- Department of Radiation Oncology, Princess Alexandra Hospital, Brisbane, Australia
- School of Medicine, University of Queensland, Brisbane, Australia
- Department of Radiation Oncology, Sunnybrook Health Sciences Centre, Toronto, Canada
- Department of Radiation Oncology, University of Toronto, Toronto, Canada
- Department of Radiation Oncology, Johns Hopkins University, Baltimore, USA
- Department of Radiation Oncology, University of Washington School of Medicine, Seattle, USA
Correspondence Address:
Michael Huo
Department of Radiation Oncology, Princess Alexandra Hospital, Brisbane, Australia
School of Medicine, University of Queensland, Brisbane, Australia
DOI:10.4103/2152-7806.200581
Copyright: © 2017 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Michael Huo, Arjun Sahgal, David Pryor, Kristin Redmond, Simon Lo, Matthew Foote. Stereotactic spine radiosurgery: Review of safety and efficacy with respect to dose and fractionation. 20-Feb-2017;8:30
How to cite this URL: Michael Huo, Arjun Sahgal, David Pryor, Kristin Redmond, Simon Lo, Matthew Foote. Stereotactic spine radiosurgery: Review of safety and efficacy with respect to dose and fractionation. 20-Feb-2017;8:30. Available from: http://surgicalneurologyint.com/surgicalint_articles/stereotactic-spine-radiosurgery-review-of-safety-and-efficacy-with-respect-to-dose-and-fractionation/
Abstract
Background:Stereotactic body radiotherapy (SBRT) is an emerging treatment option for spinal metastases with demonstrated efficacy in the upfront, postoperative, and re-treatment settings, as well as for tumor histologies considered radioresistant. Uncertainty exists regarding the optimal dose and fractionation schedule, with single and multifraction regimens commonly utilized.
Methods:A literature search of the PubMed and Medline databases was conducted to identify papers specific to spine SBRT and the effect of varying dose/fractionation regimens on outcomes. Bibliographies of relevant papers were searched for further references, and international spine SBRT experts were consulted.
Results:Local control rates generally exceed 80% at 1 year, while high rates of pain control have been attained. There is insufficient evidence to suggest superiority of either single or multiple fraction regimens with respect to local control and pain control. Low rates of toxicity have been reported, assuming strict dose constraints are respected. Radiation myelopathy may be the most morbid toxicity, although the rates are low. The risk of vertebral compression fracture appears to be associated with higher doses per fraction such as those used in single-fraction regimens. The Spinal Instability Neoplastic Score should be considered when evaluating patients for spine SBRT, and prophylactic stabilisation may be warranted. Pain flare is a relatively common toxicity which may be mediated with prophylactic dexamethasone. Because of the treatment complexity and potentially serious toxicities, strict quality assurance should occur at the organizational, planning, dosimetric, and treatment delivery levels.
Conclusion:Both single and multifraction regimens are safe and efficacious in spine SBRT for spinal metastases. There may be advantages to hypofractionated treatment over single-fraction regimens with respect to toxicity. Ongoing investigation is underway to define optimal dose and fractionation schedules.
Keywords: Metastases, radiotherapy, SBRT, spine, stereotactic body radiation
INTRODUCTION
Spinal metastases are relatively common, with their incidence projected to increase as modern systemic therapies prolong patients’ overall survival. There is increasing focus on optimizing the rates of local control and pain relief, and the durability of both outcomes in appropriate patients. Spine stereotactic body radiation therapy (SBRT) is an emerging treatment option which may offer significant advantages in these aspects over conventional radiotherapy, with a growing body of evidence demonstrating safety and efficacy.[
SBRT utilizes ablative radiation doses in few fractions (typically 1–5) to the target. Intensity-modulated radiation therapy (IMRT)/volumetric modulated arc therapy (VMAT) and image-guidance allow the generation of steep dose gradients millimetres from the target, allowing spinal tumors to be treated with high biologically effective doses (BED) while protecting the adjacent spinal cord.[
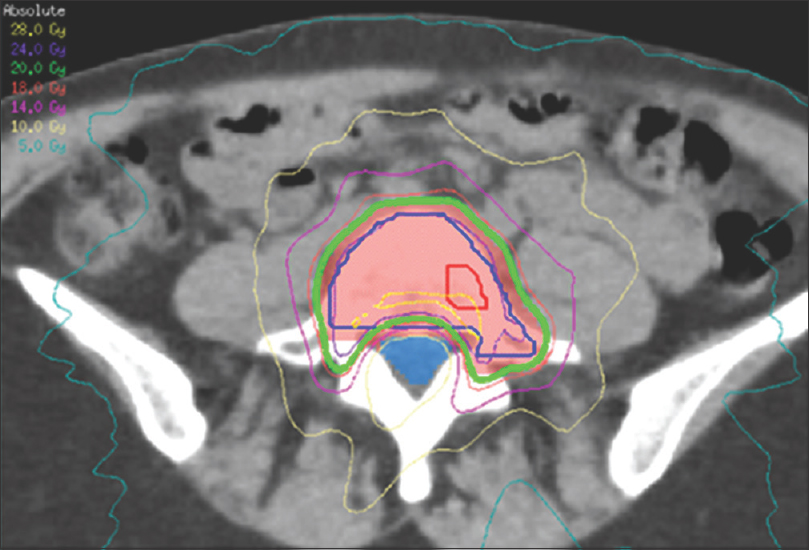
Figure 1
Axial CT scan demonstrating dosimetry for a lumbar spine lesion treated with VMAT, highlighting the steep dose gradient generated between the involved vertebral body and the thecal sac (blue). Adapted with permission[
High rates of local control and pain relief are achieved via high BEDs and possibly enhanced antitumor immune response.[
Because of the ablative doses involved, spine SBRT exposes patients to potential toxicities uncommonly observed in conventional radiotherapy such as radiation myelopathy and vertebral compression fracture (VCF), which fortunately occur at a relatively low rate.[
With increasing early phase data, spine SBRT has been widely adopted in the developed world. Nonetheless, there is a relative paucity of phase III data,[
PATIENT SELECTION
Predictive scoring systems have been developed to assess which patients benefit most from spine SBRT.[
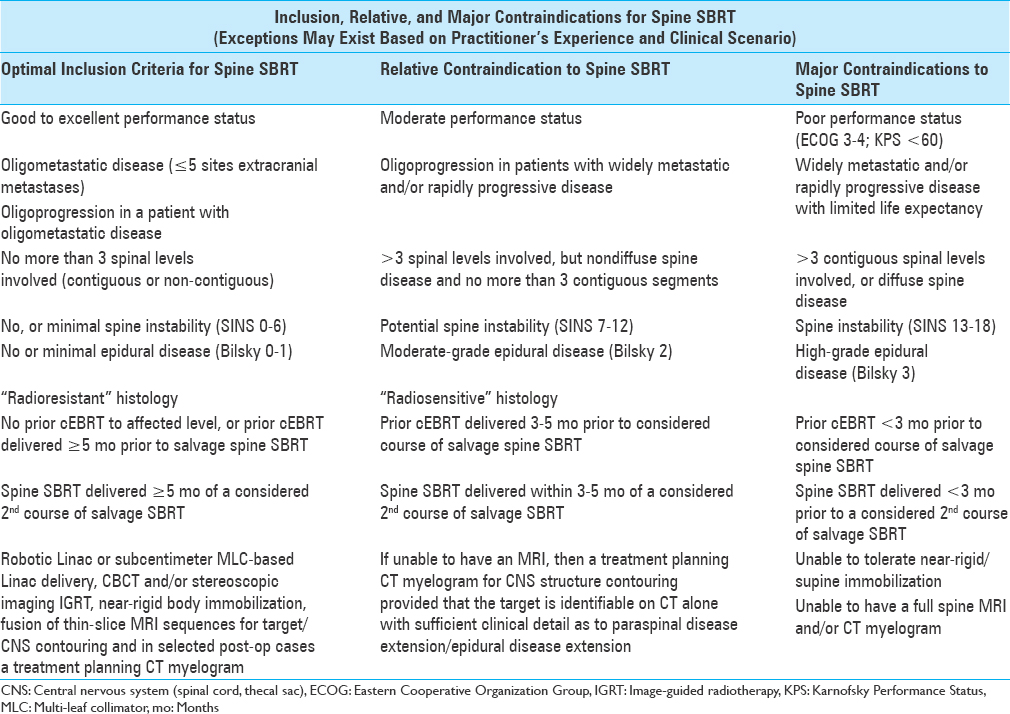
Table 1
Inclusion, relative, and major contraindications for spine SBRT. Adapted with permission[
In general terms, disease should be limited to three spinal levels at most, and ideally be at least 2–3 mm from the spinal cord itself to allow for a region of dose drop-off.[
SBRT should not be used for the primary treatment of spinal cord compression outside of a clinical trial setting because of the amount of time required for treatment planning/delivery and risk of under-dosing tumor immediately adjacent to the spinal cord.[
LOCAL CONTROL
The primary rationale for spine SBRT is to deliver a significantly higher BED, thereby improving local control compared with conventional radiotherapy, with published evidence demonstrating efficacy in the upfront, retreatment, and postoperative settings even in radioresistant histologies. Uncertainty exists regarding the optimal dose and fractionation regimen.
Upfront
Both prospective and retrospective data suggest high local control rates for spine SBRT in the upfront setting. Prospective studies reveal local control rates of between 80.5–95% at 12 to 18 months.[
A multi-institutional analysis of 301 patients treated upfront with spine SBRT with a variety of dose and fractionation schedules (from 8–60 Gy in 1–20 fractions) found local control at 2 years to be 83.9%, with a highly favorable toxicity profile.[
Postoperative
Spine SBRT is highly efficacious in the postoperative setting despite the potential for target volume uncertainty following surgical intervention. Tao et al. evaluated the outcomes of 66 patients treated via phase I or II trials, all following laminectomy or vertebrectomy. The 1 and 2-year local control rates were 85% and 79%, respectively.[
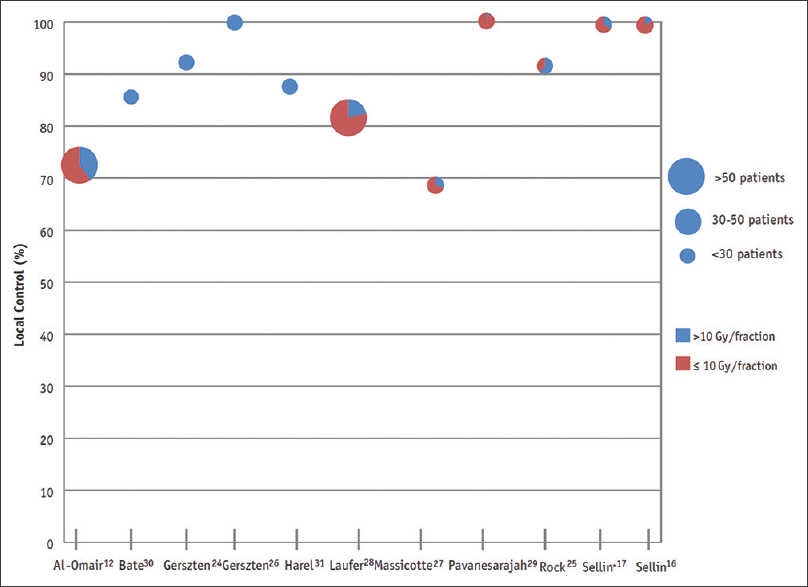
Figure 2
Local control following postoperative spine SBRT, as reviewed by Redmond et al. Dose per fraction is represented via each colour. Adapted with permission[
For patients with thecal sac compression, local control after spine SBRT was significantly improved when patients with high grade preoperative epidural disease (Bilsky grade 2 or 3) were downgraded to Bilsky grade 0 or 1, compared with those who had postoperative grade 2 disease.[
Retreatment
Local failure following conventional radiotherapy is an increasingly common scenario as modern systemic therapies increase patients’ overall survival. The challenge lies in delivering sufficient dose to control recurrent disease while maintaining an acceptable risk of spinal cord myelopathy.
The only multi-institutional study on retreatment is a pooled outcome analysis by Hashmi et al. demonstrating a high degree of efficacy and safety.[
The number of cases where local failure occurs following spine SBRT is not insignificant and the rationale for conventional radiotherapy in this setting is limited. Retreatment with fractionated spine SBRT safely confers high rates of local control. Thibault et al. reported 40 patients treated with a second course of SBRT with 20–35 Gy in 2–5 fractions; the median dose from initial SBRT was 24 Gy in 2 fractions. Local control at 1 year was 81%, while no cases of vertebral compression fracture nor radiation myelopathy were seen.[
Single fraction vs. multifraction
There is no prospective data demonstrating an optimal fractionation schedule for spine SBRT, with both single and multiple fraction regimens commonly utilised. Fractionation exploits intrinsic differences in radiosensitivity between tumour and spinal cord, allowing high tumouricidal doses to be safely delivered to lesions within the epidural space – where up to half of all recurrences occur following conventional radiotherapy.[
Conversely, single fraction treatments greater than 15 Gy per fraction are postulated to cause additional cell death via apoptosis. There is no conclusive data demonstrating an advantage in local control with either method – though it is possible that toxicities such as vertebral compression fractures, pain flare, and radiation myelopathy may be reduced with fractionation. Fractionated SBRT may be more appropriate for larger treatment volumes, or in the postoperative and re-irradiation settings.[
Published data comparing single to multifraction SBRT is limited to retrospective series. Heron et al. compared single fraction spine SBRT to a mean dose of 16.3 Gy with fractionated SBRT to mean doses of 20.6 Gy to 24.5 Gy in 3–5 fractions.[
Histology
Tumor histologies such as renal cell carcinoma, melanoma, and sarcoma have traditionally been regarded as radioresistant. Nonetheless, high local control rates have been observed for these tumor types when treated with spine SBRT.
For sarcoma metastases treated with 24–28.5 Gy in 1–3 fractions, the 12-month local control rate was 87.9%, which is significantly higher than historical 1-year local control rates of 50–77% with conventional radiotherapy.[
For renal cell carcinoma, 1- and 2-year local control rates of 83.5% and 66.2%, respectively, have been reported. The median dose used was 24 Gy in 2 fractions.[
Gerszten et al. demonstrated efficacy for melanoma spinal metastases using single fraction treatments of 17.5–25 Gy.[
It can be seen that SBRT provides superior local control compared with conventional radiotherapy for radioresistant histologies. The impact of the number of fractions is unknown, though it is likely that a high BED is required to adequately ablate these metastases. SBRT allows for feasible delivery of such high doses, resulting in the high rates of local control observed.
PAIN CONTROL
Spine SBRT is also efficacious for symptom palliation, an important outcome for patients of reasonable prognosis where the goal is to prolong progression-free survival. Conventional radiotherapy is associated with partial pain relief rates of approximately 60%, with complete pain response rates of 24%.[
Reported pain outcomes in 149 prospectively treated patients with a mean baseline pain score of 3.4 on an 11-point Brief Pain Inventory scale fell to 2.1 at 4 weeks post-SBRT. At baseline, 34% of the patients rated pain as moderate to severe, falling to 15% at 2 months. At 6 months, 53.9% of patients reported no pain.[
High rates of pain response in the retreatment setting have also been reported, with 84% of the patients experiencing improvement in pain and 74% of patients becoming pain free. Mean pain scores (measured via visual analog scale score of 0–100) were 51 at baseline, and 17.5 at 1 year.[
COMPLICATIONS FROM SPINE SBRT
The available literature suggests low complication rates for spine SBRT, highlighting its utility in the palliative setting. A phase II feasibility study of spine SBRT found no cases of grade 4–5 toxicity, a 2.3% rate of grade 3 toxicity, and a 25% rate of grade 1–2 toxicity among 44 patients.[
The specific toxicities reported in the literature will be outlined, with the most prominent issues being vertebral compression fracture (VCF) and radiation myelopathy.
Vertebral compression fracture
Radiation-induced tissue necrosis may be the underlying mechanism leading to vertebral instability and subsequent fracture.[
Analyses by Sahgal et al. and Cunha et al. found that 3 components of SINS were risk factors for VCF following spine SBRT, including pre-existing VCF, lytic tumor type, and spinal deformity. Furthermore, the risk of VCF was found to progressively decrease as dose per fraction decreased, suggesting that high doses per fraction are inappropriate for patients with high SINS.[
Chang et al. reviewed 24 studies which reported VCF rates and found a crude VCF rate of 13.7%. Where VCF occurred, the rate of salvage intervention was 45%. Of these, 50% were cement augmentation, 2% percutaneous instrumentation, and 48% decompression and reconstruction.[
Careful consideration should be given to response assessment posttreatment, as osteoradionecrosis, fibrosis, or VCF can be mistaken for local progression. Thus, biopsy may be required in some cases where local progression is suspected following SBRT.[
VCF is thus a well-recognized complication of spine SBRT. SINS should always be considered and caution should be taken when considering high-dose single fractions.
Myelopathy
Radiation myelopathy may be the most feared of all potential complications from spine SBRT. Permanent neurological deficit resulting from palliative treatment is a catastrophic outcome, particularly because patients selected for SBRT typically have a reasonable prognosis.
Prospective studies reveal rates of radiation myelopathy between 0% and 3%,[
Spinal cord tolerance can be affected by small dose inhomogeneities, and particular attention is paid to the point maximum dose within the spinal cord.[
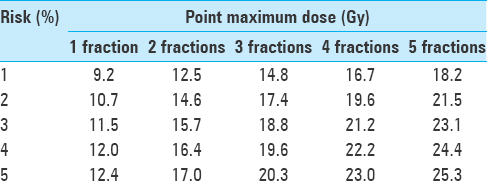
Table 2
Point maximum doses to spinal cord, categorized by myelopathy risk and fractionation schedule. Adapted with permission[
Sahgal et al. analyzed patients re-irradiated with SBRT following conventional external beam treatment.[
It is apparent there is a correlation between dose/fractionation and the risk of myelopathy, which needs to be considered when selecting treatment regimens. Treatment and dose constraints applied should be balanced against the likely greater risks of neurological deficit caused by uncontrolled tumor progression.
Pain flare
Acute pain flare occurs more commonly for spine SBRT compared with conventional RT. The onset is typically within days of treatment. Its incidence has been reported as high as 68%, though evidence suggests that the risk of pain flare significantly reduces with increasing fraction number.[
Standard management for pain flare consists of dexamethasone, with recent evidence suggesting a benefit to prophylactic use. Prophylactic dexamethasone reduces the incidence of pain flare to 19.2% at a dose of 4 mg daily for 5 days, commencing on the day of the first fraction.[
Esophageal toxicity
Esophageal toxicity can occur in patients receiving spine SBRT though rates are low, even when target volumes directly abut the eosphagus. Two studies report low rates of esophageal toxicity with 24 Gy single-fraction regimens. Cox et al. found a 1.5% rate of grade 3 or higher acute esophageal toxicity when treating spinal metastases directly abutting the esophagus. The rate of grade 3 late toxicity (stenosis, fistula, or ulcer) was 5%.[
A report of lung and liver SBRT treated to a median dose of 50 Gy in 5 fractions found a grade 3 esophageal toxicity rate of 3.8%. It was postulated that the use of bevazicumab contributed to esophageal toxicity.[
Damage to great vessels
Rates of radiotherapy-related complications for the aorta are generally low, and other organ constraints typically take precedence. Xue et al. reported 625 cases treated with SBRT over 1–5 fractions with aorta or major vessel contours. It was not reported how many patients specifically received spine SBRT versus non-spine SBRT sites, though no grade 3 or higher toxicity was observed. The authors’ recommended dose constraints were all higher than typical prescription doses for spine SBRT, suggesting that this complication is unlikely.[
QUALITY ASSURANCE
The planning and treatment process for spine SBRT is complex and the potential for serious toxicity has been outlined. A focus on optimizing treatment quality is needed to achieve the favorable rates of local control and toxicity demonstrated in the literature. Multiple factors within each institution need to be considered when assessing its ability to deliver high quality treatment.
Organizational quality
There is international consensus on the need for specialized teams of physicians, physicists, and radiation therapists with SBRT-specific training.[
Planning dosimetric quality
Magnetic resonance imaging (MRI) is a prerequisite. T1 and T2 noncontrast images are recommended, with T2 especially useful for delineating spinal cord/thecal sac and paraspinal disease extension. Slice thickness should be fine at 1–2 millimetres. In the postoperative setting, particular attention should be paid to the preoperative imaging findings, as this is associated with patterns of local recurrence.[
Consensus guidelines exist regarding volume definition for upfront and retreatment spine SBRT based on expert recommendations. In general, coverage of the entire vertebral body is recommended unless tumor involves only the lamina, spinous process, or in select cases, the pedicle. This is detailed more comprehensively in the original article by Cox et al.[
There is less certainty regarding the target volumes in the postoperative setting, though a recent postoperative review paper recommends image fusion of preoperative scans, utilization of documented operative findings, and direct communication with the surgeon to guide target delineation. CT myelogram can be used where surgical hardware obscures visualization of spinal cord/cauda equina.[
Planning Target Volumes (PTV) and Planning organ-at-Risk Volumes (PRV) are required to account for intrafraction movement and setup error, though these can be minimized with appropriate image guidance and correction techniques to approximately 2 mm.[
Planning and delivery quality
Near rigid immobilization systems are recommended to minimize intrafraction motion, given the typically steep dose gradients that are generated between the spinal cord and target volume.[
Commonly utilized treatment machines include linear accelerators with multileaf collimators and treatment couches capable of fine translational and rotational repositioning, and a linear accelerator mounted on a robotic arm (Cyberknife, Accuray, Sunnyvale, CA, USA). Intensity modulated protons have also been deemed adequate on a dosimetric basis, although these are not widely available.[
ONGOING RANDOMIZED TRIALS
Further randomized studies are underway to provide higher quality data for spine SBRT in a variety of treatment scenarios.
To evaluate the role of spine SBRT in the upfront setting, the Canadian Cancer Trials Group (NCIC) are recruiting for a randomized phase 2 study (NCT02512965) comparing spine SBRT with conventional radiotherapy with respect to pain palliation, via regimens of 24 Gy in 2 fractions for SBRT and 20 Gy in 5 fractions for conventional radiotherapy. Along similar lines, the Radiation Therapy Oncology Group (RTOG) are recruiting for a phase III randomized comparison of single-fraction SBRT to single-fraction conventional radiotherapy (NCT00922974, or RTOG 06-31).
For patients deemed at high risk of VCF following spine SBRT, an MD Anderson-based phase 2 study (NCT02387905) is underway to evaluate the efficacy of prophylactic vertebral cement augmentation. If efficacy is demonstrated, this may spare patients from subsequent fracture-related morbidity and additional surgical procedures.
For patients with early spinal cord compression and minor neurological deficits, investigators in Denmark are comparing spine SBRT of 16 Gy in 1 fraction with decompressive surgery and postoperative conventional radiotherapy, with the primary endpoint being the ability to walk at 6 weeks (NCT02167633). If efficacy is demonstrated, some patients may be spared the morbidity of surgery.
To investigate optimal dose and fractionation regimens with respect to local control, the Memorial Sloan Kettering Cancer Center is performing two randomized phase 3 trials comparing single fraction SBRT of 24 Gy with hypofractionated SBRT of 27 Gy in 3 fractions. The first (NCT02320825) is specific to patients in the postoperative spine setting, whereas the second (NCT01223248) includes a variety of metastatic sites including bone, spine, soft tissue, and lymph nodes. With an overall enrolment of 220 patients, spine-specific data will be of interest.
CONCLUSION
Spine SBRT is a highly efficacious and safe treatment in the treatment of spinal tumors in appropriately selected patients, though focus is needed on ensuring treatment quality across a range of domains.
High quality randomized data regarding optimal dose and fractionation is lacking, though the available evidence suggests hypofractionated spine SBRT may have advantages with respect to toxicity when compared with single fraction regimens. This should be considered when evaluating each patient on a case-by-case basis. Ongoing investigation is underway to further guide clinicians’ dose/fractionation decisions.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Al-Omair A, Masucci L, Masson-Cote L, Campbell M, Atenafu EG, Parent A. Surgical resection of epidural disease improves local control following postoperative spine stereotactic body radiotherapy. Neurooncology. 2013. 15: 1413-9
2. Al-Omair A, Smith R, Kiehl TR, Lao L, Yu E, Massicotte EM. Radiation-induced vertebral compression fracture following spine stereotactic radiosurgery: Clinicopathological correlation. J Neurosurg Spine. 2013. 18: 430-5
3. Amdur RJ, Bennett J, Olivier K, Wallace A, Morris CG, Liu C. A prospective, phase II study demonstrating the potential value and limitation of radiosurgery for spine metastases. Am J Clin Oncol. 2009. 32: 515-20
4. Anwar M, Barani IJ. Role of stereotactic body radiotherapy in spinal metastasis and subsequent fracture risk: Identifying and treating the at-risk patient. CNS Oncol. 2013. 2: 437-45
5. Bilsky MH, Laufer I, Fourney DR, Groff M, Schmidt MH, Varga PP. Reliability analysis of the epidural spinal cord compression scale. J Neurosurg Spine. 2010. 13: 324-8
6. Brown JM, Carlson DJ, Brenner DJ. The tumor radiobiology of SRS and SBRT: Are more than the 5 Rs involved?. Int J Radiat Oncol Biol Phys. 2014. 88: 254-262
7. Chan MW, Thibault I, Atenafu EG, Yu E, John Cho BC, Letourneau D. Patterns of epidural progression following postoperative spine stereotactic body radiotherapy: Implications for clinical target volume delineation. J Neurosurg Spine. 2016. 24: 652-9
8. Chang JH, Shin JH, Yamada Y, Mesfin A, Fehlings MG, Rhines LD. Stereotactic Body Radiotherapy for Spinal Metastases: What are the Risks and how do we Minimize Them?. Spine. 2016. 41: S238-45
9. Chiang A, Zeng L, Zhang L, Lochray F, Korol R, Loblaw A. Pain flare is a common adverse event in steroid-naive patients after spine stereotactic body radiation therapy: A prospective clinical trial. Int J Radiat Oncol Biol Phys. 2013. 86: 638-42
10. Choi CY, Adler JR, Gibbs IC, Chang SD, Jackson PS, Minn AY. Stereotactic radiosurgery for treatment of spinal metastases recurring in close proximity to previously irradiated spinal cord. Int J Radiat Oncol Biol Phys. 2010. 78: 499-506
11. Chow E, Harris K, Fan G, Tsao M, Sze WM. Palliative radiotherapy trials for bone metastases: A systematic review. J Clin Oncol. 2007. 25: 1423-36
12. Corbin KS, Hellman S, Weichselbaum RR. Extracranial oligometastases: A subset of metastases curable with stereotactic radiotherapy. J Clin Oncol. 2013. 31: 1384-90
13. Cox BW, Jackson A, Hunt M, Bilsky M, Yamada Y. Esophageal toxicity from high-dose, single-fraction paraspinal stereotactic radiosurgery. Int J Radiat Oncol Biol Phys. 2012. 83: e661-7
14. Cox BW, Spratt DE, Lovelock M, Bilsky MH, Lis E, Ryu S. International Spine Radiosurgery Consortium consensus guidelines for target volume definition in spinal stereotactic radiosurgery. Int J Radiat Oncol Biol Phys. 2012. 83: e597-605
15. Cunha MV, Al-Omair A, Atenafu EG, Masucci GL, Letourneau D, Korol R. Vertebral compression fracture (VCF) after spine stereotactic body radiation therapy (SBRT): Analysis of predictive factors. Int J Radiat Oncol Biol Phys. 2012. 84: e343-9
16. Dahele M, Fehlings MG, Sahgal A. Stereotactic radiotherapy: An emerging treatment for spinal metastases. Can J Neurol Sci. 2011. 38: 247-50
17. Dahele M, Hatton M, Slotman B, Guckenberger M. Stereotactic body radiotherapy: A survey of contemporary practice in six selected European countries. Acta Oncol. 2015. 54: 1237-41
18. Degen JW, Gagnon GJ, Voyadzis JM, McRae DA, Lunsden M, Dieterich S. CyberKnife stereotactic radiosurgical treatment of spinal tumors for pain control and quality of life. J Neurosurg Spine. 2005. 2: 540-9
19. Finnigan R, Burmeister B, Barry T, Jones K, Boyd J, Pullar A. Technique and early clinical outcomes for spinal and paraspinal tumours treated with stereotactic body radiotherapy. J Clin Neurosci. 2015. 22: 1258-63
20. Finnigan R, Lamprecht B, Barry T, Jones K, Boyd J, Pullar A. Inter- and intra-fraction motion in stereotactic body radiotherapy for spinal and paraspinal tumours using cone-beam CT and positional correction in six degrees of freedom. J Med Imaging Radiat Oncol. 2016. 60: 112-8
21. Folkert MR, Bilsky MH, Tom AK, Oh JH, Alektiar KM, Laufer I. Outcomes and toxicity for hypofractionated and single-fraction image-guided stereotactic radiosurgery for sarcomas metastasizing to the spine. Int J Radiat Oncol Biol Phys. 2014. 88: 1085-91
22. Foote M, Bailey M. Guidelines for safe practice of stereotactic body (ablative) radiation therapy. J Med Imaging Radiat Oncol. 2015. 59: 646-53
23. Garg AK, Shiu AS, Yang J, Wang XS, Allen P, Brown BW. Phase 1/2 trial of single-session stereotactic body radiotherapy for previously unirradiated spinal metastases. Cancer. 2012. 118: 5069-77
24. Germano IM, Carai A, Pawha P, Blacksburg S, Lo YC, Green S. Clinical outcome of vertebral compression fracture after single fraction spine radiosurgery for spinal metastases. Clin Exp Metastasis. 2016. 33: 143-9
25. Gerszten PC, Burton SA, Quinn AE, Agarwala SS, Kirkwood JM. Radiosurgery for the treatment of spinal melanoma metastases. Stereotact Funct Neurosurg. 2005. 83: 213-21
26. Gerszten PC, Sahgal A, Sheehan JP, Kersh R, Chen S, Flickinger JC. A multi-national report on methods for institutional credentialing for spine radiosurgery. Radiat Oncol. 2013. 8: 158-
27. Ghia AJ, Chang EL, Bishop AJ, Pan HY, Boehling NS, Amini B. Single-fraction versus multifraction spinal stereotactic radiosurgery for spinal metastases from renal cell carcinoma: Secondary analysis of Phase I/II trials. J Neurosurg Spine. 2016. 24: 829-36
28. Gomez DR, Hunt MA, Jackson A, O’Meara WP, Bukanova EN, Zelefsky MJ. Low rate of thoracic toxicity in palliative paraspinal single-fraction stereotactic body radiation therapy. Radiother Oncol. 2009. 93: 414-8
29. Guckenberger M, Mantel F, Gerszten PC, Flickinger JC, Sahgal A, Letourneau D. Safety and efficacy of stereotactic body radiotherapy as primary treatment for vertebral metastases: A multi-institutional analysis. Radiat Oncol. 2014. 9: 226-
30. Guckenberger M, Sweeney RA, Flickinger JC, Gerszten PC, Kersh R, Sheehan J. Clinical practice of image-guided spine radiosurgery-results from an international research consortium. Radiat Oncol. 2011. 6: 172-
31. Hall WA, Stapleford LJ, Hadjipanayis CG, Curran WJ, Crocker I, Shu HK. Stereotactic body radiosurgery for spinal metastatic disease: An evidence-based review. Int J Surg Oncol 2011. 2011. p.
32. Hashmi A, Guckenberger M, Kersh R, Gerszten PC, Mantel F, Grills IS. Re-irradiation stereotactic body radiotherapy for spinal metastases: A multi-institutional outcome analysis. J Neurosurg Spine. 2016. 25: 646-53
33. Heron DE, Rajagopalan MS, Stone B, Burton S, Gerszten PC, Dong X. Single-session and multisession CyberKnife radiosurgery for spine metastases-University of Pittsburgh and Georgetown University experience. J Neurosurg Spine. 2012. 17: 11-8
34. Husain ZA, Thibault I, Letourneau D, Ma L, Keller H, Suh J. Stereotactic body radiotherapy: A new paradigm in the management of spinal metastases. CNS Oncol. 2013. 2: 259-70
35. Hyde D, Lochray F, Korol R, Davidson M, Wong CS, Ma L. Spine stereotactic body radiotherapy utilizing cone-beam CT image-guidance with a robotic couch: Intrafraction motion analysis accounting for all six degrees of freedom. Int J Radiat Oncol Biol Phys. 2012. 82: e555-62
36. Jabbari S, Gerszten PC, Ruschin M, Larson DA, Lo SS, Sahgal A. Stereotactic Body Radiotherapy for Spinal Metastases: Practice Guidelines, Outcomes, and Risks. Cancer J. 2016. 22: 280-9
37. Jain AK, Yamada YJ. The role of stereotactic body radiotherapy and stereotactic radiosurgery in the re-irradiation of metastatic spinal tumors. Exp Rev Anticancer Ther. 2014. 14: 1141-52
38. Khan L, Chiang A, Zhang L, Thibault I, Bedard G, Wong E. Prophylactic dexamethasone effectively reduces the incidence of pain flare following spine stereotactic body radiotherapy (SBRT): A prospective observational study. Support Care Cancer. 2015. 23: 2937-43
39. Kim MS, Kim W, Park IH, Kim HJ, Lee E, Jung JH. Radiobiological mechanisms of stereotactic body radiation therapy and stereotactic radiation surgery. Radiat Oncol J. 2015. 33: 265-75
40. Kirkbride PBurton KCassoni ACooper TEccles CLast accessed on 2016 Jun 12. Available from: http://www.cancerresearchuk.org/cancer-info/prod_consump/groups/cr_common/@nre/@new/@gen/documents/generalcontent/cr_074729.pdf .
41. Laufer I, Iorgulescu JB, Chapman T, Lis E, Shi W, Zhang Z. Local disease control for spinal metastases following “separation surgery” and adjuvant hypofractionated or high-dose single-fraction stereotactic radiosurgery: Outcome analysis in 186 patients. J Neurosurg Spine. 2013. 18: 207-14
42. Li W, Sahgal A, Foote M, Millar BA, Jaffray DA, Letourneau D. Impact of immobilization on intrafraction motion for spine stereotactic body radiotherapy using cone beam computed tomography. Int J Radiat Oncol Biol Phys. 2012. 84: 520-6
43. Lo SS, Foote M, Siva S, Slotman BJ, Teh BS, Guckenberger M. Technical know-how in stereotactic ablative radiotherapy (SABR). J Med Radiat Sci. 2016. 63: 5-8
44. Lo SS, Sahgal A, Chang EL, Mayr NA, Teh BS, Huang Z. Serious complications associated with stereotactic ablative radiotherapy and strategies to mitigate the risk. Clin Oncol. 2013. 25: 378-87
45. Lo SS, Sahgal A, Wang JZ, Mayr NA, Sloan A, Mendel E. Stereotactic body radiation therapy for spinal metastases. Discov Med. 2010. 9: 289-96
46. Lo SS, Slotman BJ, Lock M, Nagata Y, Guckenberger M, Siva S. The development of stereotactic body radiotherapy in the past decade: A global perspective. Future Oncol. 2015. p.
47. Moulding HD, Elder JB, Lis E, Lovelock DM, Zhang Z, Yamada Y. Local disease control after decompressive surgery and adjuvant high-dose single-fraction radiosurgery for spine metastases. J Neurosurg Spine. 2010. 13: 87-93
48. Moussazadeh N, Lis E, Katsoulakis E, Kahn S, Svoboda M, DiStefano NM. Five-Year Outcomes of High-Dose Single-Fraction Spinal Stereotactic Radiosurgery. Int J Radiat Oncol Biol Phys. 2015. 93: 361-7
49. Pan HY, Allen PK, Wang XS, Chang EL, Rhines LD, Tatsui CE. Incidence and predictive factors of pain flare after spine stereotactic body radiation therapy: Secondary analysis of phase 1/2 trials. Int J Radiat Oncol Biol Phys. 2014. 90: 870-6
50. Potters L, Kavanagh B, Galvin JM, Hevezi JM, Janjan NA, Larson DA. American Society for Therapeutic Radiology and Oncology (ASTRO) and American College of Radiology (ACR) practice guideline for the performance of stereotactic body radiation therapy. Int J Radiat Oncol Biol Phys. 2010. 76: 326-32
51. Redmond KJ, Lo SS, Fisher C, Sahgal A. Postoperative Stereotactic Body Radiation Therapy (SBRT) for Spine Metastases: A Critical Review to Guide Practice. Int J Radiat Oncol Biol Phys. 2016. 95: 1414-28
52. Redmond KJ, Sahgal A, Foote M, Knisely J, Gerszten PC, Chao ST. Single versus multiple session stereotactic body radiotherapy for spinal metastasis: The risk-benefit ratio. Future Oncol. 2015. 11: 2405-15
53. Rose PS, Laufer I, Boland PJ, Hanover A, Bilsky MH, Yamada J. Risk of fracture after single fraction image-guided intensity-modulated radiation therapy to spinal metastases. J Clin Oncol. 2009. 27: 5075-9
54. Ryu S, Pugh SL, Gerszten PC, Yin FF, Timmerman RD, Hitchcock YJ. RTOG 0631 phase 2/3 study of image guided stereotactic radiosurgery for localized (1-3) spine metastases: Phase 2 results. Pract Radiat Oncol. 2014. 4: 76-81
55. Sahgal A, Ames C, Chou D, Ma L, Huang K, Xu W. Stereotactic body radiotherapy is effective salvage therapy for patients with prior radiation of spinal metastases. Int J Radiat Oncol Biol Phys. 2009. 74: 723-31
56. Sahgal A, Atenafu EG, Chao S, Al-Omair A, Boehling N, Balagamwala EH. Vertebral compression fracture after spine stereotactic body radiotherapy: A multi-institutional analysis with a focus on radiation dose and the spinal instability neoplastic score. J Clin Oncol. 2013. 31: 3426-31
57. Sahgal A, Ma L, Fowler J, Weinberg V, Gibbs I, Gerszten PC. Impact of dose hot spots on spinal cord tolerance following stereotactic body radiotherapy: A generalized biological effective dose analysis. Technol Cancer Res Treat. 2012. 11: 35-40
58. Sahgal A, Ma L, Gibbs I, Gerszten PC, Ryu S, Soltys S. Spinal cord tolerance for stereotactic body radiotherapy. International journal of radiation oncology, biology, physics. 2010. 77: 548-553
59. Sahgal A, Ma L, Weinberg V, Gibbs IC, Chao S, Chang UK. Reirradiation human spinal cord tolerance for stereotactic body radiotherapy. Int J Radiat Oncol Biol Phys. 2012. 82: 107-16
60. Sahgal A, Roberge D, Schellenberg D, Purdie TG, Swaminath A, Pantarotto J. The Canadian Association of Radiation Oncology scope of practice guidelines for lung, liver and spine stereotactic body radiotherapy. Clin Oncol. 2012. 24: 629-39
61. Sahgal A, Weinberg V, Ma L, Chang E, Chao S, Muacevic A. Probabilities of radiation myelopathy specific to stereotactic body radiation therapy to guide safe practice. Int J Radiat Oncol Biol Phys. 2013. 85: 341-7
62. Stephans KL, Djemil T, Diaconu C, Reddy CA, Xia P, Woody NM. Esophageal dose tolerance to hypofractionated stereotactic body radiation therapy: Risk factors for late toxicity. Int J Radiat Oncol Biol Phys. 2014. 90: 197-202
63. Tang C, Hess K, Bishop AJ, Pan HY, Christensen EN, Yang JN. Creation of a Prognostic Index for Spine Metastasis to Stratify Survival in Patients Treated With Spinal Stereotactic Radiosurgery: Secondary Analysis of Mature Prospective Trials. Int J Radiat Oncol Biol Phys. 2015. 93: 118-25
64. Tao R, Bishop AJ, Brownlee Z, Allen PK, Settle SH, Chang EL. Stereotactic Body Radiation Therapy for Spinal Metastases in the Postoperative Setting: A Secondary Analysis of Mature Phase 1-2 Trials. Int J Radiat Oncol Biol Phys. 2016. 95: 1405-13
65. Thibault I, Al-Omair A, Masucci GL, Masson-Cote L, Lochray F, Korol R. Spine stereotactic body radiotherapy for renal cell cancer spinal metastases: Analysis of outcomes and risk of vertebral compression fracture. J Neurosurg Spine. 2014. 21: 711-8
66. Thibault I, Campbell M, Tseng CL, Atenafu EG, Letourneau D, Yu E. Salvage Stereotactic Body Radiotherapy (SBRT) Following In-Field Failure of Initial SBRT for Spinal Metastases. Int J Radiat Oncol Biol Phys. 2015. 93: 353-60
67. Thibault I, Chang EL, Sheehan J, Ahluwalia MS, Guckenberger M, Sohn MJ. Response assessment after stereotactic body radiotherapy for spinal metastasis: A report from the SPIne response assessment in Neuro-Oncology (SPINO) group. Lancet Oncol. 2015. 16: e595-603
68. Tseng CL, Sussman MS, Atenafu EG, Letourneau D, Ma L, Soliman H. Magnetic resonance imaging assessment of spinal cord and cauda equina motion in supine patients with spinal metastases planned for spine stereotactic body radiation therapy. Int J Radiat Oncol Biol Phys. 2015. 91: 995-1002
69. van Mourik AM, Elkhuizen PH, Minkema D, Duppen JC, van Vliet-Vroegindeweij C. Multiinstitutional study on target volume delineation variation in breast radiotherapy in the presence of guidelines. Radiother Oncol. 2010. 94: 286-91
70. Wang H, Shiu A, Wang C, O’Daniel J, Mahajan A, Woo S. Dosimetric effect of translational and rotational errors for patients undergoing image-guided stereotactic body radiotherapy for spinal metastases. Int J Radiat Oncol Biol Phys. 2008. 71: 1261-71
71. Wang XS, Rhines LD, Shiu AS, Yang JN, Selek U, Gning I. Stereotactic body radiation therapy for management of spinal metastases in patients without spinal cord compression: A phase 1-2 trial. Lancet Oncol. 2012. 13: 395-402
72. Xue J, Kubicek G, Patel A, Goldsmith B, Asbell SO, LaCouture TA. Validity of Current Stereotactic Body Radiation Therapy Dose Constraints for Aorta and Major Vessels. Semin Radiat Oncol. 2016. 26: 135-9