- Department of Neurosurgery, Shiga University of Medical Science, Seta-Tsukinowa-Cho, Otsu, Shiga, Japan
- National Epilepsy Center, Shizuoka Institute of Epilepsy and Neurological Disorders, Aoi-ku, Shizuoka, Japan
- Department of Clinical Laboratory Medicine and Division of Diagnostic Pathology, Shiga University of Medical Science, Seta-Tsukinowa-Cho, Otsu, Shiga, Japan
Correspondence Address:
Naoki Nitta
Department of Neurosurgery, Shiga University of Medical Science, Seta-Tsukinowa-Cho, Otsu, Shiga, Japan
DOI:10.4103/sni.sni_289_17
Copyright: © 2018 Surgical Neurology International This is an open access journal, and articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as appropriate credit is given and the new creations are licensed under the identical terms.How to cite this article: Hitoshi Kayatani, Naoki Nitta, Suzuko Moritani, Kazuhiko Nozaki. Primary diffuse large B-cell lymphoma of the choroid plexus: A case report and review of the literature. 25-May-2018;9:110
How to cite this URL: Hitoshi Kayatani, Naoki Nitta, Suzuko Moritani, Kazuhiko Nozaki. Primary diffuse large B-cell lymphoma of the choroid plexus: A case report and review of the literature. 25-May-2018;9:110. Available from: http://surgicalneurologyint.com/surgicalint-articles/primary-diffuse-large-b%e2%80%91cell-lymphoma-of-the-choroid-plexus-a-case-report-and-review-of-the-literature/
Abstract
Background:Primary lymphomas in the choroid plexus are much less frequent than primary lymphomas in the brain parenchyma.
Case Description:A 66-year-old male patient was referred to our department with a right intraventricular mass that had been diagnosed by biopsy at another hospital as anaplastic ependymoma. The patient underwent subtotal removal of the tumor via a transcortical inferior temporal gyrus approach. The mass was attached to the choroid plexus in the right atrium. Histopathological examination showed diffuse large B-cell lymphoma. Ophthalmological examination, blood tests, computed tomography of the whole body, and bone marrow biopsy did not show any other lesion, leading to the diagnosis of primary choroid plexus lymphoma. The patient underwent chemotherapy with three courses of high-dose methotrexate and one course of carboplatin and etoposide followed by whole-brain irradiation (1.8 Gy × 22).
Conclusion:We present a rare case of primary choroid plexus lymphoma, which should be considered in the differential diagnosis of choroid plexus tumors.
Keywords: Diffuse large B-cell lymphoma, lateral ventricle, primary choroid plexus lymphoma
INTRODUCTION
Primary central nervous system lymphoma (PCNSL) is typically a diffuse large B-cell lymphoma that accounts for about 2.4–3% of all central nervous system (CNS) tumors with an overall annual incidence rate of 0.47 cases per 100,000 population.[
PCNSL frequently presents as a solitary mass with profound peritumoral edema. The supratentorial compartment is involved in about 60% of PCNSLs, whereas the posterior fossa or spinal cord is less frequently affected.[
CASE DESCRIPTION
A 66-year-old male patient was referred to our department with a right intraventricular mass that had been diagnosed as anaplastic ependymoma on open biopsy at another hospital.
He had been followed because of diabetes mellitus, hyperlipidemia, and carotid artery stenosis but had been otherwise well. Two months before admission to our department, he had undergone magnetic resonance imaging (MRI) of the head, which incidentally revealed a mass lesion in the right lateral ventricle, for which he had been referred to the neurosurgery department at another hospital. He underwent a biopsy and the tumor was diagnosed as an anaplastic ependymoma. Although he had been initially referred to a Radiation Oncology Department for adjuvant radiation therapy, the provider referred him to our Department of Neurosurgery because of concerns that the residual tumor was not suitable for radiotherapy without prior cytoreduction.
Physical examination showed some short-term memory impairment, and ophthalmological examination revealed a visual field compromise (quadrantanopia) likely related to access corridor chosen for the biopsy. There was no focal motor weakness, major sensory deficit, dysmetria, or ataxia.
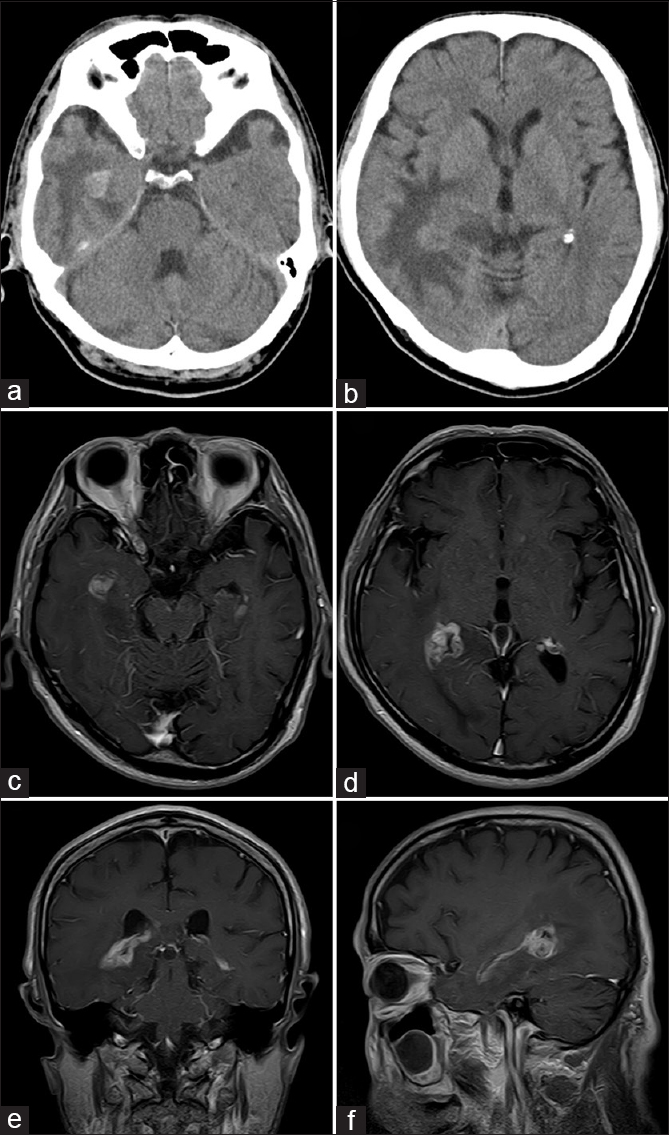
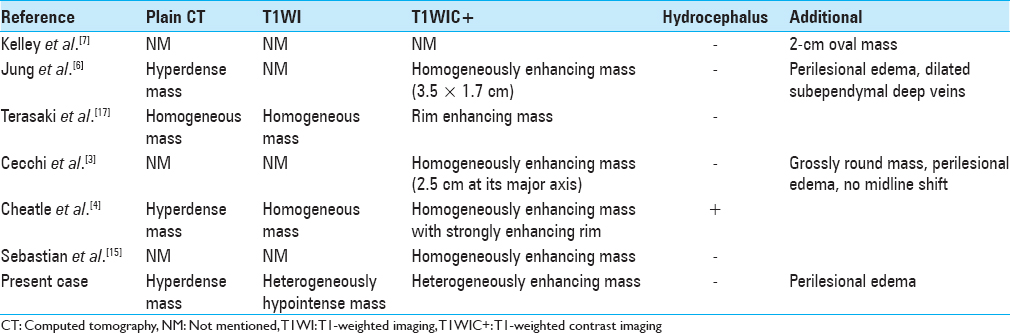
Computed tomography (CT) showed two hyperdense foci in the inferior horn and the atrium of the right lateral ventricle with peri-lesional edema of the ipsilateral temporo-occipital parenchyma [Figure
The patient was taken to surgery and underwent subtotal removal of the tumor in the right lateral ventricle via an inferior temporal gyrus approach [Figure
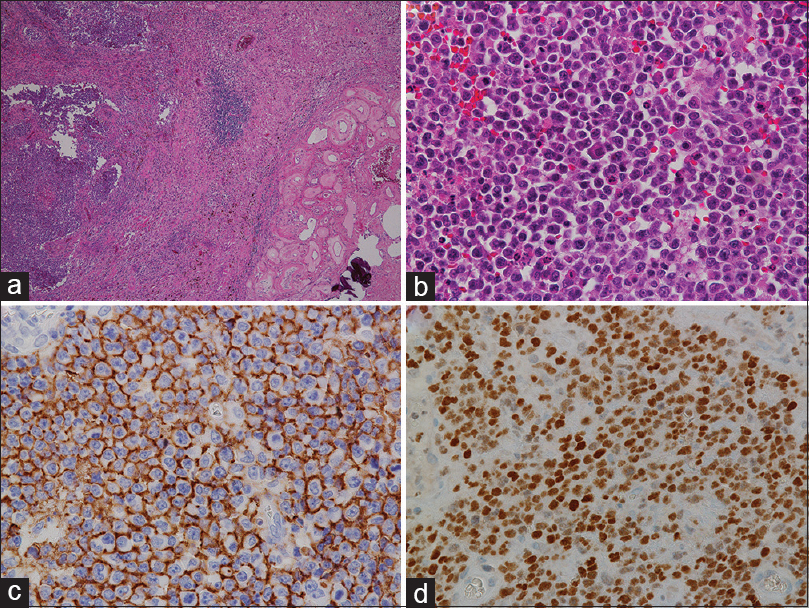
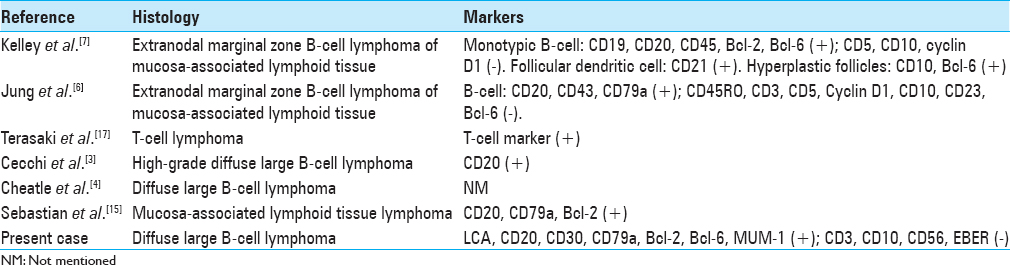
Histopathologically, the tumor showed diffuse, tightly packed monotonous neoplastic cells with large, round, or slightly irregular nuclei and a scant to moderate amount of eosinophilic cytoplasm [
Ophthalmological examination showed no intraocular lesions. CT of the chest, abdomen, and pelvis was interpreted as normal and bone marrow biopsy showed no atypical lymphocytic infiltrate. The patient was thus diagnosed with primary diffuse large B-cell CNS lymphoma likely originating in the choroid plexus.
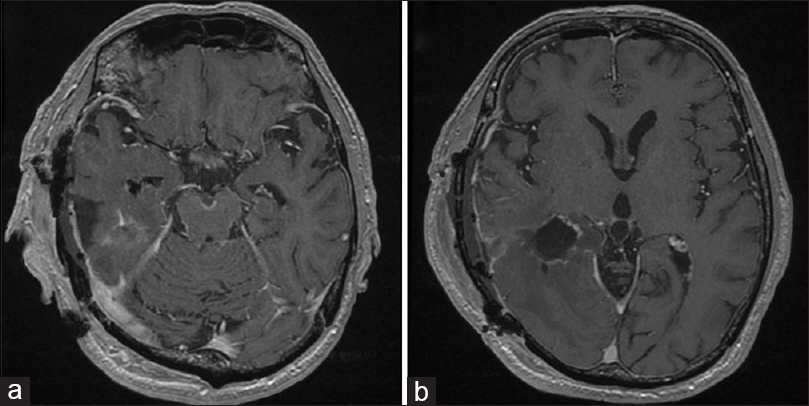
The patient underwent chemotherapy with high-dose methotrexate (3500 mg/m2) every 14 days for three courses, followed by a course of chemotherapy with carboplatin (360 mg calculated by using the Calvert formula) and etoposide (80 mg/m2) 14 days after the third administration of high-dose methotrexate. After chemotherapy, cerebrospinal fluid (CSF) samples were obtained for cytology, which was negative for malignant cells. After the patient had undergone whole-brain irradiation (1.8 Gy × 22), postcontrast T1WI showed complete resolution of all previously visible CNS disease. Physical examination showed persistent quadrantanopia—unchanged from the patients status prior to subtotal removal of tumor—and amelioration of his short-term memory impairment. Because meningeal dissemination recurred during the follow-up period, the patient is currently again receiving chemotherapy with high-dose methotrexate in our department (8 months after surgery).
DISCUSSION
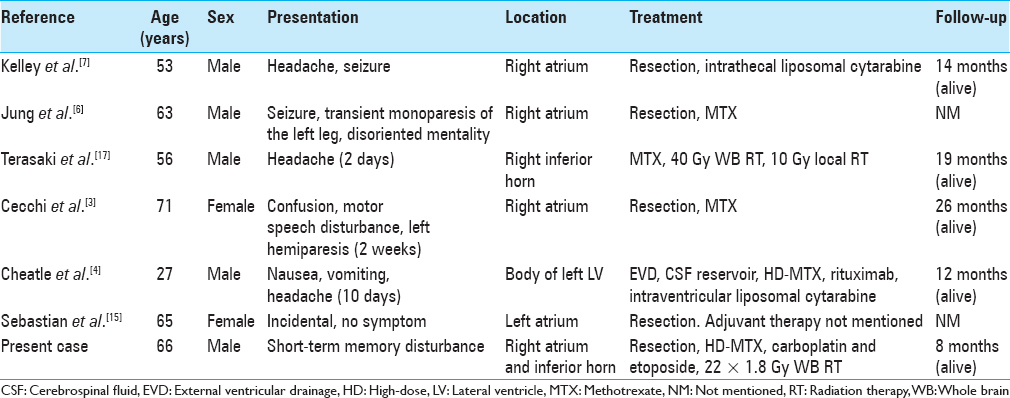
Primary lymphomas in the brain have been reported to occur as single or multiple space occupying lesions that usually occur in the parenchyma, are deep-seated and adjacent to the ventricular system. Lymphomas originating in the choroid plexus itself are rare. To our knowledge, this is only the seventh reported case of such primary choroid plexus lymphoma.[
The choroid plexus is a lobulated structure found in the walls of the ventricles. It has a central highly vascularized stroma surrounded by neuroectodermal cells.[
More typical choroid plexus tumors consist of choroid plexus papilloma, atypical choroid plexus papilloma, and choroid plexus carcinoma, accounting for as little as 0.77% of all brain tumors and 14% of those occurring in the first year of life.[
In all reported cases of primary choroid plexus lymphoma, as well as in our case, the tumors were located in the lateral ventricle [
Our patient was initially diagnosed as suffering from ependymoma according to the biopsy material obtained at another hospital, possibly because of the intraventricular location and the tightly packed proliferation of tumor cells which may mimic the epithelioid arrangement characteristic of ependymoma. Although the tumor was already disseminated in the subarachnoid space, we performed subtotal removal for mass reduction prior to radiation therapy of the lesion. However, because the final diagnosis was re-interpreted as diffuse large B-cell lymphoma, we subsequently performed systemic chemotherapy as well as whole-brain radiation therapy.
Differentiation of choroid plexus tumors by imaging characteristics alone remains difficult. Calcification is frequently observed in meningiomas and choroid plexus papillomas as well as in native choroid plexus, and hydrocephalus can be seen as a frequent presenting feature in choroid plexus papilloma.[
Given the rare scenario of this lymphoma presentation, the best algorithm for the establishment of the correct diagnosis remains a tissue-based diagnosis, until specific biomarkers can be identified.
CONCLUSION
Here, we present a rare case of choroid plexus lymphoma, which should be considered in the differential diagnosis of choroid plexus tumors.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form the patient(s) has/have given his/her/their consent for his/her/their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
The authors declare no conflicts of interest in association with this study.
References
1. Camilleri-Broët S, Crinière E, Broët P, Delwail V, Mokhtari K, Moreau A. A uniform activated B-cell-like immunophenotype might explain the poor prognosis of primary central nervous system lymphomas: Analysis of 83 cases. Blood. 2006. 107: 190-6
2. Cannon DM, Mohindra P, Gondi V, Kruser TJ, Kozak KR. Choroid plexus tumor epidemiology and outcomes: Implications for surgical and radiotherapeutic management. J Neurooncol. 2015. 121: 151-7
3. Cecchi PC, Billio A, Colombetti V, Rizzo P, Ricci UM, Schwarz A. Primary high-grade B-cell lymphoma of the choroid plexus. Clin Neurol Neurosurg. 2008. 110: 75-9
4. Cheatle JT, Aizenberg MR, Weinberg JS, Surdell DL. Atypical presentation of primary central nervous system non-Hodgkin lymphoma in immunocompetent young adults. World Neurosurg. 2013. 79: 593.e9-13
5. Josephson SA, Papanastassiou AM, Berger MS, Barbaro NM, McDermott MW, Hilton JF. The diagnostic utility of brain biopsy procedures in patients with rapidly deteriorating neurological conditions or dementia. J Neurosurg. 2007. 106: 72-5
6. Jung TY, Jung S, Lee MC, Lee KH. Extranodal marginal zone B-cell lymphoma mimicking meningioma in lateral ventricle: A case report and possible pathogenesis. J Neurooncol. 2006. 80: 63-7
7. Kelley TW, Prayson RA, Barnett GH, Stevens GH, Cook JR, Hsi ED. Extranodal marginal zone B-cell lymphoma of mucosa-associated lymphoid tissue arising in the lateral ventricle. Leuk Lymphoma. 2005. 46: 1423-7
8. Kitagawa Y, Higuchi F, Abe Y, Matsuda H, Kim P, Ueki K. Metastasis to the choroid plexus from thyroid cancer: Case report. Neurol Med Chir (Tokyo). 2013. 53: 832-6
9. Louis DN, Ohgaki H, Wiestler OD, Cavenee WK.editors. Choroid plexus tumours. WHO Classification of Tumors of the Central Nervous System. Geneva: WHO Press; 2016. p. 124-6
10. Louis DN, Ohgaki H, Wiestler OD, Cavenee WK.editors. Lymphomas. WHO Classification of Tumors of the Central Nervous System. Geneva: WHO Press; 2016. p. 272-277
11. Nakamura M, Roser F, Bundschuh O, Vorkapic P, Samii M. Intraventricular meningiomas: A review of 16 cases with reference to the literature. Surg Neurol. 2003. 59: 491-503
12. Nieuwenhuys R, Voogd J, van Huijzen C.editors. The Human Central Nervous System. Berlin: Springer; 2008. p.
13. Palmedo H, Urbach H, Bender H, Schlegel U, Schmidt-Wolf IG, Matthies A. FDG-PET in immunocompetent patients with primary central nervous system lymphoma: Correlation with MRI and clinical follow-up. Eur J Nucl Med Mol Imaging. 2006. 33: 164-8
14. Rosenfeld SS, Hoffman JM, Coleman RE, Glantz MJ, Hanson MW, Schold SC. Studies of primary central nervous system lymphoma with fluorine-18-fluorodeoxyglucose positron emission tomography. J Nucl Med. 1992. 33: 532-6
15. Sebastián C, Vela AC, Figueroa R, Marín MÁ, Alfaro J. Primary intracranial mucosa-associated lymphoid tissue lymphoma. A report of two cases and literature review. Neuroradiol J. 2014. 27: 425-30
16. Stadnik TW, Chaskis C, Michotte A, Shabana WM, van Rompaey K, Luypaert R. Diffusion-weighted MR imaging of intracerebral masses: Comparison with conventional MR imaging and histologic findings. AJNR Am J Neuroradiol. 2001. 22: 969-76
17. Terasaki M, Abe T, Tajima Y, Fukushima S, Hirohata M, Shigemori M. Primary choroid plexus T-cell lymphoma and multiple aneurysms in the CNS. Leuk Lymphoma. 2006. 47: 1680-2
18. Weiler-Sagie M, Bushelev O, Epelbaum R, Dann EJ, Haim N, Avivi I. (18) F-FDG avidity in lymphoma readdressed: A study of 766 patients. J Nucl Med. 2010. 51: 25-30
19. Zukotynski K, Fahey F, Kocak M, Kun L, Boyett J, Fouladi M. 18F-FDG PET and MR imaging associations across a spectrum of pediatric brain tumors: A report from the pediatric brain tumor consortium. J Nucl Med. 2014. 55: 1473-80