- Department of Otolaryngology, Johns Hopkins School of Medicine, Baltimore, Maryland, USA
- Department of Neurology, Johns Hopkins School of Medicine, Baltimore, Maryland, USA
Correspondence Address:
Bryan K. Ward
Department of Otolaryngology, Johns Hopkins School of Medicine, Baltimore, Maryland, USA
DOI:10.4103/sni.sni_442_15
Copyright: © 2017 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Bryan K. Ward, Angela Wenzel, Eva K. Ritzl, John P. Carey. Electrocochleography summating potential seen on auditory brainstem response in a case of superior semicircular canal dehiscence. 26-May-2017;8:90
How to cite this URL: Bryan K. Ward, Angela Wenzel, Eva K. Ritzl, John P. Carey. Electrocochleography summating potential seen on auditory brainstem response in a case of superior semicircular canal dehiscence. 26-May-2017;8:90. Available from: http://surgicalneurologyint.com/surgicalint-articles/electrocochleography-summating-potential-seen-on-auditory-brainstem-response-in-a-case-of-superior-semicircular-canal-dehiscence/
Abstract
Background:Superior canal dehiscence syndrome (SCDS) is a condition in which an abnormal communication between the superior semicircular canal and the middle cranial fossa causes patients to hear internal noises transmitted loudly to their affected ear as well as to experience vertigo with pressure changes or loud sounds. Patients with SCDS can have an elevated ratio of summating potential (SP) to action potential (AP) as measured by electrocochleography (ECochG). Changes in this ratio have been observed during surgical intervention to correct this abnormal communication.
Case Description:We present a case of SCDS along with history, physical examination, vestibular function testing, and computed tomography imaging. Due to the disabling symptoms, the patient elected to undergo surgery for plugging of the superior semicircular canal by middle cranial fossa approach. Simultaneous intraoperative ECochG and auditory brainstem response (ABR) were performed. Changes in SP/AP ratio, SP amplitude, and ABR wave I latency were observed during surgery, with a large ECochG SP amplitude generating a new wave, identifiable on the ABR and preceding the traditional wave I. The patient's symptoms resolved after surgery, and no long-term detriment to hearing was observed.
Conclusions:This case demonstrates the intraoperative changes in ECochG during surgery for repair of a SCDS. The substantial intraoperative changes in the summating potential can create a novel wave on intraoperative ABR.
Keywords: Electrocochleography, auditory brainstem response, superior canal dehiscence syndrome
INTRODUCTION
Superior canal dehiscence syndrome (SCDS) is a clinical syndrome described by Minor et al. in 1998, in which a hole in the bone overlying the superior semicircular canal causes a collection of symptoms including hearing the body's internal noises transmitted loudly (i.e., autophony), hearing one's own pulse (i.e., pulse-synchronous tinnitus), and sound or pressure-induced vertigo and nystagmus.[
Electrocochleography (ECochG) is an electrophysiologic test used for the diagnosis of endolymphatic hydrops, especially as seen in Ménière's disease.[
CASE REPORT
A 49-year-old woman presented with the chief complaint of “I can hear my eyes move.” Her symptoms included chronic dizziness, sensitivity to loud sounds, and autophony. Approximately 10 years prior to presentation, she began noticing that she would feel vertigo when near sirens, during applause in stadiums, or when the radio volume was high while driving in the car. She would experience momentary dizziness with coughing or straining, and could hear her heartbeat constantly in her left ear. In addition to hearing her eyes move, she heard her back cracking in her left ear and each swallow transmitted loudly to that ear. On physical examination, her ears appeared normal by otoscopy. She had normal vestibulo-ocular reflex function to rapid head impulses in the planes of each semicircular canal. Dix–Hallpike positioning maneuvers did not elicit any positional nystagmus or vertigo. Using infrared goggles, however, vertical nystagmus was seen in the plane of the left superior semicircular canal when 500 Hz and 750 Hz tones were applied to the left ear at 110 dB hearing level (HL). Simultaneous head movements in the plane of the left superior semicircular canal were also observed with application of these tones.
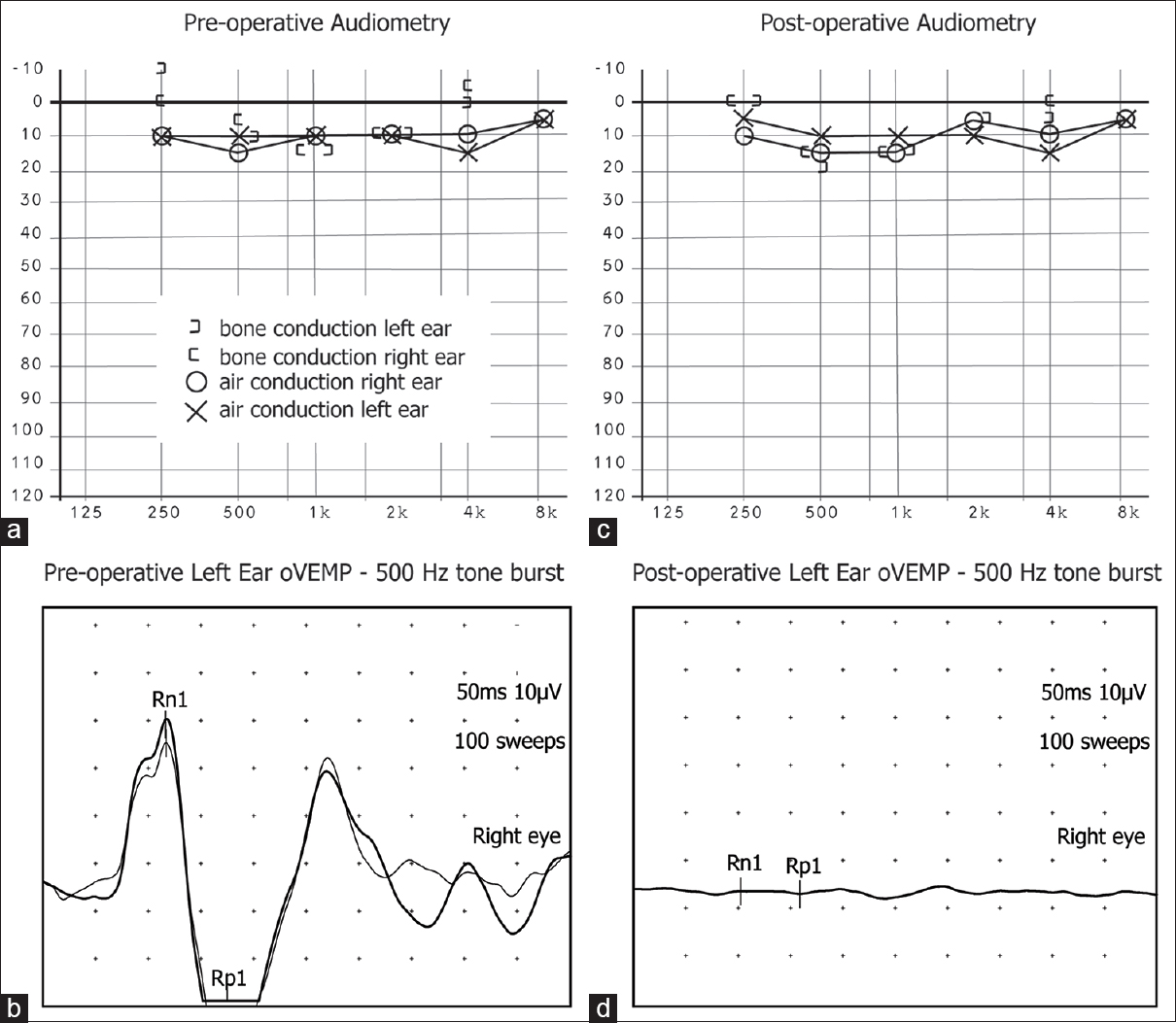
An audiogram showed normal hearing bilaterally, with a 20 dB gap between air conduction and bone conduction thresholds at 250 Hz in the left ear and a bone-conduction threshold of −10 dB HL indicating bone-conduction hyperacusis [
Figure 1
Preoperative audiometry and ocular VEMP data (a and b) demonstrated a low-frequency air-bone gap for the symptomatic left ear, as well as elevated ocular VEMP amplitudes in the contralateral eye in response to 500 Hz tone bursts in the symptomatic ear. Three months after surgery (c and d) the low-frequency air-bone gap decreases, without any sensorineural hearing loss, and the ocular VEMP amplitudes normalize, both consistent with the patient's resolution of symptoms
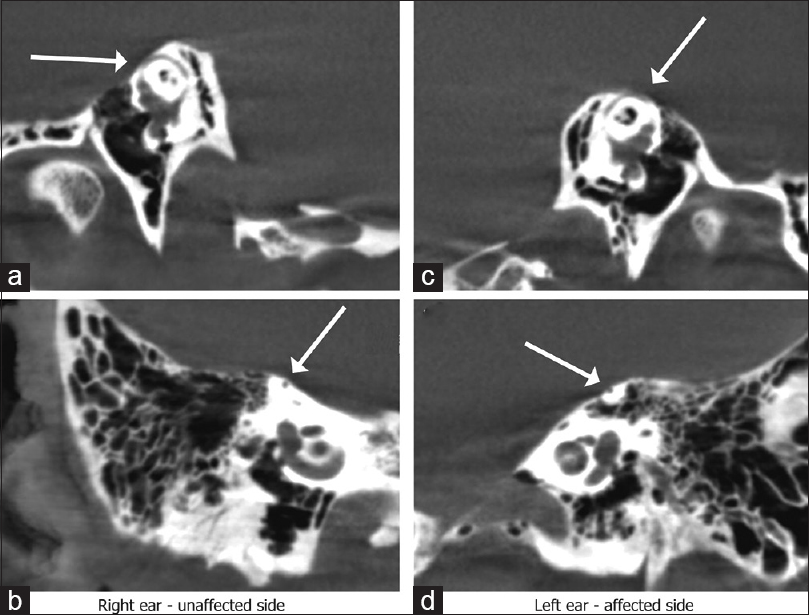
Figure 2
High-resolution computed tomography (CT) demonstrated a thin but intact covering of bone over the right superior semicircular canal on reformation in the plane of the affected canal (a) and orthogonal to that plane (b). A dehiscence was seen affecting the superior semicircular canal on the left side (c and d)
During surgery, ECochG and ABR were simultaneously recorded, and are shown in Figures
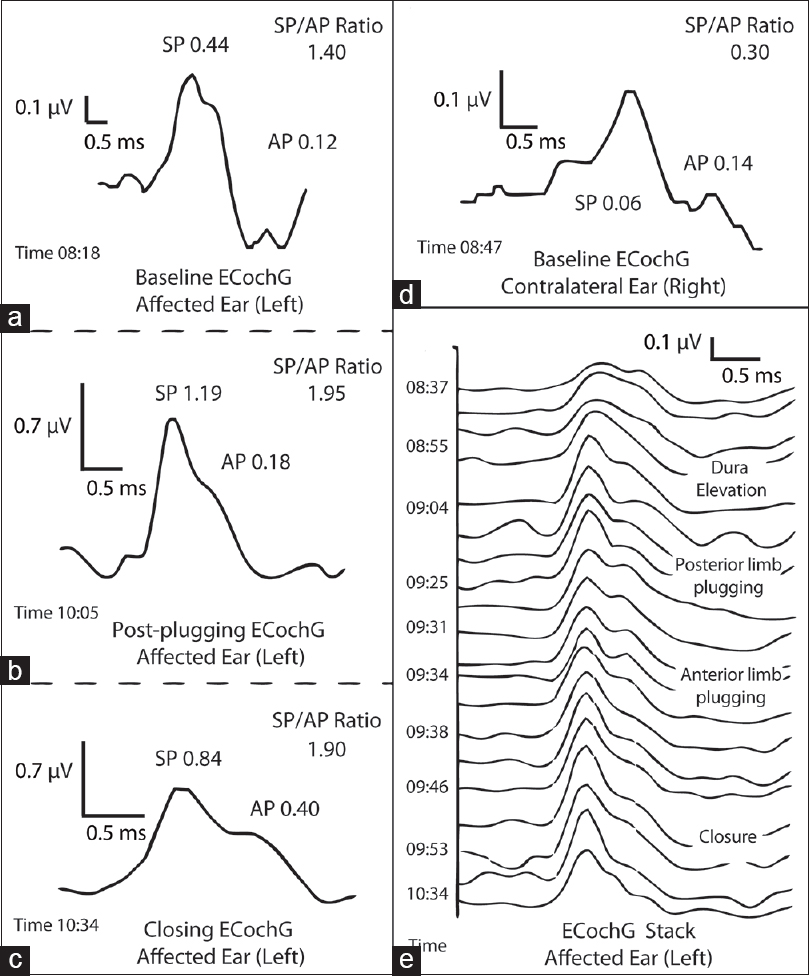
Figure 3
Intraoperative ECochG revealed significantly elevated SP/AP ratio that did not decrease after plugging of the affected superior semicircular canal. In fact, the elevated SP/AP ratio further increased during surgery. (a) Baseline ECochG prior to plugging, followed by (b) ECochG immediately after plugging the canal and (c) at the completion of surgery (each window is labeled with time of day). (d) Contralateral ear demonstrates a normal (<0.4) SP/AP ratio. (e) ECochG tracings stacked over an approximately 2-hour intraoperative time interval. Time of day and notable intraoperative events are labeled
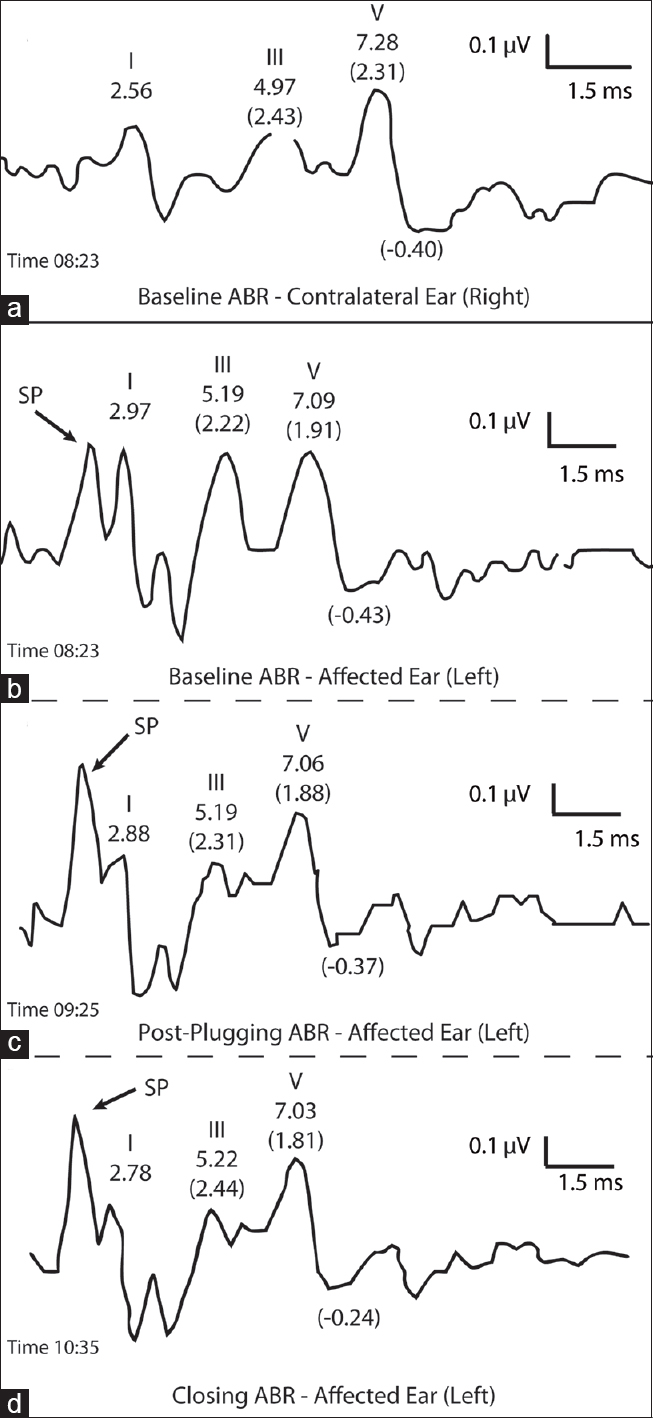
Figure 4
Intraoperative ABR is normal for the contralateral ear (a) and shows the appearance of a separate wave prior to wave I, and corresponding to the ECochG summating potential in the affected ear (b). Similar to the SP on ECochG, the new wave becomes elevated during the case and remains elevated at the conclusion of surgery (c and d, each window is labeled with time of day)
She recovered well from surgery, with complete resolution of her symptoms of autophony and sound or pressure-induced vertigo. As expected, she transiently had oscillopsia with head movements in the plane of the superior semicircular canal that symptomatically improved after vestibular physical therapy. Follow-up audiogram 3 months after surgery demonstrated resolution of the air-bone gap and bone conduction hyperacusis, with no sensorineural hearing loss [
METHODS
Preoperative evaluation included high resolution CT imaging, ocular VEMP responses to 500 Hz tone burst stimuli and cervical VEMP in response to clicks.[
Intraoperative image guidance was used to localize the dehiscent canal. To obliterate the lumen of the canal, the lumen of the dehiscent canal was plugged for 2–3 mm beyond either end of the dehiscent opening using fascia strips, bone dust, and bone chips. Care was taken not to induce unnecessary force or suction near the membranous labyrinth. The area was covered with hydroxyapatite cement, followed by a layer of fascia and fibrin glue.
This study was a review of existing clinical data with patient identifiers removed. It qualified for exemption from an institutional review board protocol on the basis of the United States Department of Health and Human Services criteria 45 CFR 46.101(b4). This exemption was approved by the Johns Hopkins Institutional Review Board.
DISCUSSION
ECochG as a diagnostic and intraoperative adjunct has been reported in cases of SCDS.[
ECochG has been historically a test used for the assessment of endolymphatic hydrops, associated with Ménière's disease.[
CONCLUSION
We present a case of superior semicircular canal dehiscence syndrome during which intraoperative ECochG recordings showed an SP/AP ratio >1, and a summating potential large enough to be identified as a new wave on the ABR. While the clinical consequences of a large spike in the summating potential is unknown, this finding likely reflects a change in inner ear physiology and may lead to new understandings of origin of the ECochG summating potential.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Acknowledgements
The authors would like to thank “Chely” Nirma Carballido Martinez for her assistance with acquiring intraoperative electrocochleography and auditory brainstem response data and images for review.
References
1. Adams ME, Kileny PR, Telian SA, El-Kashlan HK, Heidenreich KD, Mannarelli GR. Electrocochleography as a diagnostic and intraoperative adjunct in superior semicircular canal dehiscence syndrome. Otol Neurotol. 2011. 32: 1506-12
2. Arts HA, Adams ME, Telian SA, El-Kashlan H, Kileny PR. Reversible electrocochleographic abnormalities in superior canal dehiscence. Otol Neurotol. 2009. 30: 79-86
3. Büki B, de Kleine E, Wit HP, Avan P. Detection of intracochlear and intracranial pressure changes with otoacoustic emissions: A gerbil model. Hear Res. 2002. 167: 180-91
4. Büki B, Giraudet F, Avan P. Non-invasive measurements of intralabyrinthine pressure changes by electrocochleography and otoacoustic emissions. Hear Res. 2009. 251: 51-9
5. Carey JP, Migliaccio AA, Minor LB. Semicircular canal function before and after surgery for superior canal dehiscence. Otol Neurotol. 2007. 28: 356-64
6. Crane BT, Lin FR, Minor LB, Carey JP. Improvement in autophony symptoms after superior canal dehiscence repair. Otol Neurotol. 2010. 31: 140-46
7. Crane BT, Minor LB, Carey JP. Superior canal dehiscence plugging reduces dizziness handicap. Laryngoscope. 2008. 118: 1809-13
8. Ferraro J, Best LG, Arenberg IK. The use of electrocochleography in the diagnosis, assessment, and monitoring of endolymphatic hydrops. Otolaryngol Clin N Am. 1983. 16: 69-82
9. Ikino CM, de Almeida ER. Summating potential-action potential waveform amplitude and width in the diagnosis of Meniere's disease. Laryngoscope. 2006. 116: 1766-9
10. Margolis RH, Rieks D, Fournier EM, Levine SE. Tympanic electrocochleography for diagnosis of Meniere's disease. Arch Otolaryngol Head Neck Surg. 1995. 121: 44-55
11. Minor LB, Cremer PD, Carey JP, Della Santina CC, Streubel SO, Weg N. Symptoms and signs in superior canal dehiscence syndrome. Ann N Y Acad Sci. 2001. 942: 259-73
12. Minor LB, Solomon D, Zinreich JS, Zee DS. Sound- and/or pressure-induced vertigo due to bone dehiscence of the superior semicircular canal. Arch Otolaryngol Head Neck Surg. 1998. 124: 249-58
13. Nguyen KD, Welgampola MS, Carey JP. Test-retest reliability and age-related characteristics of the ocular and cervical vestibular evoked myogenic potential tests. Otol Neurotol. 2010. 31: 793-802
14. Park JH, Lee SY, Song J-J, Choi BY, Koo J-W. Electrocochleographic findings in superior canal dehiscence syndrome. Hear Res. 2015. 323: 61-7
15. Pou AM, Hirsch BE, Durrant JD, Gold SR, Kamerer DB. The efficacy of tympanic electrocochleography in the diagnosis of endolymphatic hydrops. Am J Otol. 1996. 17: 607-11
16. Rosowski JJ, Songer JE, Nakajima HH, Brinsko KM, Merchant SN. Clinical, experimental, and theoretical investigations of the effect of superior semicircular canal dehiscence on hearing mechanisms. Otol Neurotol. 2004. 25: 323-32
17. Sass K. Sensitivity and specificity of transtympanic electrocochleography in Meniere's disease. Acta Otolaryngol. 1998. 118: 150-6
18. Sone M, Yoshida T, Morimoto K, Teranishi M, Nakashima T, Naganawa S. Endolymphatic hydrops in superior canal dehiscence and large vestibular aqueduct syndromes. Laryngoscope. 2016. 126: 1446-50
19. Ward BK, Agrawal Y, Nguyen E, Della Santina CC, Limb CJ, Francis HW. Hearing Outcomes After Surgical Plugging of the Superior Semicircular Canal by a Middle Cranial Fossa Approach. Otol Neurotol. 2012. 33: 1386-91
20. Ward BK, Wenzel A, Ritzl EK, Gutierrez-Hernandez S, Della Santina CC, Minor LB. Near-dehiscence: Clinical findings in patients with thin bone over the superior semicircular canal. Otol Neurotol. 2013. 34: 1421-8
21. Welgampola MS, Myrie OA, Minor LB, Carey JP. Vestibular-evoked myogenic potential thresholds normalize on plugging superior canal dehiscence. Neurology. 2008. 70: 464-72
22. Wenzel A, Ward BK, Ritzl EK, Gutierrez-Hernandez S, Della Santina CC, Minor LB. Intraoperative neuromonitoring for superior semicircular canal dehiscence and hearing outcomes. Otol Neurotol. 2015. 36: 139-45
23. Zuniga MG, Janky KL, Nguyen KD, Welgampola MS, Carey JP. Ocular versus cervical VEMPs in the diagnosis of superior semicircular canal dehiscence syndrome. Otol Neurotol. 2013. 34: 121-6