- Professor of Clinical Neurosurgery, Schoold of Medicine, State University of New York at Stony Brook and Chief of Neurosurgical Spine and Education, NYU Winthrop Hospital, NYU Winthrop NeuroScience, NY, USA
Correspondence Address:
Nancy E. Epstein
Professor of Clinical Neurosurgery, Schoold of Medicine, State University of New York at Stony Brook and Chief of Neurosurgical Spine and Education, NYU Winthrop Hospital, NYU Winthrop NeuroScience, NY, USA
DOI:10.4103/sni.sni_302_17
Copyright: © 2017 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Nancy E. Epstein. Tisseel's impact on hemostasis for 2–3 and 4–6-level lumbar laminectomies. 11-Dec-2017;8:299
How to cite this URL: Nancy E. Epstein. Tisseel's impact on hemostasis for 2–3 and 4–6-level lumbar laminectomies. 11-Dec-2017;8:299. Available from: http://surgicalneurologyint.com/surgicalint-articles/tisseels-impact-on-hemostasis-for-2-3-and-4-6%e2%80%91level-lumbar-laminectomies/
Abstract
Background:Tisseel (Baxter International Inc., Westlake Village, CA, USA), a fibrin sealant, was originally devised to strengthen repairs of spinal cerebrospinal fluid (CSF) fistulas. Here, we evaluated how Tisseel correlated with hemostasis (e.g., defined as reduced postoperative drainage, time to drain removal, length of stay (LOS), and postoperative transfusion requirements) in 58 patients undergoing 2–3 vs. 79 patients having 4–6 level lumbar laminectomies.
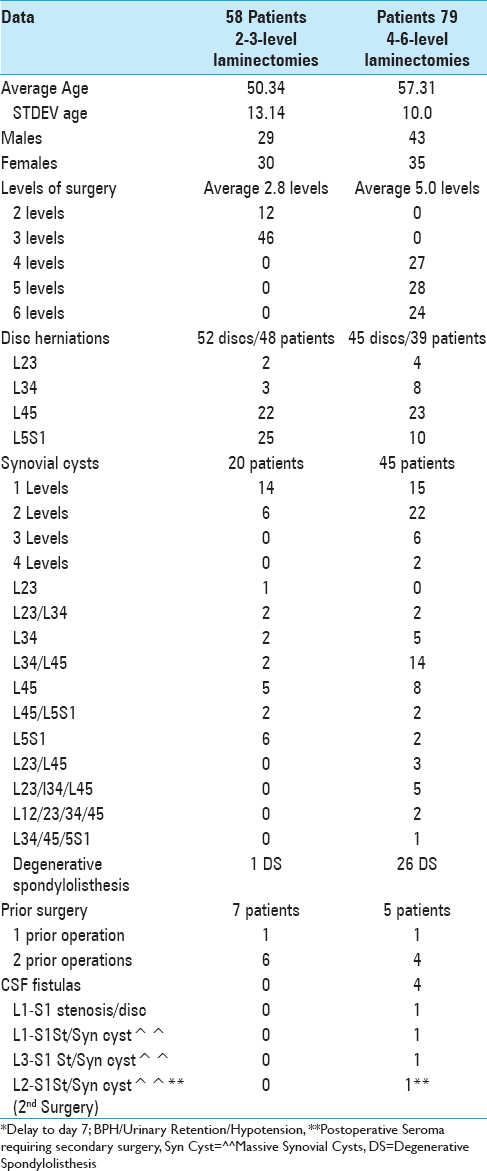
Methods:We assessed how Tisseel correlated with hemostasis in 58 patients undergoing 2–3 level laminectomies/stenosis (with 48 herniated discs and 20 synovial cysts, 1 degenerative spondylolisthesis) vs. 79 having 4–6 level laminectomies/stenosis (with 39 lumbar discs, 45 synovial cysts, and 26 degenerative spondylolisthesis).
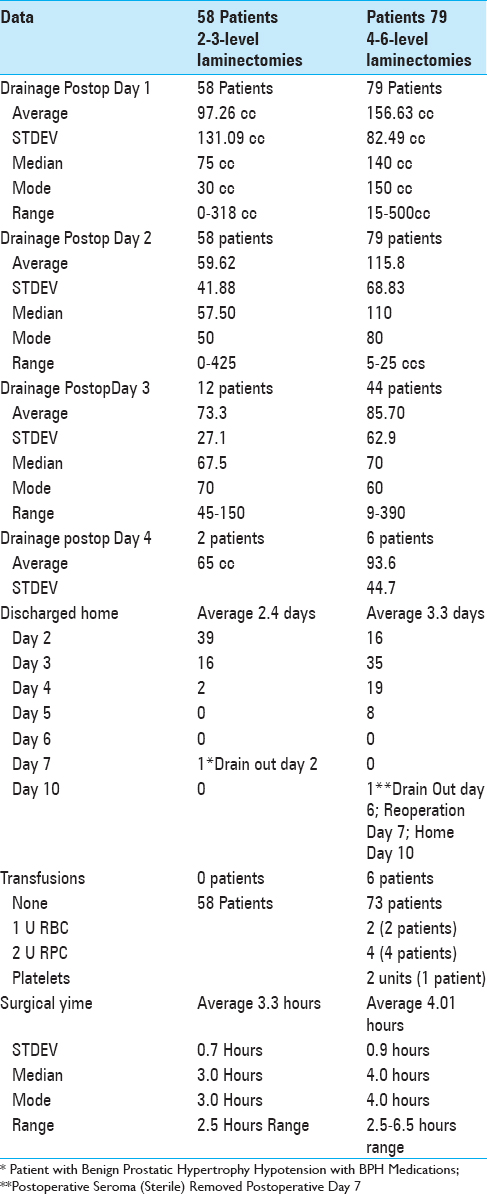
Results:Following 2–3 level laminectomies, the average drainage on postoperative day 1 was 87.26 cc, and on day 2 was 59.62 cc; most drains were removed and the majority of patients were discharged on postoperative day 2, requiring no transfusions. After 4–6 level decompressions, greater postoperative drainage was observed on postoperative days 1 (e.g., 156.63 cc), and 2 (115.8 cc), and many were continued for 3 (85.7 cc; 44 patients), and 4 postoperative days (93.6: 6 patients) respectively. Drains were typically removed and patients were discharged on postoperative days 3 and 4, with just 6 requiring transfusions. Notably, there were four CSF fistulas for patients undergoing 4–6 level laminectomies; one had a large disc hernation in conjunction with postoperative scare, while three had massive calcified synovial cysts extending to/through the dura.
Conclusions:Utilizing Tisseel as a hemostatic allowed us to quantitate hemostasis (the average postoperative drainage, time to drain removal, LOS, and postoperative transfusion requirements) for those undergoing 2–3 level laminectomies vs. 4–6 level procedures with large subsets also exhibiting herniated discs, synovial cysts, and degenerative spondylolisthesis.
Keywords: Discs, hemostatic agent, lumbar spine surgery, no CSF fistulas, no fusions length of stay, stenosis, timing drain removal, Tisseel, transfusion requirements
INTRODUCTION
Tisseel (Baxter International Inc., Westlake Village, CA, USA), a fibrin sealant, was originally introduced to strengthen repairs of cerebrospinal fluid (CSF) fistulas (traumatic and deliberate) resulting from lumbar spinal surgery. However, Tisseel is increasingly being utilized in the absence of CSF fistulas as a hemostatic agent for lumbar surgery, with hemostasis variably defined by the amount of postoperative drainage, time to drain removal, length of stay (LOS), and postoperative transfusion requirements. Here we quantitated the comparative impact of Tisseel on hemostasis for patients undergoing 2–3 vs. 4–6 level laminectomies for stenosis with varying frequencies (e.g. with/without) of disc herniations, synovial cysts, and degenerative spondylolisthesis (DS).
MATERIALS AND METHODS
We prospectively followed 58 patients undergoing 2–3 level laminectomies/stenosis and 79 patients undergoing 3–6 level laminectomies/stenosis [
RESULTS
Postoperative drainage/other variables
Data for 2–3-level laminectomies
The average operative times for 58 patients undergoing 2–3 level laminectomies was 3.3 h (STDEV 0.7 h) [
Data for 4–6-level laminectomies
For the 79 patients undergoing 4–6 level laminectomies, the average operative time was 4.0 h (STDEV 0.9) [
Postoperative cerebrospinal fluid fistulas
Correlation of intraoperative cerebrospinal fluid fistulas with massive calcified synovial cysts extending to/through the dura
Intraoperative traumatic CSF fistulas were only observed in four patients undergoing the more extensive 4–6 level laminectomies/stenosis. This correlated with a higher incidence (45 cases) of one to four level synovial cysts observed amongst these 79 patients. One patient developed a CSF fistula following an L1-S1 laminectomy for stenosis and excision of a massive disc herniation; this patient had prior surgery, and the fistula was largely attributed to postoperative scar. The other three CSF fistulas occurred during decompressions/removals of massive calcified synovial cysts extending to/through the dura resected during L1-S1, L2-S1, and L3-S1 laminectomies. Notably, although 20 of 58 patients undergoing 2-3 level laminectomies had single/multiple synovial cysts, none developed intraoperative CSF fistulas [
Lack of correlation of cerebrospinal fluid fistulas with postoperative scar
Prior surgery did not appear to uniquely contribute to CSF fistulas during secondary or tertiary surgery for patients undergoing 2–3 or 4–6 level laminectomies/stenosis [
Transfusion requirements: 6 (7.6%) of 79 patients undergoing4-6-level laminectomies
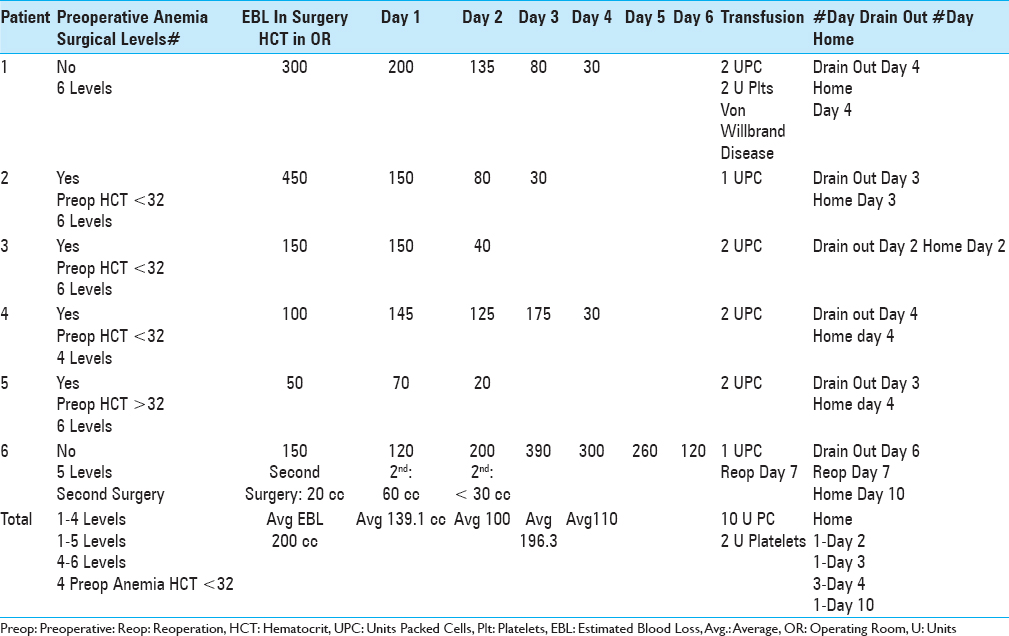
Utilizing Tisseel to facilitate hemostasis, no transfusions were required for the 58 patients undergoing 2–3 level laminectomies, while 6 (7.6%) of the 79 undergoing 4–6 level laminectomies required transfusions [Tables
DISCUSSION
Fibrocaps (dry powder fibrin sealant) contributes to spinal hemostasis
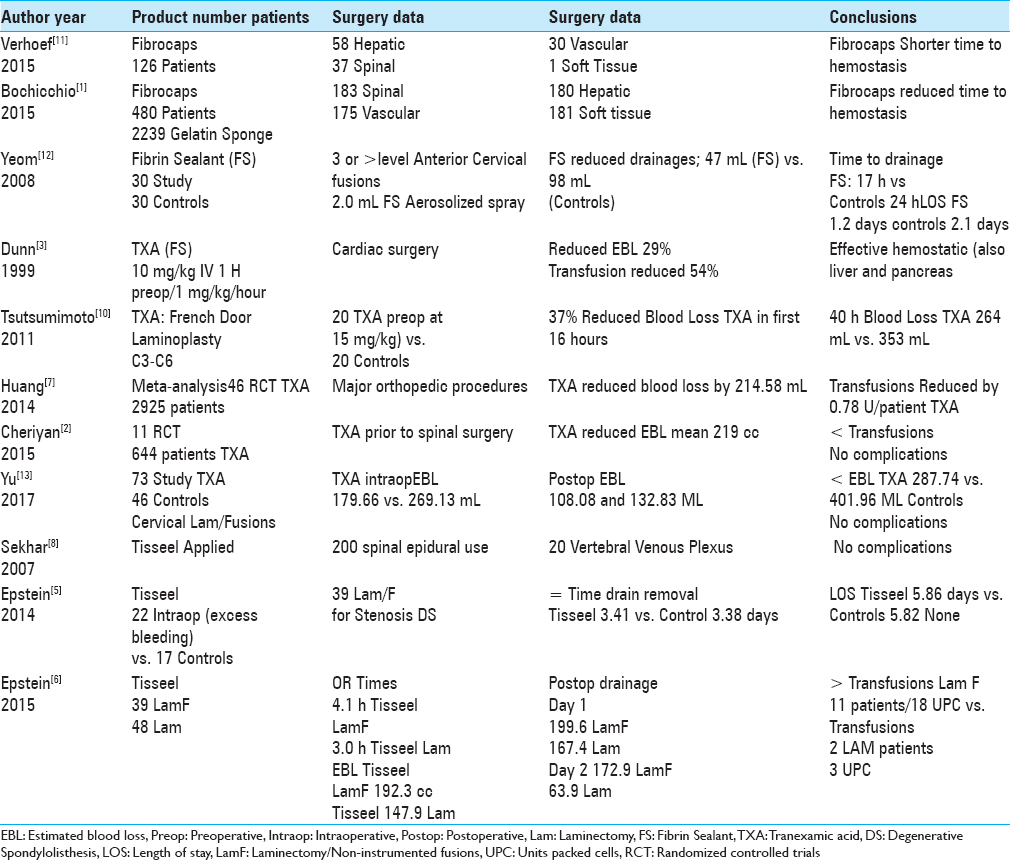
Two studies in 2015 utilized Fibrocaps for epidural spinal hemostasis and other surgical procedures [
Role of fibrin sealants/fibrin glues in spinal and orthopedic surgery
Multiple studies have documented the efficacy of fibrin sealant/fibrin glue (FS/FG) in promoting postoperative spinal hemostasis [
Tranexamic acid (fibrin Sealant) reduces blood loss in spine surgery
Several studies demonstrated how preoperative intravenous administration of tranexamic acid (TXA) (FS) contributed to hemostasis and reduced postoperative blood loss following spinal surgery [
Tranexamic acid reduces blood loss in cardiac and orthopedic surgery
In 1999, Dunn and Goa defined TXA as a “synthetic derivative of the amino acid lysine that exerts its antifibrinolytic effect through the reversible blockade of lysine binding sites on plasminogen molecules [
Tranexamic acid reduces blood loss in cervical spine surgery
In 2011, Tsutsumimoto et al. performed 40 consecutive French door cervical laminoplasties (C3-C6); 20 patients received intravenous TXA prior to the incision (15 mg/kg body weight) vs. 20 receiving placebo [
Tranexamic acid reduces blood loss in spinal surgery overall
In 2014, when Zhang et al. performed a meta-analysis of six RCTs (randomized controlled trials) involving spinal surgery (RCTs; 411 patients), TXA-treated patients exhibited significantly reduced blood loss and transfusion requirements vs. placebo patients, without increasing thromboembolic complications [
Tisseel's (fibrin sealant) role in spinal hemostasis
Several studies have unsuccessfully utilized Tisseel FS/FG to promote epidural spinal hemostasis [
CONCLUSIONS
One may utilize the data presented in this study to advise patients undergoing 2-3 and 4-6 level laminectomies regarding their anticipated time to drain removal, LOS, risk of a CSF fistula, and potential requirement for a transfusion. Here, the addition of Tisseel to facilitate hemostasis for 79 patients undergoing the more extensive 4-6 level laminectomies (without non instrumented fusions), correlated with greater postoperative drainage, time to drain removal, LOS, and postoperative transfusion requirements. Alternatively, for those undergoing more restricted 2-3 level laminectomies (without non instrumented fusions), all parameters were appropriately/respetively decreased. Notably, utilizing Tisseel, there were no neurological complications, no infections, and no readmissions for patients in either operative group.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Bochicchio GV, Gupta N, Porte RJ, Renkens KL, Pattyn P, Topal B. The FINISH-3 trial: A phase 3, international, randomized, single-blind, controlled trial of topical fibrocaps in intraoperative surgical hemostasis. J Am Coll Surg. 2015. 220: 70-81
2. Cheriyan T, Maier SP 2nd, Bianco K, Slobodyanyuk K, Rattenni RN, Lafage V. Efficacy of tranexamic acid on surgical bleeding in spine surgery: A meta-analysis. Spine J. 2015. 15: 752-61
3. Dunn CJ, Goa KL. Tranexamic acid: A review of its use in surgery and other indications. Drugs. 1999. 57: 1005-32
4. Epstein NE. Hemostasis and other benefits of fibrin sealants/glues in spine surgery beyond cerebrospinal fluid leak repairs. Surg Neurol Int. 2014. 5: S304-14
5. Epstein NE. Tisseel utilized as hemostatic in spine surgery impacts time to drain removal and length of stay. Surg Neurol Int. 2014. 5: S354-61
6. Epstein NE. Tisseel does not reduce postoperative drainage, length of stay, and transfusion requirements for lumbar laminectomy with noninstrumented fusion versus laminectomy alone. Surg Neurol Int. 2015. 6: S172-6
7. Huang F, Wu D, Ma G, Yin Z, Wang Q. The use of tranexamic acid to reduce blood loss and transfusion in major orthopedic surgery: A meta-analysis. J Surg Res. 2014. 186: 318-27
8. Sekhar LN, Natarajan SK, Manning T, Bhagawati D. The use of fibrin glue to stop venous bleeding in the epidural space, vertebral venous plexus, and anterior cavernous sinus: Technical note. Neurosurgery. 2007. 61: E51-
9. Thoms RJ, Marwin SE. The role of fibrin sealants in orthopaedic surgery. J Am Acad Orthop Surg. 2009. 17: 727-36
10. Tsutsumimoto T, Shimogata M, Ohta H, Yui M, Yoda I, Misawa H. Tranexamic acid reduces perioperative blood loss in cervical laminoplasty: A prospective randomized study. Spine (Phila Pa 1976). 2011. 36: 1913-8
11. Verhoef C, Singla N, Moneta G, Muir W, Rijken A, Lockstadt H. Fibrocaps for surgical hemostasis: Two randomized, controlled phase II trials. J Surg Res. 2015. 194: 679-87
12. Yeom JS, Buchowski JM, Shen HX, Liu G, Bunmaprasert T, Riew KD. Effect of fibrin sealant on drain output and duration of hospitalization after multilevel anterior cervical fusion: A retrospective matched pair analysis. Spine (Phila Pa 1976). 2008. 33: E543-7
13. Yu CC, Gao WJ, Yang JS, Gu H, Md MZ, Sun K, Hao DJ. Can tranexamic acid reduce blood loss in cervical laminectomy with lateral mass screw fixation and bone grafting: A retrospective observational study. Medicine (Baltimore). 2017. 96: e6043-
14. Zhang F, Wang K, Li FN, Huang X, Li Q, Chen Z. Effectiveness of tranexamic acid in reducing blood loss in spinal surgery: A meta-analysis. BMC Musculoskelet Disord. 2014. 15: 448-