- Geisel School of Medicine, New Hampshire, Lebanon, New Hampshire, United States
- Section of Neurological Surgery, Department of Surgery, Dartmouth-Hitchcock Medical Center, Lebanon, New Hampshire, United States
Correspondence Address:
Vyacheslav Makler
Section of Neurological Surgery, Department of Surgery, Dartmouth-Hitchcock Medical Center, Lebanon, New Hampshire, United States
DOI:10.4103/sni.sni_52_18
Copyright: © 2018 Surgical Neurology International This is an open access journal, and articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as appropriate credit is given and the new creations are licensed under the identical terms.How to cite this article: Erin D'Agostino, Vyacheslav Makler, David F. Bauer. Epidural abscess presenting as severe depression with suicidal ideations: Case report. 16-Apr-2018;9:83
How to cite this URL: Erin D'Agostino, Vyacheslav Makler, David F. Bauer. Epidural abscess presenting as severe depression with suicidal ideations: Case report. 16-Apr-2018;9:83. Available from: http://surgicalneurologyint.com/surgicalint-articles/epidural-abscess-presenting-as-severe-depression-with-suicidal-ideations-case-report/
Abstract
Background:Epidural abscess (EDA) is an uncommon form of intracranial infection that generally presents with fever, headache, and focal neurologic deficit. Imaging generally reveals a lentiform collection with diffusion restriction on diffusion weighted image. We present an interesting case in which a patient with EDA presented with three weeks of depression with suicidal ideations. The patient displayed no notable infectious signs and the imaging was suggestive of chronic subdural hematoma (SDH) rather than EDA.
Case Description:The patient is a 57-year-old man with past medical history significant for epilepsy and left hemiplegia secondary to remote traumatic brain injury who presented with a three-week history of depression, anxiety, and active suicidal ideation, resulting in psychiatric admission to an outside hospital. He had undergone three previous craniotomies for SDH many years ago and had no significant psychiatric history. Magnetic resonance imaging was consistent with subacute right SDH. On presentation, patient was at neurologic baseline and was afebrile with unremarkable labs. Operative findings demonstrated frank purulence in the epidural space. The patient was treated with antibiotics and both depression and suicidal ideations resolved postoperative day 5.
Conclusions:EDA can present in atypical ways, especially in patients who have undergone previous cranial procedures. Depression is one possible atypical presentation.
Keywords: Depression, epidural abscess, subdural empyema, suicidal ideations
INTRODUCTION
Subdural empyema (SDE) and epidural abscess (EDA) comprise between 6 and 20% of intracranial infections.[
CASE REPORT
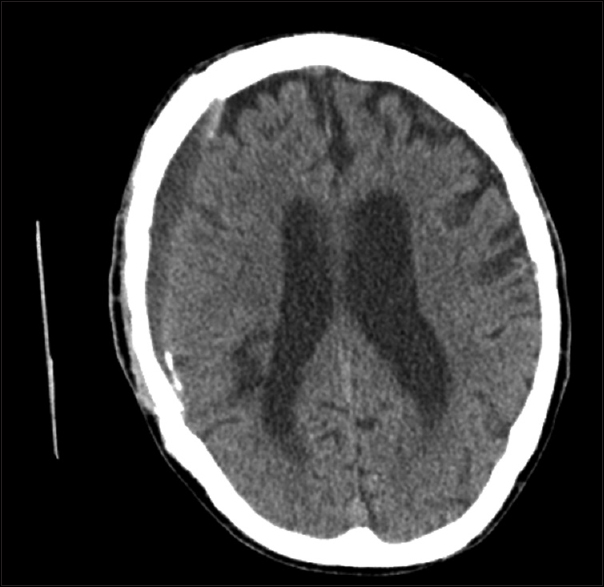
The patient is a 57-year-old man with past medical history significant for epilepsy and left hemiplegia secondary to right-sided traumatic brain injury (TBI) 28 years prior to presentation. He presented with a 3-week history of depression, anxiety, and active suicidal ideation resulting in psychiatric admission to an outside hospital. He had three prior craniotomies for right subdural hematoma (SDH), one at the time of his TBI, one 8 years prior to presentation, and one 5 years prior to presentation. On follow-up imaging 3 years after his last craniotomy, he underwent head CT showing a small subacute SDH, which was managed nonoperatively [
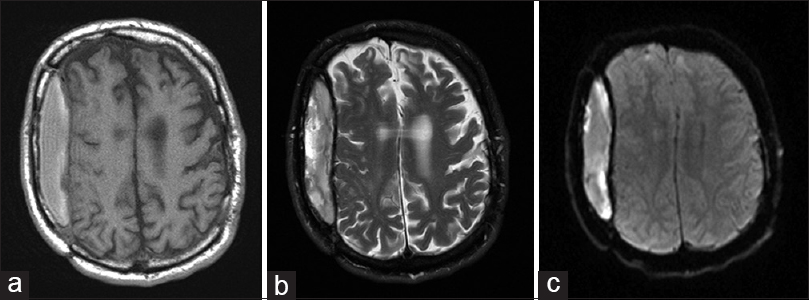
Figure 2
MRI of the brain without contrast on the day of admission. (a) T1WI showing hyperintense extraaxial fluid collection within the bounds of previous craniotomy on the right side. (b) T2WI showing hyperintense extraaxial fluid collection within the bounds of previous craniotomy on the right side. Both of these findings were consistent with late subacute SDH. (c) DWI shows some diffusion restriction within the extraaxial fluid collection on the right side. This was felt to be nonspecific secondary to history of previous extraaxial blood
DISCUSSION
EDA and SDE are typically the result of paranasal sinusitis, otomastoiditis, postoperative infection, trauma, or meningitis.[
The pathogenesis of SDE involves bacterial seeding followed by a robust inflammatory response and rapid progression due to little resistance to expansion offered by the leptomeninges. Local meningeal irritation can become diffuse meningitis if the arachnoid layer is breached. Septic thrombosis of bridging veins can result in cortical inflammation and vascular congestion, causing ischemia and edema within neighboring brain parenchyma. These complications can result in a fulminant clinical deterioration secondary to inflammation and mass effect.[
SDE and EDA can typically be differentiated by imaging, with SDE having a crescent shape that does not cross the midline and EDA having a lentiform shape that can cross the midline. However, following surgery adherence to suture lines may be interrupted, causing deviation from typical shape of collection. On MRI, SDE demonstrates isointense signal on T1WI and high signal on T2WI, and is bright on DWI with corresponding low apparent coefficient diffusion values.[
Rarely, patients present without any of the typical symptoms of infection. There are four cases in the literature of depression as the presenting feature of SDE or hematoma. Three patients with chronic SDH[
CONCLUSION
EDA typically presents with subacute onset of fever, headache, and focal neurologic deficits. We describe a patient who presented with several weeks history of psychiatric symptoms and absence of infectious signs. Index of suspicion for EDA should be higher in patients who have had cranial procedures. EDA may not demonstrate typical imaging findings due to altered anatomy and chronicity of the collection.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Agrawal A, Timothy J, Pandit L, Shetty L, Shetty J. A review of subdural empyema and its management. Infect Dis Clin Pract. 2007. 15: 149-53
2. Bartt RE. Cranial epidural abscess and subdural empyema. Handb Clin Neurol. 2010. 96: 75-89
3. Farrell CJ, Hoh BL, Pisculli ML, Henson JW, Barker FG, Curry Jr WT. Limitations of diffusion-weighted imaging in the diagnosis of postoperative infections. Neurosurgery. 2008. 62: 577-83
4. Foerster BR, Thurnher MM, Malani PN, Petrou M, Carets-Zumelzu F, Sundgren PC. Intracranial infections: Clinical and imaging characteristics. Acta Radiol. 2007. 48: 875-93
5. French H, Schaefer N, Keijzers G, Barison D, Olson S. Intracranial subdural empyema: A 10-year case series. Ochsner J. 2014. 14: 188-94
6. Fukui T, Ueda A, Murate KI, Hikichi C, Ito S, Asakura K. Depressive state as an initial symptom for subdural abscess. Neurol Clin Neurosci. 2016. 4: 31-3
7. Harris LF, Haws FP, Triplett JJ, Maccubbin DA. Subdural empyema and epidural abscess: Recent experience in a community hospital. South Med J. 1987. 80: 1254-8
8. Hlavin ML, Kaminski HJ, Fenstermaker RA, White RJ. Intracranial suppuration: A modern decade of postoperative subdural empyema and epidural abscess. Neurosurgery. 1994. 34: 974-81
9. Nagatomo I, Ueyama K, Fukuzako H, Matsumoto K. Three cases of chronic subdural hematoma with depressive state. Psychiatry Clin Neurosci. 1990. 44: 703-7
10. Nathoo N, Nadvi SS, van Dellen JR. Cranial extradural empyema in the era of computed tomography: A review of 82 cases. Neurosurgery. 1999. 44: 748-53
11. Rich PM, Deasy NP, Jarosz JM. Intracranial dural empyema. Br J Radiol. 2000. 73: 1329-36
12. Singh B, Dellen JV, Ramjettan S, Maharaj TJ. Sinogenic intracranial complications. J Laryngol Otol. 2007. 109: 945-50
13. Tsai YD, Chang WN, Shen CC, Lin YC, Lu CH, Liliang PC. Intracranial suppuration: A clinical comparison of subdural empyemas and epidural abscesses. Surg Neurol. 2003. 59: 191-6
14. Tsuchiya K, Makita K, Furui S, Kusano S, Inoue Y. Contrast-enhanced magnetic resonance imaging of sub-and epidural empyemas. Neuroradiology. 1992. 34: 494-6
15. Tsuchiya K, Osawa A, Katase S, Fujikawa A, Hachiya J, Aoki S. Diffusion-weighted MRI of subdural and epidural empyemas. Neuroradiology. 2003. 45: 220-3
16. Wong AM, Zimmerman RA, Simon EM, Pollock AN, Bilaniuk LT. Diffusion-weighted MR imaging of subdural empyemas in children. Am J Neuroradiol. 2004. 25: 1016-21