- Department of Neurosurgery, University of Kansas Medical Center, Kansas City, KS, USA
- Department of Mechanical Engineering, University of Kansas, Lawrence, KS, USA
Correspondence Address:
Domenico A. Gattozzi
Department of Neurosurgery, University of Kansas Medical Center, Kansas City, KS, USA
DOI:10.4103/sni.sni_273_18
Copyright: © 2018 Surgical Neurology International This is an open access journal, and articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as appropriate credit is given and the new creations are licensed under the identical terms.How to cite this article: Domenico A. Gattozzi, Lisa A. Friis, Paul M. Arnold. Surgery for traumatic fractures of the upper thoracic spine (T1–T6). 19-Nov-2018;9:231
How to cite this URL: Domenico A. Gattozzi, Lisa A. Friis, Paul M. Arnold. Surgery for traumatic fractures of the upper thoracic spine (T1–T6). 19-Nov-2018;9:231. Available from: http://surgicalneurologyint.com/surgicalint-articles/9070/
Abstract
Background:The management of traumatic upper thoracic spine fractures (T1–T6) is complex due to the unique biomechanical/physiological characteristics of these levels and the nature of the injuries. They are commonly associated with multiple other traumatic injuries and severe spinal cord injuries. We describe the safety and efficacy of surgery for achieving stability and maintaining reduction of upper thoracic T1–T6 spine fractures.
Methods:We retrospectively analyzed a series of traumatic unstable upper thoracic (T1–T6) spine fractures treated at one institution between 1993 and 2016. All patients were assessed neurologically and underwent complete preoperative radiographic analysis of their T1–T6 spine fractures including assessment of instability. Neurological and radiographic outcomes including fusion rates, kyphotic deformity, and successful reduction of the fracture were evaluated along with hospital length of stay (LOS), intensive care unit LOS, and overall complication rates.
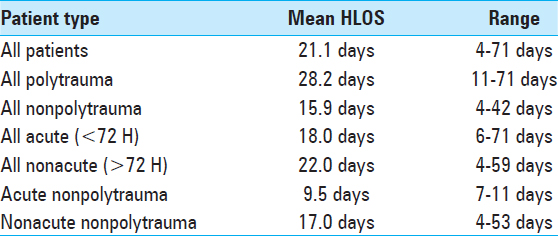
Results:There were 43 patients (29 males, 14 females) with an average age of 37.7 years. Between 1993 and 1999, 8 patients were treated with hook/rod constructs, whereas between 1995 and 2016, 35 patients received pedicle screw fixation utilizing intraoperative fluoroscopy or computed tomography (CT) navigation. Forty-three patients had a total of 178 levels fused. In this series, there were no intraoperative vascular or neurological complications. Instrumentation was removed in five patients due to pain, wound infection, or hardware failure. The mean hospital LOS was 21.1 days (range 4–59 days), and there was a 95% fusion rate based on follow-up imaging (X-rays or CT scan).
Conclusions:Surgical treatment of upper thoracic spine fractures (T1–T6), although complex, is safe and effective. Reduction and fixation of these fractures decreases the risk of further neurological complications, allows for earlier mobilization, and correlates with shorter hospital LOS and improved outcomes.
Keywords: Fracture, spine, surgery, thoracic, trauma
INTRODUCTION
The management of fractures of the upper thoracic spine (T1–T6) is technically and biomechanically challenging due to the transition from the mobile cervical spine to the fixed thoracic spine.[
MATERIALS AND METHODS
Patient demographics
Forty-three patients with T1–T6 fractures were included in this study (1993–2016). The AO/Magerl and thoracolumbar injury classification system (TLICS) classification system were used to describe fracture patterns.[
RESULTS
Patient demographics
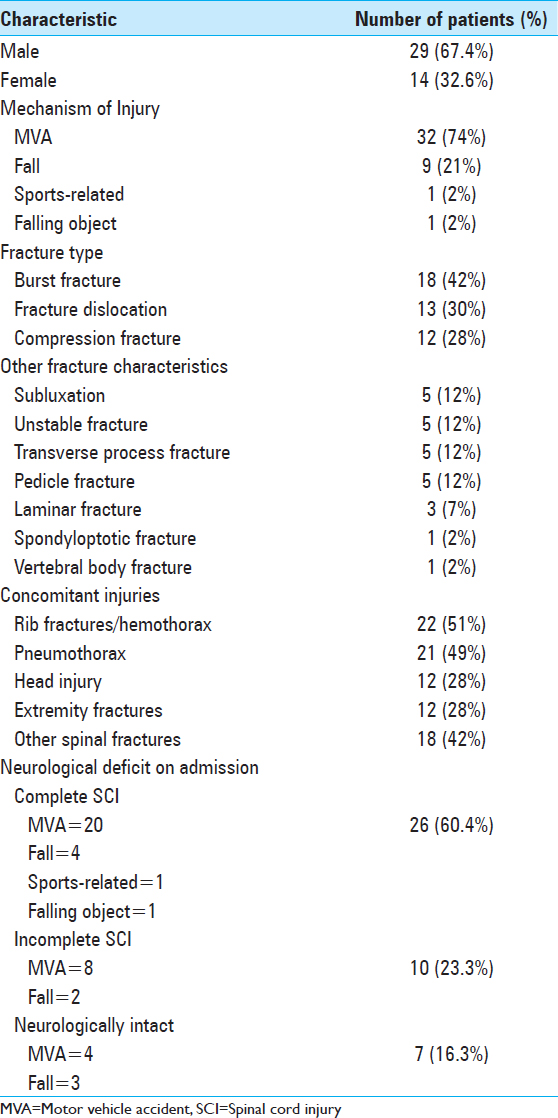
Forty-three patients were included in this study (1993-2016). Average age was 37.7 years old (range 14-83 years). For full patient demographics, fracture characteristics, and preoperative neurological assessments, see
Timing of surgery dependent on neurological deficit
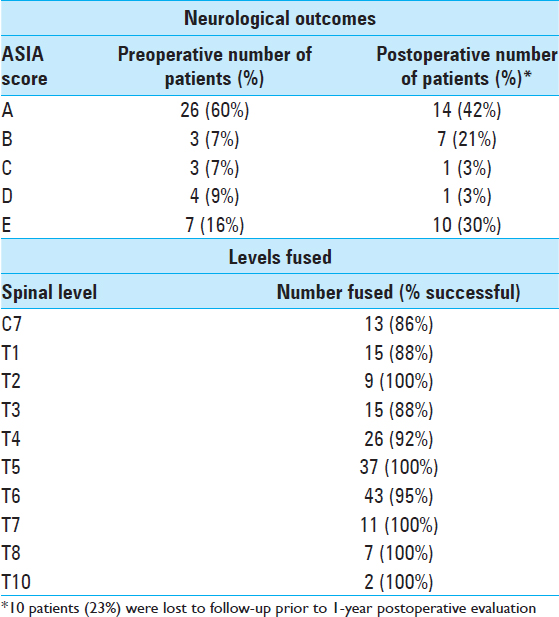
Patients with incomplete injuries (10, 23.3%) had emergent surgery within 72 hours, whereas those with no deficits or with complete spinal cord injuries had surgery once medically stable from comorbid injuries. All patients had surgery within 1 month after injury. The average time to surgery was 7.78 days. Twenty-three patients (53.5%) also underwent cord decompression (laminectomy and/or ventral decompression) in addition to instrumented fusion. Patients were mobilized on postoperative day 1. Neurologic deficits were graded with the ASIA scale at 3, 6, and 12 months postoperatively. Of 33 patients, 11 (33.3%) improved 1 year postoperatively [
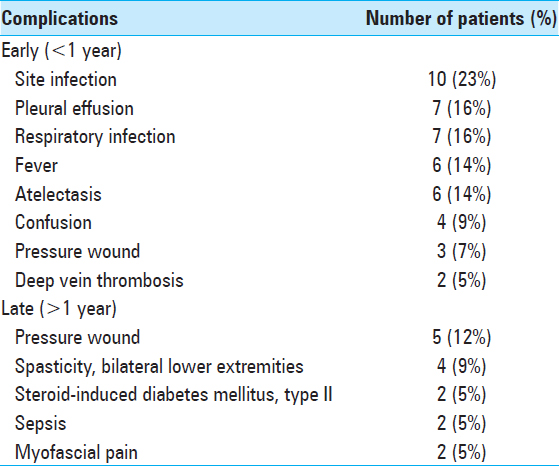
Complications and indications for reoperation
There were no intraoperative vascular or neurological complications. Other complications were noted and divided into early and late, depending on whether they occurred before or after 1 year from the time of surgery [
Assessment of radiological fusion rates/outcomes
The fusion rate across all levels was 95%. There were no broken screws or instances of instrumentation failure. Given the increased cost and higher level of radiation to the patients, we do not routinely perform CT scans after spinal fusion at our institution and prefer X-rays. Postoperative CT analysis was reserved for patients with no evidence of fusion on plain films or where there was concern regarding the instrumented construct. Using the Cobb method, the mean preoperative kyphosis was 22.25°. At 3 and 12 months, the mean kyphosis calculations were 13.5° and 17.6°, respectively. Seven of the 43 patients died an average of 4.2 years postoperatively. The deaths were due to causes unrelated to the upper thoracic spine surgery. These patients averaged 47 years of age (range 23–78 years). For a sample patient case see
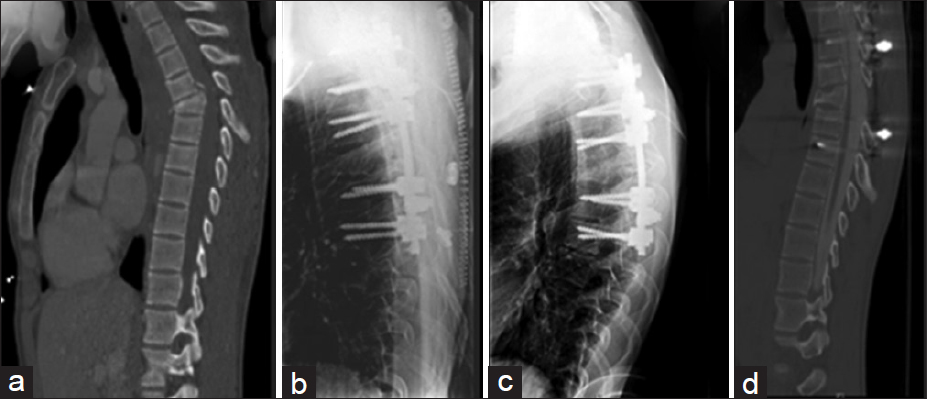
Figure 1
Representative case: 19-year-old male who sustained a motorcycle collision with a thoracic-5 anterior wedge flexion-compression fracture and paraplegia. He underwent T5 laminectomy, with T2–T7 posterior instrumentation under O-arm guidance. (a) Original fracture on mid-sagittal view of CT chest obtained as part of trauma workup. (b) Immediate postoperative lateral T-Spine X-ray showing pedicle screws at T2, 3, 4, 6, and 7. (c) Lateral T-Spine X-ray obtained 6 months postoperatively. (d) Mid-sagittal CT myelogram at 18 months showing continued patency of spinal canal and maintenance of correction of the preoperative kyphotic deformity
DISCUSSION
Traumatic upper thoracic spine fractures are a complex surgical problem often complicated by multiple comorbidities due to the high forces required to cause these types of fractures.[
CONCLUSION
The surgical treatment of traumatic upper thoracic spine fractures T1–T6, although complex, is safe and effective. Reduction and fixation of these fractures results in a reduced risk of further neurological damage, and allows for earlier mobilization, shorter hospital LOS, and improved patient outcomes.
Prior Presentation
Portions of this work were presented as an oral abstract presentation at the Joint Section for Spine and Peripheral Nerves on March 16, 2018 in Orlando, FL, and as an electronic poster abstract at the AANS 2018 annual scientific meeting in New Orleans, LA.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Benzel EC, Ball PA, Baldwin NG, Marchand EP. The sequential hook insertion technique for universal spine instrumentation application. Technical note. J Neurosurg. 1993. 79: 608-11
2. Fisher C, Singh S, Boyd M, Kingwell S, Kwon B, Li MJ. Clinical and radiographic outcomes of pedicle screw fixation for upper thoracic spine (T1-5) fractures: A retrospective cohort study of 27 cases. J Neurosurg Spine. 2009. 10: 207-13
3. Fisher CG, Sahajpal V, Keynan O, Boyd M, Graeb D, Bailey C. Accuracy and safety of pedicle screw fixation in thoracic spine trauma. J Neurosurg Spine. 2006. 5: 520-6
4. Han W, Gao ZL, Wang JC, Li YP, Peng X, Rui J. Pedicle screw placement in the thoracic spine: A comparison study of computer-assisted navigation and conventional techniques?. Orthopedics. 2010. 33:
5. Joaquim AF, Daubs MD, Lawrence BD, Brodke DS, Cendes F, Tedeschi H. Retrospective evaluation of the validity of the thoracolumbar injury classification system in 458 consecutively treated patients. Spine J. 2013. 13: 1760-5
6. Laudato PA, Pierzchala K, Schizas C. Pedicle screw insertion accuracy using o-arm, robotic guidance, or freehand technique: A comparative study. Spine (Phila Pa 1976). 2018. 43: E373-8
7. Magerl F, Aebi M, Gertzbein SD, Harms J, Nazarian S. A comprehensive classification of thoracic and lumbar injuries. Eur Spine J. 1994. 3: 184-201
8. McLain RF, Ferrara L, Kabins M. Pedicle morphometry in the upper thoracic spine: Limits to safe screw placement in older patients. Spine (Phila Pa 1976). 2002. 27: 2467-71
9. Quinlan JF, Harty JA, O'Byrne JM. The need for multidisciplinary management of patients with upper thoracic spine fractures caused by high-velocity impact: A review of 32 surgically stabilised cases. J Orthop Surg (Hong Kong). 2005. 13: 34-9
10. Stankovic M, Milicic A, Savic D, Milankov M, Kecojevic V, Arbutinov V. Comparative radiographic analysis of surgical and conservative treatment of unstable injuries of the thoracic and lumbar spine. Med Pregl. 2001. 54: 315-22
11. Vaccaro AR, Lehman RA, Hurlbert RJ, Anderson PA, Harris M, Hedlund R. A new classification of thoracolumbar injuries: The importance of injury morphology, the integrity of the posterior ligamentous complex, and neurologic status. Spine (Phila Pa 1976). 2005. 30: 2325-33