- Department of Neurological Surgery, Instituto Nacional de Neurología y Neurocirugía “Manuel Velasco Suárez”, City, Mexico
- Neurological Surgery, Hospital Angeles Pedregal, Mexico City, Mexico
- Faculty of Health Sciences, Universidad Anáhuac, Mexico City, Mexico
- Department of Neurosurgery, Hospital Universitario Dr. José Eleuterio González, Monterrey, Mexico
- Department of Neurosurgery, The Johns Hopkins University School of Medicine, Baltimore, USA
Correspondence Address:
Juan Luis Gómez-Amador
Department of Neurological Surgery, Instituto Nacional de Neurología y Neurocirugía “Manuel Velasco Suárez”, City, Mexico
DOI:10.4103/sni.sni_165_17
Copyright: © 2017 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Luis Alberto Ortega-Porcayo, Alexander Perdomo-Pantoja, Isaac Jair Palacios-Ortíz, Salomon Cohen Cohen, Juan Pablo González-Mosqueda, Juan Luis Gómez-Amador. Endoscopic management of a cavernous malformation on the floor of third ventricle and aqueduct of Sylvius: Technical case report and review of the literature. 26-Sep-2017;8:237
How to cite this URL: Luis Alberto Ortega-Porcayo, Alexander Perdomo-Pantoja, Isaac Jair Palacios-Ortíz, Salomon Cohen Cohen, Juan Pablo González-Mosqueda, Juan Luis Gómez-Amador. Endoscopic management of a cavernous malformation on the floor of third ventricle and aqueduct of Sylvius: Technical case report and review of the literature. 26-Sep-2017;8:237. Available from: http://surgicalneurologyint.com/?post_type=surgicalint_articles&p=8623
Abstract
Background:Intraventricular cavernous malformations are unusual intracranial vascular malformations; their deep anatomical location complicates their surgical management. Microsurgical approaches are the gold standard approaches for the resection of ventricular lesions, however, they imply considerable neurovascular risks.
Case Description:A 51-year-old patient presented with acute headache, diplopia, vertigo, blurred vision, and a depressed level of consciousness. A ventricular hemorrhage was treated with a ventriculostomy and the patient was discharged without hydrocephalus. After 11 days, he developed ataxia, diplopia, and a depressed level of consciousness. The patient was diagnosed with hydrocephalus secondary to the previous third ventricle hemorrhage. An endoscopic exploration using a 30° rigid ventricular endoscope was performed; after the third ventriculostomy, an intraventricular cavernous malformation located on the floor of the third ventricle and the aqueduct of Sylvius was resected.
Conclusions:Three days after the surgery, magnetic resonance imaging demonstrated a gross total resection and adequate third ventriculostomy flow. One year after the surgery, the patient was asymptomatic. Neuroendoscopy has evolved towards minimally invasiveness, and in selected cases is an equally effective surgical approach to ventricular lesions. It provides minimal cerebral cortex disruption and vascular manipulation.
Keywords: Aqueduct of Sylvius, cavernous malformation, endoscopy, intraventricular, minimally invasive, third ventricle
INTRODUCTION
A cavernous malformation (CM) is an unusual vascular lesion characterized by its bleeding predisposition and composed of sinusoidal vascular spaces lined by a single layer of endothelium not separated by parenchyma.[
CASE DESCRIPTION
Clinical findings
A 51-year-old patient presented with acute headache, diplopia, vertigo, and blurred vision. During the following hours, the level of consciousness was acutely depressed. He required ventilatory support. Computed tomography (CT) demonstrated an intraventricular hemorrhage Graeb[
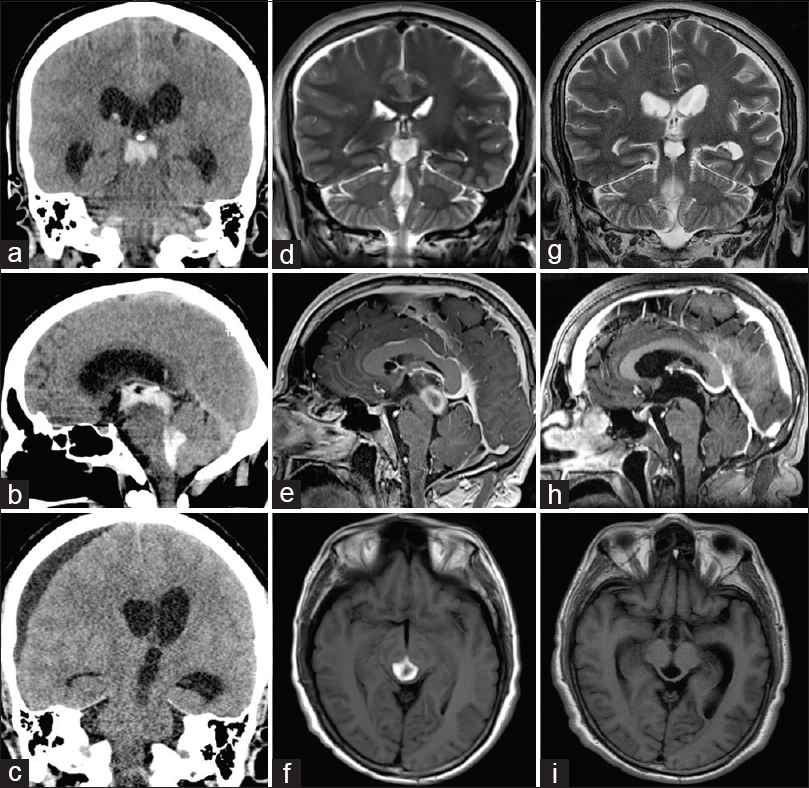
Figure 1
(a and b) Initial coronal and sagittal CT study that demonstrated an intraventricular hemorrhage modified Graeb of 11. (c) Notice in the second preoperative CT a right subdural hygroma and obstructive hydrocephalus. (d-f) Magnetic resonance imaging (MRI) showed a late subacute hemorrhage at the posterior TV and the aqueduct of Sylvius. T2-weighted, T1-weighted simple and contrasted T1 were all hyperintense. (g-i) MRI three days after the surgery demonstrated a gross total resection
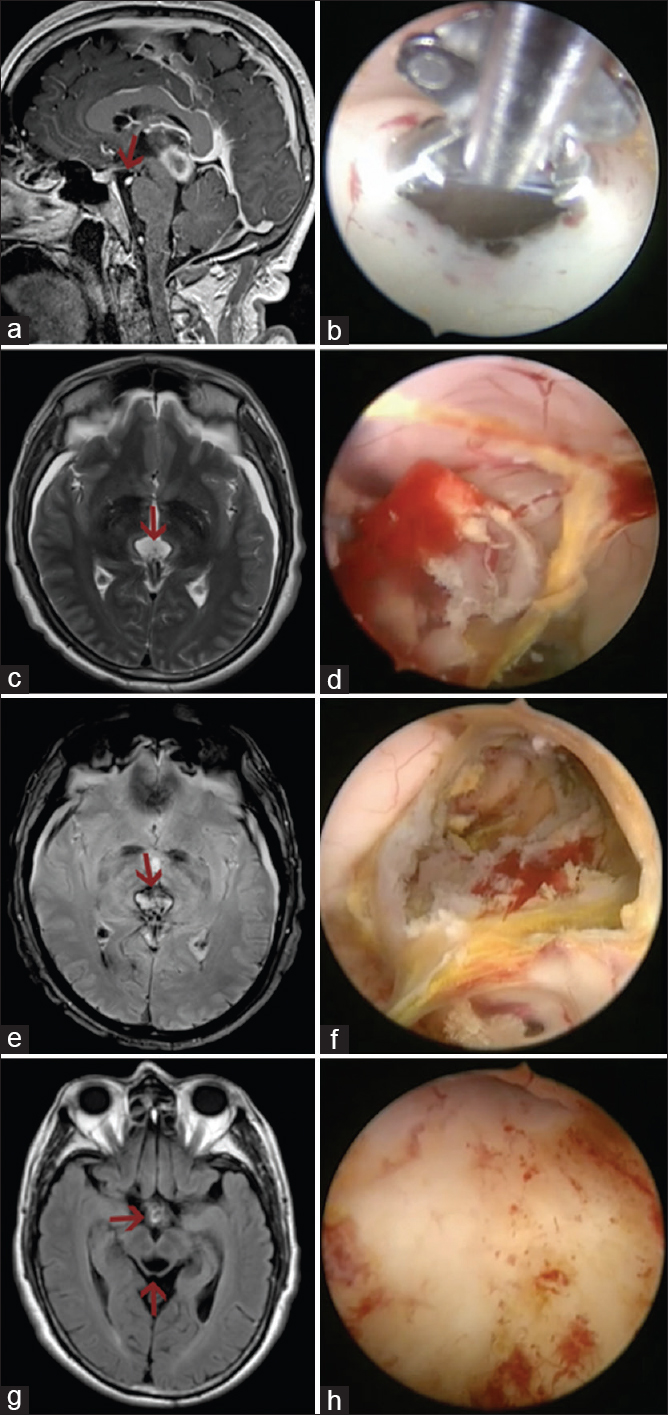
Figure 2
(a and b) Endoscopic third ventriculostomy was performed using blunt forceps. (c-f) A subacute hemorrhagic lesion was observed. Grasping forceps and bipolar coagulator were used to dissect away the CM and clots from the borders of the aqueduct and TV. Notice that T2 and gradient-echo images showed a hypointense rim due to hemosiderin. (g and h) Endoscopic inspection revealed no residual lesion. Postoperative MRI demonstrated a gross total resection (caudal arrow) and adequate third ventriculostomy flow (rostral arrow)
Surgical technique
Neuroanesthesiology used an armored oral endotracheal tube and total intravenous anesthesia. The patient was placed supine with a 30° head elevation so that the burr hole site was at the highest point. The site of the burr hole and the trajectory was planned using preoperative MRI measurements; the trajectory was planned in between the aqueduct and tuber cinereum through the foramen of Monro. A 30° 6 mm ventricular endoscope (DECQ neuroendoscope, Karl Storz, Tuttlingen, Deutschland) was used for the procedure. An endoscopic third ventriculostomy (ETV) was performed using blunt forceps [
Postoperative course
Immediately after the surgery, the patient was awake and his ocular movements improved. The patient was discharged on the fifth postoperative day. MRI obtained 3 days after the surgery demonstrated a gross total resection and adequate third ventriculostomy flow [
DISCUSSION
Surgical approach
Microsurgical approaches have been considered the gold standard for managing intraventricular lesions.[
The TV can be accessed from above using either an interhemispheric anterior transcallosal approach (IATcA) or a frontal transcortical approach (FTA). Once within lateral ventricle, it is possible to reach the TV through several pathways – transforaminal, transchoroidal, subchoroidal, or interforniceal.[
Three main approaches have been described to reach the posterior TV through the velum interpositum of the suprapineal recess: the supracerebellar infratentorial approach (ScItA), the interhemispheric posterior transcallosal approach (IPTcA), and the occipital transtentorial approach (OTtA).[
Although microsurgical techniques to reach intraventricular lesions with less morbidity have improved, the endoscopic transfrontal approach offers interesting benefits.[
Intraventricular cavernous malformations
Faropoulos et al.[
CONCLUSIONS
Ventricular endoscopy should be considered as a useful minimally invasive technique for some well-selected patients harboring intraventricular lesions.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Video Available on: www.surgicalneurologyint.com
References
1. Abdulrauf SI, Kaynar MY, Awad IA. A comparison of the clinical profile of cavernous malformations with and without associated venous malformations. Neurosurgery. 1999. 44: 41-
2. Abe M, Hagihara N, Tabuchi K, Uchino A, Miyasaka Y. Histologically classified venous angiomas of the brain: A controversy. Neurol Med Chir (Tokyo). 2003. 43: 1-10
3. Awad IA, Robinson JR, Mohanty S, Estes ML. Mixed vascular malformations of the brain: Clinical and pathogenetic considerations. Neurosurgery. 1993. 33: 179-88
4. Barrow DL, Schuette AJ. Cavernous malformations: A paradigm for progress. Clin Neurosurg. 2011. 58: 27-41
5. Behari S, Garg P, Jaiswal S, Nair A, Naval R, Jaiswal AK. Major surgical approaches to the posterior third ventricular region: A pictorial review. J Pediatr Neurosci. 2010. 5: 97-101
6. Biller J, Toffol GJ, Shea JF, Fine M, Azar-Kia B. Cerebellar venous angiomas. A continuing controversy. Arch Neurol. 1985. 42: 367-70
7. Brown A, Thompson B, Spetzler R. The two-point method: Evaluating brainstem lesions. BNI Q. 1996. 12: 20-4
8. Chourmouzi D, Kaltsikis I, Zountsas B, Drevelegas A. Third ventricle cavernoma associated with multiple intracerebral cavernomas. Acta Neurol Belg. 2013. 113: 99-101
9. Chowdhry SA, Cohen AR. Intraventricular neuroendoscopy: Complication avoidance and management. World Neurosurg. 2013. 79: S15 e11-10
10. Cikla U, Swanson KI, Tumturk A, Keser N, Uluc K, Cohen-Gadol A. Microsurgical resection of tumors of the lateral and third ventricles: Operative corridors for difficult-to-reach lesions. J Neurooncol. 2016. 130: 331-40
11. Collmann H, Kazner E, Sprung C. Supratentorial intraventricular tumors in childhood. Acta Neurochir Suppl (Wien). 1985. 35: 75-9
12. Del Curling O, Kelly DL, Elster AD, Craven TE. An analysis of the natural history of cavernous angiomas. J Neurosurg. 1991. 75: 702-8
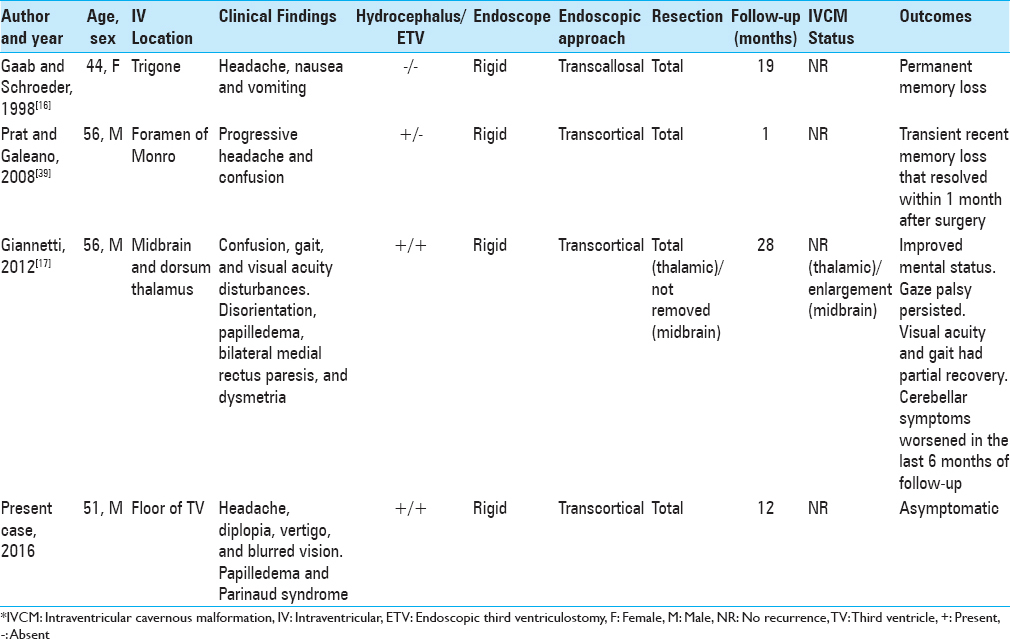
13. Faropoulos K, Panagiotopoulos V, Partheni M, Tzortzidis F, Konstantinou D. Therapeutic management of intraventricular cavernoma: Case series and review of the literature. J Neurol Surg A Cent Eur Neurosurg. 2015. 76: 233-9
14. Feletti A, Dimitriadis S, Pavesi G. Cavernous angioma of the cerebral aqueduct. World Neurosurg. 2017. 98: 876-
15. Gaab MR, Schroeder HW. Neuroendoscopic approach to intraventricular lesions. Neurosurg Focus. 1999. 6: e5-
16. Gaab MR, Schroeder HW. Neuroendoscopic approach to intraventricular lesions. J Neurosurg. 1998. 88: 496-505
17. Giannetti AV. Purely neuroendoscopic resection of an intraventricular cavernous angioma: Case report. J Neurol Surg A Cent Eur Neurosurg. 2013. 74: 47-50
18. Graeb DA, Robertson WD, Lapointe JS, Nugent RA, Harrison PB. Computed tomographic diagnosis of intraventricular hemorrhage. Etiology and prognosis. Radiology. 1982. 143: 91-6
19. Gross BA, Lin N, Du R, Day AL. The natural history of intracranial cavernous malformations. Neurosurg Focus. 2011. 30: E24-
20. Hassani FD, Gana R, El Abbadi N, El Fatemi N, Maaqili MR. [Cavernoma of the fourth ventricle: Report of a case and review of the literature]. Pan Afr Med J. 2014. 18: 331-
21. Kim DS, Park YG, Choi JU, Chung SS, Lee KC. An analysis of the natural history of cavernous malformations. Surg Neurol. 1997. 48: 9-
22. Kivelev J, Niemela M, Hernesniemi J. Characteristics of cavernomas of the brain and spine. J Clin Neurosci. 2012. 19: 643-8
23. Kivelev J, Niemela M, Hernesniemi J. Treatment strategies in cavernomas of the brain and spine. J Clin Neurosci. 2012. 19: 491-7
24. Kivelev J, Niemela M, Kivisaari R, Hernesniemi J. Intraventricular cerebral cavernomas: A series of 12 patients and review of the literature. J Neurosurg. 2010. 112: 140-9
25. Konovalov AN, Gorelyshev SK. Surgical treatment of anterior third ventricle tumours. Acta Neurochir (Wien). 1992. 118: 33-9
26. Lan Q, Dong J, Huang Q. Minimally invasive keyhole approaches for removal of tumors of the third ventricle. Chin Med J (Engl). 2006. 119: 1444-50
27. Li KW, Nelson C, Suk I, Jallo GI. Neuroendoscopy: Past, present, and future. Neurosurg Focus. 2005. 19: E1-
28. Liebelt BD, Hooten KG, Britz GW. The Anterior Subcallosal Approach to Third Ventricular and Suprasellar Lesions: Anatomical Description and Technical Note. World Neurosurg. 2016. 87: 187-94
29. Lobato RD, Perez C, Rivas JJ, Cordobes F. Clinical, radiological, and pathological spectrum of angiographically occult intracranial vascular malformations. Analysis of 21 cases and review of the literature. J Neurosurg. 1988. 68: 518-31
30. Louis N, Marsh R. Simultaneous and sequential hemorrhage of multiple cerebral cavernous malformations: A case report. J Med Case Rep. 2016. 10: 36-
31. Lozier AP, Bruce JN. Surgical approaches to posterior third ventricular tumors. Neurosurg Clin N Am. 2003. 14: 527-45
32. Martirosyan NL, Kalani MY, Nakaji P, Spetzler RF. The anterior interhemispheric approach to a third ventricular cavernous malformation. Neurosurg Focus. 2016. 40:
33. Mazher S, Imran M, Ashraf J, Ahmed A, Shah IU, Zulfiqar F. Outcome of open transcortical approach in the management of intraventricular lesions. J Coll Physicians Surg Pak. 2013. 23: 857-61
34. McCormick WF. The pathology of vascular (“arteriovenous”) malformations. J Neurosurg. 1966. 24: 807-16
35. Morgan TC, Dawson J, Spengler D, Lees KR, Aldrich C, Mishra NK. The Modified Graeb Score: An enhanced tool for intraventricular hemorrhage measurement and prediction of functional outcome. Stroke. 2013. 44: 635-41
36. Nagashima H, Tanaka K, Sasayama T, Okamura Y, Taniguchi M, Otani K. A large cavernous malformation of the third ventricle floor: A case report. Neurol Neurochir Pol. 2015. 49: 446-50
37. Ohbuchi H, Osaka Y, Ogawa T, Nanto M, Nakahara Y, Katsura K. Trigonal cavernous malformation with intraventricular hemorrhage: A case report and literature review. J Med Invest. 2012. 59: 275-9
38. Patibandla MR, Thotakura AK, Panigrahi MK. Third ventricular cavernous malformation: An unusual lesion. Br J Neurosurg. 2014. 28: 110-2
39. Prat R, Galeano I. Endoscopic resection of cavernoma of foramen of Monro in a patient with familial multiple cavernomatosis. Clin Neurol Neurosurg. 2008. 110: 834-7
40. Rigamonti D, Spetzler RF. The association of venous and cavernous malformations. Report of four cases and discussion of the pathophysiological, diagnostic, and therapeutic implications. Acta Neurochir (Wien). 1988. 92: 100-5
41. Robinson JR, Awad IA, Little JR. Natural history of the cavernous angioma. J Neurosurg. 1991. 75: 709-14
42. Santos MM, Souweidane MM. Purely endoscopic resection of a choroid plexus papilloma of the third ventricle: Case report. J Neurosurg Pediatr. 2015. 16: 54-7
43. Sarwar M, McCormick WF. Intracerebral venous angioma. Case report and review. Arch Neurol. 1978. 35: 323-5
44. Simard JM, Garcia-Bengochea F, Ballinger WE, Mickle JP, Quisling RG. Cavernous angioma: A review of 126 collected and 12 new clinical cases. Neurosurgery. 1986. 18: 162-72
45. Souweidane MM, Hoffman CE, Schwartz TH. Transcavum interforniceal endoscopic surgery of the third ventricle. J Neurosurg Pediatr. 2008. 2: 231-6
46. Souweidane MM, Luther N. Endoscopic resection of solid intraventricular brain tumors. J Neurosurg. 2006. 105: 271-8
47. Spina A, Gagliardi F, Bailo M, Boari N, Caputy AJ, Mortini P. Comparative Anatomical Study on Operability in Surgical Approaches to the Anterior Part of the Third Ventricle. World Neurosurg. 2016. 95: 457-63
48. Vitorino Araujo JL, Veiga JC, Wen HT, de Andrade AF, Teixeira MJ, Otoch JP. Comparative anatomical analysis of the transcallosal-transchoroidal and transcallosal-transforniceal-transchoroidal approaches to the third ventricle. J Neurosurg. 2017. 127: 209-18
49. Voigt K, Yasargil MG. Cerebral cavernous haemangiomas or cavernomas. Incidence, pathology, localization, diagnosis, clinical features and treatment. Review of the literature and report of an unusual case. Neurochirurgia (Stuttg). 1976. 19: 59-68
50. Winkler PA, Ilmberger J, Krishnan KG, Reulen HJ. Transcallosal interforniceal-transforaminal approach for removing lesions occupying the third ventricular space: Clinical and neuropsychological results. Neurosurgery. 2000. 46: 879-
51. Winslow N, Abode-Iyamah K, Flouty O, Park B, Kirby P, Howard M. Intraventricular foramen of Monro cavernous malformation. J Clin Neurosci. 2015. 22: 1690-3
52. Wurm G, Schnizer M, Fellner FA. Cerebral cavernous malformations associated with venous anomalies: Surgical considerations. Neurosurgery. 2005. 57: 42-58