- Department of Neurosurgery, St. Marianna University School of Medicine, Kawasaki, Japan
Correspondence Address:
Hiroshi Takasuna
Department of Neurosurgery, St. Marianna University School of Medicine, Kawasaki, Japan
DOI:10.4103/sni.sni_283_17
Copyright: © 2017 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Hiroshi Takasuna, Taigen Sase, Hidemichi Ito, Hajime Ono, Kotaro Oshio, Yuichiro Tanaka. Clinical significance of thickened sphenoid sinus mucosa in Rathke's cleft cyst. 01-Nov-2017;8:263
How to cite this URL: Hiroshi Takasuna, Taigen Sase, Hidemichi Ito, Hajime Ono, Kotaro Oshio, Yuichiro Tanaka. Clinical significance of thickened sphenoid sinus mucosa in Rathke's cleft cyst. 01-Nov-2017;8:263. Available from: http://surgicalneurologyint.com/surgicalint-articles/clinical-significance-of-thickened-sphenoid-sinus-mucosa-in-rathkes-cleft-cyst/
Abstract
Background:To determine the clinical significance of thickening of the sphenoid sinus mucosa (TSSM) in Rathke's cleft cyst (RCC).
Methods:We retrospectively reviewed patients with pituitary lesions. A total of 99 patients, who underwent surgery of pituitary lesions between 2008 and 2015, were analyzed to evaluate the clinical significance of TSSM using magnetic resonance imaging. The patients with TSSM were evaluated for their characteristics.
Results:Of the 99 operations, 15 patients with paranasal sinusitis, direct invasion to the sellar floor, and/or a history of transsphenoidal surgery were excluded. There were 51 pituitary adenomas (PAs), 18 RCCs, and 15 other tumors. TSSM was observed in 6 patients (7.1%). Pathologies included 3 RCCs (16.7%), 1 PA (2.0%), and 2 other lesions. Three RCCs were especially analyzed. The pituitary dysfunction was found in all 3 patients with TSSM, whereas that was noted in 2 of 15 patients (13.3%) without TSSM. The sensitivity was 60%, specificity was 86.7%, and the positive predictive value was 100%. There was no significant difference in the age, thickness of TSSM, symptoms, and squamous metaplasia between patients with and without TSSM.
Conclusions:TSSM occurs more frequently in RCCs than in PAs. TSSM can potentially suggest hypopituitarism in RCCs. The surgical and/or endocrinological interventions are required for preventing further endocrine deterioration.
Keywords: Hypophysitis, hypopituitarism, mucosal thickening, Rathke's cleft cyst sphenoid sinus
INTRODUCTION
Rathke's cleft cyst (RCC) is a nonneoplastic lesion, which is considered to arise from the remnants of Rathke's pouch, an invagination of the stomodeum. This lesion is often clinically silent all through the person's lifetime. If an RCC enlarges to affect surrounding structures such as the pituitary gland and/or the optic chiasm, headache, visual disturbance, and endocrine insufficiency are common presenting features. However, a point of controversy remains regarding the natural history, surgical indication, and recurrence; and it is difficult to determine the therapeutic intervention. Hormonal dysfunction is not uncommon but subclinical in many cases, which may lead to delayed diagnosis.[
MATERIALS AND METHODS
An exhaustive retrospective analysis was performed among 99 patients in whom pituitary and sellar lesions were diagnosed at our institution between 2008 and 2015. The presence of TSSM was determined with MRI. The thickness of the sphenoid sinus mucosa adjacent to the sellar floor was measured on T1- or Gadolinium T1- and T2-weighted images. A mucosal thickness of more than 1 mm was considered abnormal, as previously described.[
Statistical analysis
The distributed data are shown as the means. Mann–Whitney U test was used to analyze correlations between two groups. A P value of <0.05 was considered to indicate statistical significance. The sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) were calculated for the relative factor of TSSM.
RESULTS
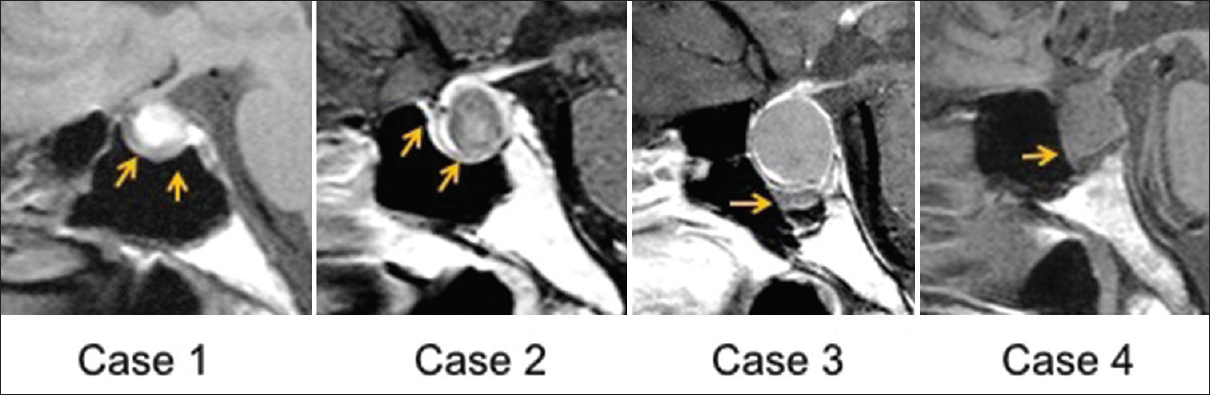
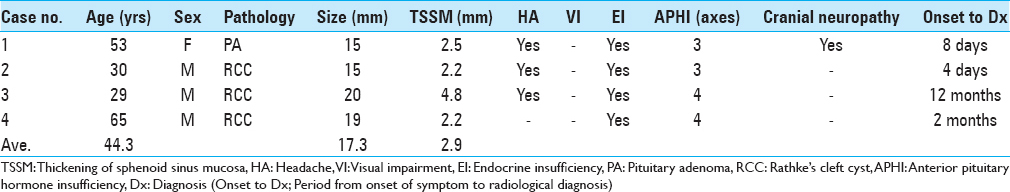
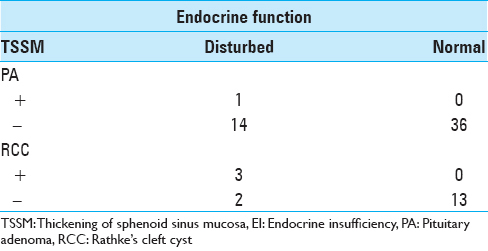
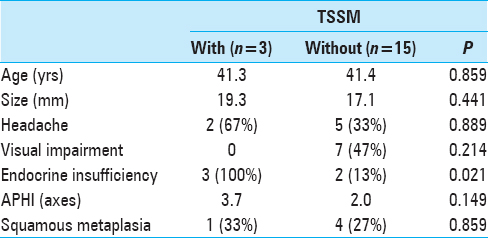
Between 2008 and 2015, we operated on 99 patients with pituitary lesions. Fifteen patients had paranasal sinusitis, direct invasion, and/or a history of transsphenoidal surgery. Among the remaining 84 patients, there were 36 men and 48 women whose mean age was 46.5 years (range 12–82 years). There were 51 PAs, the most common pathology identified, followed by 18 RCCs, and 15 other tumors (10 craniopharyngiomas, 2 germinomas, 2 xanthogranulomas, and 1 arachnoid cyst). TSSM was observed in 6 patients (7.1%) on MRI. The pathologies included 3 RCCs, 1 PA, and 2 other tumors. The patients’ mean age was 40.8 years, and the mean thickness of the mucosa adjacent to the sellar floor was 3.1 mm. Transsphenoidal surgery was performed and histological findings of the sphenoid sinus mucosa were examined in all 6 cases. The frequency of TSSM without tumor invasion proven by histological examination was 6 of 84 cases (7.1%). We compared the PAs and RCCs; and TSSM was observed in 2.0% (1 of 51) of PAs (Case 1) [
Figure 1
Thickening of sphenoid sinus mucosa (TSSM) is seen on magnetic resonance image (MRI) in patients with pituitary adenomas or Rathke's cleft cysts. Sagittal planes of MRIs show the thickened sphenoid sinus mucosa adjacent to the sellar floor (arrows). Case numbers correspond to those in
Illustrative case (Case 3)
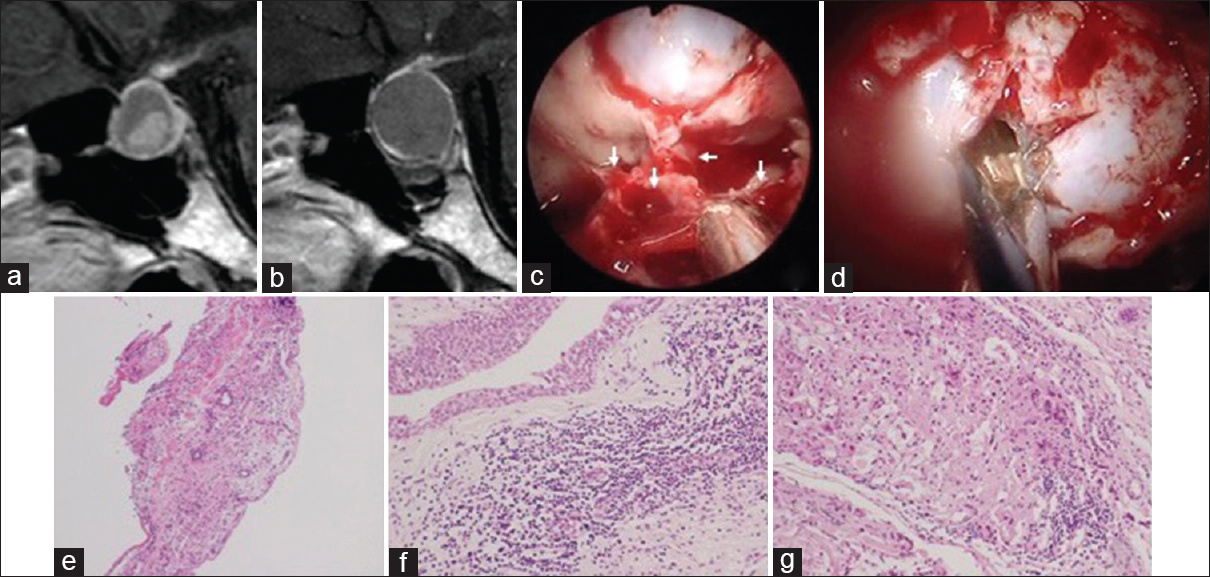
A 28-year-old man had severe headache especially retro-orbital pain. MRI revealed a 16-mm cyst in the intra- and suprasellar region. This cyst appeared as a slightly high intensity on T1-weighted image, high intensity on T2-weighted image, and without enhancement on gadolinium-enhanced T1-weighted image. An intracystic nodule can be seen which was most likely an RCC [
Figure 2
An MRI 1 year before (a) and just before the operation showing the TSSM (b). The endoscopic view revealed a thickening of the mucosa over the sellar floor (white arrows) (c), and yellowish jelly-like fluid in the cyst (d). Hematoxylin and eosin staining of the sphenoid sinus mucosa shows inflammatory cells (e). The columnar epithelium with cilia and stratified squamous epithelium, infiltration of inflammatory cells adjacent to the cyst wall were noted in the extracted cyst wall (f). Chronic inflammatory cells are found in the pituitary gland (g)
DISCUSSION
TSSM has been reported during the acute stage of pituitary apoplexy. TSSM was observed in 82%[
In some reports, MRI in patients with noninfectious granulomatous hypophysitis showed swelling of the mucosa in the sphenoid sinus.[
Hypophysitis can be classified as primary or secondary. Primary hypophysitis is very rare, and the incidence among pituitary pathologies is between 0.2% and 6.5%.[
In the present study, histological investigation showed inflammatory cells such as plasmacytes and lymphocytes in the pituitary gland around the RCCs. Infiltrations of inflammatory cells were also observed in the mucosa of the sphenoid sinus adjacent to the sellar floor in the surgical specimens in the present study. This inflammatory change suggested that the secondary inflammation caused by the RCC was extended over the sphenoid sinus mucosa. Among 3 RCCs with TSSM, the period from the development of symptoms to diagnosis was shorter than 2 months in 2 cases (Cases 2 and 4) and the other was in 12 months (Case 3). These two kinds of onset patterns suggested the possibility of two kinds of mechanisms. Hama et al. described that secondary inflammation due to the RCC causes epithelium to stratify and the inflammation spreads into the subjacent and overwhelm the hypophysis, resulting in hypopituitarism.[
Transsphenoidal surgery typically remains the preferred option for patients with symptomatic RCC and offers excellent outcomes with regard to symptomatic improvement, preservation of normal pituitary function, and minimization of complications. In 18 cases in the present study, worsening of the pituitary function, except for case 3, was not seen after the surgery. The existence of TSSM suggests clinical or subclinical hypopituitarism, endocrine insufficiency does not cause TSSM because of high NPV. Therefore, coexistence of TSSM and hypopituitarism is helpful for diagnosing RCC with pituitary inflammation. Surgical treatment is advisable to prevent further functional degradation. Endocrinological intervention is also required as soon as possible for hormonal evaluation and subsequent replacement therapy.
CONCLUSIONS
Retrospective analysis of MR images in patients with a pituitary lesion demonstrated that TSSM was observed in 16.7% of RCCs. Pituitary lesions with TSSM, especially with an RCC, are likely to already have exhibited pituitary dysfunction. TSSM may suggest hypopituitarism in RCCs. Transsphenoidal surgical intervention should be considered to prevent further endocrine deterioration, and hormone replacement therapy is also recommended as soon as possible.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Agrawal B, Dziurzynski K, Salamat MS, Baskaya M. The temporal association of sphenoid sinus mucosal thickening on MR imaging with pituitary apoplexy. Turk Neurosurg. 2012. 22: 785-90
2. Arita K, Kurisu K, Tominaga A, Sugiyama K, Ikawa F, Yoshioka H. Thickening of sphenoid sinus mucosa during the acute stage of pituitary apoplexy. J Neurosurg. 2001. 95: 897-901
3. Caturegli P, Newschaffer C, Olivi A, Pomper MG, Burger PC, Rose NR. Autoimmune hypophysitis. Endocr Rev. 2005. 26: 599-614
4. Demetri C, Shoukri KC, Taylor SL, Silva JE. Postpartum granulomatous hypophysitis with sphenoid sinus involvement: A case study. Endocr Pract. 2010. 16: 264-9
5. Eguchi K, Uozumi T, Arita K, Kurisu K, Yano T, Sumida M. Pituitary function in patients with Rathke's cleft cyst: Significance of surgical management. Endocr J. 1994. 41: 535-40
6. Gazioǧlu N, Tüzgen S, Oz B, Koçer N, Kafadar A, Akar Z. Idiopathic granulomatous hypophysitis: Are there reliable, constant radiological and clinical diagnostic criterias?. Neuroradiol. 2000. 42: 890-4
7. Hama S, Arita K, Nishisaka T, Fukuhara T, Tominaga A, Sugiyama K. Changes in the epithelium of Rathke cleft cyst associated with inflammation. J Neurosurg. 2002. 96: 209-16
8. Honegger J, Buchfelder M, Schlaffer S, Droste M, Werner S, Strasburger C. Pituitary Working Group of the German Society of Endocrinology. Treatment of primary hypophysitis in Germany. J Clin Endocrinol Metab. 2015. 100: 3460-9
9. Honegger J, Fahlbush R, Bornmann A, Hensen J, Buchfelder M, Müller M. Lymphocytic and granulomatous hypophysitis: Experience with nine cases. Neurosurgery. 1997. 40: 713-23
10. Honegger J, Schlaffer S, Menzel C, Droste M, Werner S, Elbelt U. Pituitary Working Group of the German Society of Endocrinology. Diagnosis of primary hypophysitis in Germany. J Clin Endocrinol Metab. 2015. 100: 3841-9
11. Liu JK, Couldwell WT. Pituitary apoplexy in the magnetic resonance imaging era: Clinical significance of sphenoid sinus mucosal thickening. J Neurosurg. 2006. 104: 892-8
12. Nishioka H, Haraoka J, Izawa H, Ikeda Y. Magnetic resonance imaging, clinical manifestations, and management of Rathke's cleft cyst. Clin Endocrinol (Oxf). 2006. 64: 184-8
13. Semple PL, Webb MK, de Villiers JC, Laws ER. Pituitary apoplexy. Neurosurgery. 2005. 56: 65-73