-AIDS-related cerebral infection. Neurosurg Clin N Am. 1992. 3: 291-302
5. Garvey G. Current concepts of bacterial infections of the central nervous system. Bacterial meningitis and bacterial brain abscess. J Neurosurg. 1983. 59: 735-44
6. Greenberg MS.editors. Cerebral Abscess. Handbook of Neurosurgery. New York: Thieme Publishers; 2010. p. 350-6
7. Hakan T. Management of bacterial brain abscess. Neurosurg Focus. 2008. 24: E4-
8. Helweg-Larsen J, Astradsson A, Richhall H, Erdal J, Laursen A, Brennum J. Pyogenic brain abscess, a 15 year survey. BMC Infect Dis. 2012. 12: 332-
9. Kariev MK, Kadyrbekov RT, Akhmediev MM, Akmedov SC, Khuzhaniiazov SB. Comparative analysis of surgical methods in the treatment of brain abscesses. Zh Vopr Neirokhir Im N N Burdenko. 2001. 2: 17-20
10. Leuthardt EC, Wippold FJ, Oswood MC, Rich KM. Diffusion-weighted MR imaging in the preoperative assessment of brain abscesses. Surg Neurol. 2002. 58: 395-402
11. Lu CH, Chang WN, Lui CC. Strategies for the management of bacterial brain abscess. J Clin Neurosci. 2006. 13: 979-85
12. Rosenblum ML, Hoff JT, Norman D, Edwards MS, Berg BO. Nonoperative treatment of brain abscesses in selected high-risk patients. J Neurosurg. 1980. 52: 217-25
13. Mathisen GE, Johnson JP. Brain abscess. Clin Infect Dis. 1997. 25: 763-79
14. Nathoo N, Nadvi SS, Narotam PK, van Dellen JR. Brain abscess: Management and outcome analysis of a computed tomography era experience with 973 patients. World Neurosurg. 2011. 75: 716-26
15. Obana WG, Rosenblum ML. Nonoperative treatment of neurosurgical infections. Neurosurg Clin N Am. 1992. 3: 359-73
16. Ratnaike TE, Das S, Gregson BA, Mendelow AD. A review of brain abscess surgical treatment – 78 years: Aspiration versus excision. World Neurosurg. 2011. 76: 431-6
17. Sharma BS, Gupta SK, Khosla VK. Current concepts in the management of pyogenic brain abscess. Neurol India. 2000. 48: 105-
18. Singh I, Rohilla S, Kumawat M. Twist drill aspiration of pyogenic brain abscesses: Our experience in 103 cases. J Neurol Surg. 2014. 74: 189-94
19. Stevens DC, Asfora WT. 8-year-old patient with multipule large cerebral abscesses successfully treated with stereotactic aspiration: Case report and literature review. S D Med. 2013. 66: 420-3
20. Garvey L, Winston A, Walsh J, Post F, Porter K. HIV-associated central nervous system diseases in the recent combination antiretroviral therapy era. Eur J Neurol. 2011. 18: 527-34
21. Zhai Y, Wei X, Chen R, Guo Z, Raj Singh R, Zhang Y. Surgical outcome of encapsulated brain abscess in superficial non-eloquent area: A systematic review. Br J Neurosurg. 2016. 30: 29-34