- Department of Neurosurgery, Graduate School of Medical Science, Kanazawa University, Kanazawa, Ishikawa, Japan
Correspondence Address:
Mitsutoshi Nakada
Department of Neurosurgery, Graduate School of Medical Science, Kanazawa University, Kanazawa, Ishikawa, Japan
DOI:10.4103/2152-7806.196931
Copyright: © 2016 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Takehiro Uno, Masashi Kinoshita, Takuya Furuta, Katsuyoshi Miyashita, Hemragul Sabit, Mitsutoshi Nakada. Volumetric growth analysis of an insular dysembryoplastic neuroepithelial tumor over a 10-year follow-up. 28-Dec-2016;7:
How to cite this URL: Takehiro Uno, Masashi Kinoshita, Takuya Furuta, Katsuyoshi Miyashita, Hemragul Sabit, Mitsutoshi Nakada. Volumetric growth analysis of an insular dysembryoplastic neuroepithelial tumor over a 10-year follow-up. 28-Dec-2016;7:. Available from: http://surgicalneurologyint.com/surgicalint_articles/volumetric-growth-analysis-of-an-insular-dysembryoplastic-neuroepithelial-tumor-over-a-10%e2%80%91year-follow%e2%80%91up/
Abstract
Background:Dysembryoplastic neuroepithelial tumors (DNETs) are benign tumors characterized by a cortical location; they result in symptoms of drug-resistant partial seizures in children. The development of DNETs is poorly understood because most of them are resected immediately upon diagnosis without any observation period owing to the intractable seizures.
Case Description:We report the first DNET case with the growth rate analyzed in the natural course of development for a period of 10 years. The patient was a right-handed man who was initially referred to another hospital with mild head injury when he was 8 years old. A tumor located in the right insular cortex was incidentally detected on magnetic resonance imaging (MRI) and followed-up with annual MRI for 10 years.
Conclusion:In this case, the volume of the DNET increased in direct proportion to the length of time in its clinical course. The tumor doubling time was approximately 10 years. This case suggests DNET is a slow-growing but not stable tumor.
Keywords: Dysembryoplastic neuroepithelial tumor, insular, growth analysis
INTRODUCTION
Dysembryoplastic neuroepithelial tumors (DNETs) are benign, hamartomatous tumors thought to arise from the cortical gray matter. They are mixed neuronal-glial tumors, classified as grade I by the World Health Organization (WHO). Progression or post-surgical recurrence of DNETs is perceived to be extremely rare. DNETs typically cause intractable seizures in children, and are removed surgically without observation.[
CASE REPORT
Our patient was initially referred to another hospital with mild head injury when he was 8 years old. An intra-axial tumor located in the right insular cortex was incidentally detected on MRI. Surgical resection was waived and followed-up for 10 years until the patient was 18 years old because of slowly growing tumor without symptoms. At the end of the observation period, the tumor size was measured to be one and a half times the diameter measured on the first MRI scan. After the end of the follow-up period, he visited our hospital for intensive examination and treatment.
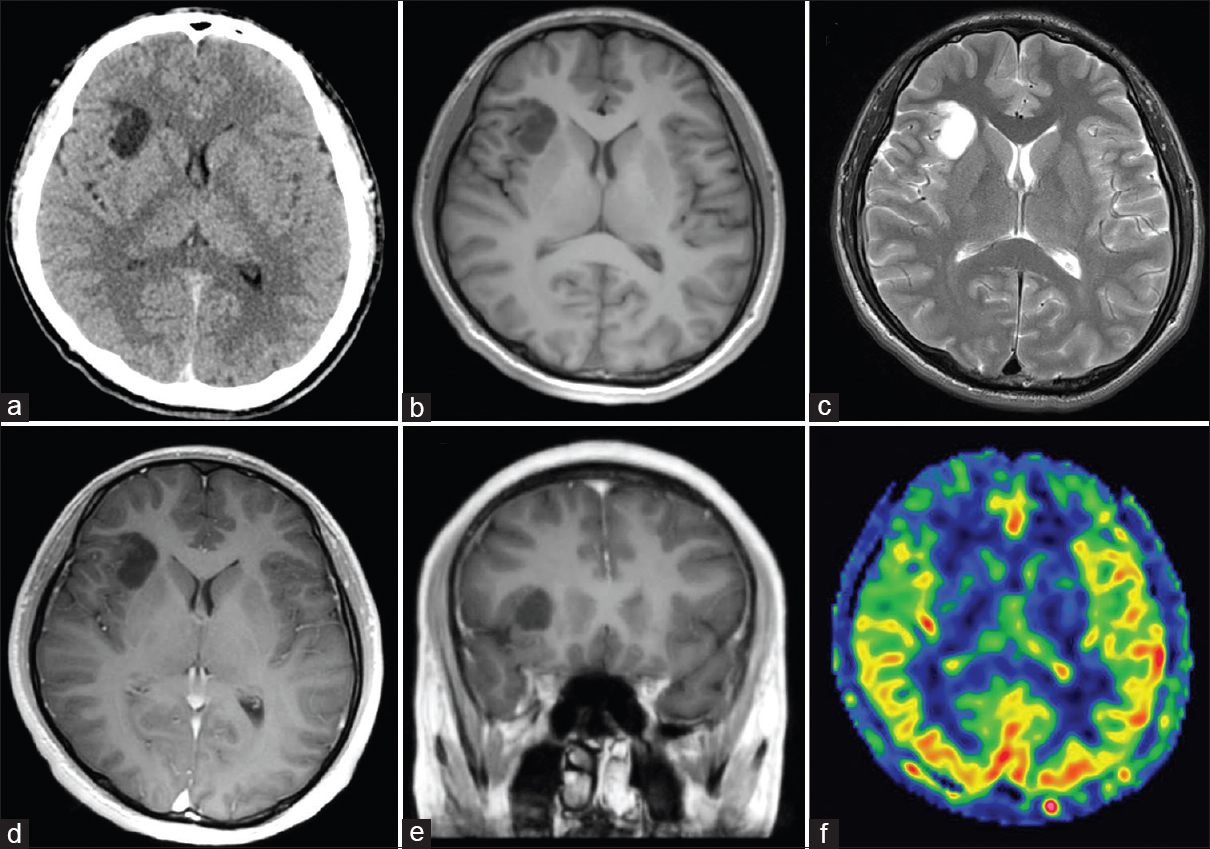
The patient had no neurological deficit. Computed tomography (CT) imaging showed a low-density lesion with no calcification located in the right insular cortex [
Figure 1
Axial computed tomography (CT) (a) and magnetic resonance imaging (MRI) scans (b-d, f) and coronal MRI scan (e). (a) CT scan shows a low-density lesion in right insular cortex with no calcification. (b) T1-weighted MRI demonstrates a hypointense lesion. (c) T2-weighted MRI shows a hyperintense lesion that corresponds with the hypointensity on the T1-weighted MRI image. (d, e) T1-weighted MRI with gadolinium administration did not demonstrate any enhanced lesion. (f) Arterial spin labeling study shows decreased blood flow at the lesion
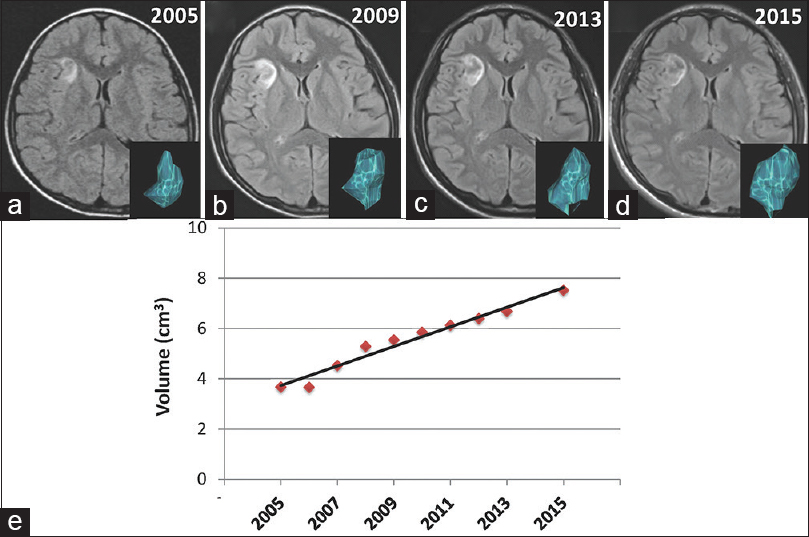
The lesion located in the right insular cortex demonstrated gradual growth for 10 years [Figure
Figure 2
(a-d) T2-FLAIR MRI scans in the axial plane from age 8 to 18 years show gradual growth. An inserted picture in the corner of images is a three-dimensional reconstructed model of the tumor in each figure. (e) A dot graph with an almost straight line shows that the increase in tumor volume is directly proportional to time
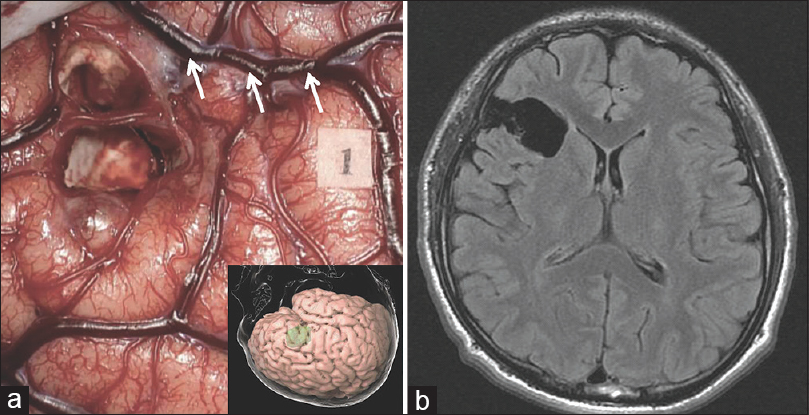
In order to remove the lesion and obtain histopathological diagnosis, an awake craniotomy was performed using cortical and subcortical stimulation mapping with a bipolar direct electrical stimulator at 3.5 mA/60 Hz biphasic current to monitor motor and somatosensory response, speech or language difficulties, and other higher brain functions.[
Figure 3
Intraoperative (a) and postoperative (b) images. (a) Intraoperative view after tumor resection during awake surgery. Tag 1 suggests the area where anarthria was induced on the ventral precentral gyrus by direct electrical stimulation. An inserted picture is the three-dimensional cortical view. (b) Fluid-attenuated inversion recovery magnetic resonance imaging performed 3 months after the operation shows gross total resection. Arrows indicate the Sylvian fissure
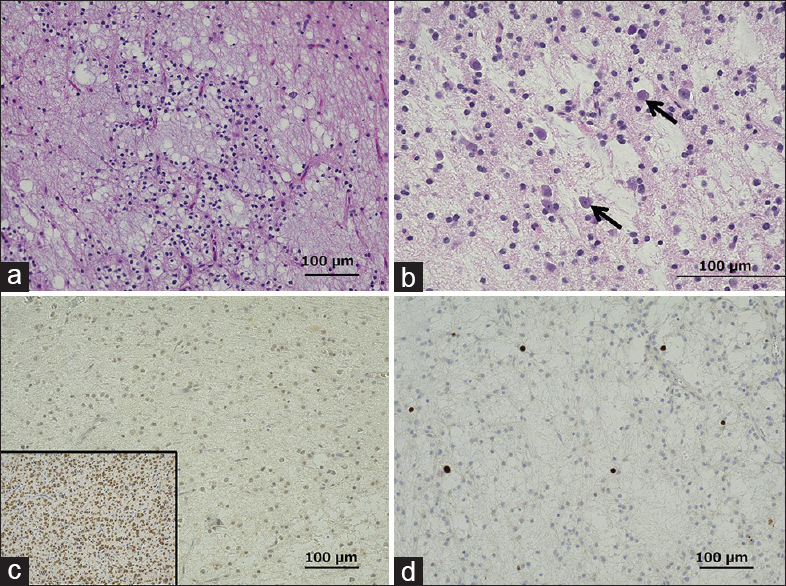
Histological examination of the hypointense area on T1-weighted MRI showed multiple cystic structures with myxomatous background and proliferation of oligodendroglia-like cells with oval nuclei in the wall of the cystic spaces [
Figure 4
Photomicrographs of the surgical specimen showing the hypointense lesion on T1-weighted magnetic resonance imaging. (a) Multiple cystic structures are observed on a mucinous background. Hematoxylin and eosin (H and E) staining, original magnification ×100. (b) High-power view of a, showing floating neurons (arrows) scattered in the cystic structure. H and E staining, ×200. (c) Negative staining for IDH1 mutation, ×100. Inset: Positive control (d) The Ki-67 staining index was 1%, ×100
DISCUSSION
This rare case of DNET was followed for 10 years with annual MRI. Our statistical analysis of growth rate showed that the tumor volume gradually increased in direct proportion to time. According to this analysis, the tumor doubling time was determined to be approximately 3473 days. To date, only a few studies on DNET growth pattern have been reported.[
DNETs are usually stable tumors. However, malignant transformation in DNETs results in rapid growth rates.[
The lesion was located in the right insular cortex in our patient. DNETs typically occur in the temporal lobe in 62%, the frontal lobe in 31%, the parietal and/or occipital lobe in 7% of cases,[
DNET is generally positive for Olig2, S-100, and synaptophysin. The genetic background of DNETs has not been systemically investigated. Loss of heterozygosity at 1p/19q and TP53[
The patient underwent an awake craniotomy. Gross total resection of the lesion was achieved. We expected an extremely low risk of tumor recurrence.[
Despite being recognized for only less than three decades, DNETs are becoming an important part of epilepsy neurosurgical practice. However, their natural growth rate remains poorly understood. We present this case to increase the knowledge related to the growth pattern of these tumors, and to suggest that the growth rate of DNETs is directly proportional to time.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Acknowledgments
We thank Erika Komura for assistance with preparation of the immunohistochemistry.
References
1. Alexander H, Tannenburg A, Walker DG, Coyne T. Progressive dysembryoplastic neuroepithelial tumour. J Clin Neurosci. 2015. 22: 221-4
2. Capper D, Reuss D, Schittenhelm J, Hartmann C, Bremer J, Sahm F. Mutation-specific IDH1 antibody differentiates oligodendrogliomas and oligoastrocytomas from other brain tumors with oligodendroglioma-like morphology. Acta Neuropathol. 2011. 121: 241-52
3. Daghistani R, Miller E, Kulkarni AV, Widjaja E. Atypical characteristics and behavior of dysembryoplastic neuroepithelial tumors. Neuroradiology. 2013. 55: 217-24
4. Daumas-Duport C, Scheithauer BW, Chodkiewicz JP, Laws ER, Vedrenne C. Dysembryoplastic neuroepithelial tumor: A surgically curable tumor of young patients with intractable partial seizures. Report of thirty-nine cases. Neurosurgery. 1988. 23: 545-56
5. Fujisawa H, Marukawa K, Hasegawa M, Tohma Y, Hayashi Y, Uchiyama N. Genetic differences between neurocytoma and dysembryoplastic neuroepithelial tumor and oligodendroglial tumors. J Neurosurg. 2002. 97: 1350-5
6. Herbet G, Lafargue G, Almairac F, Moritz-Gasser S, Bonnetblanc F, Duffau H. Disrupting the right pars opercularis with electrical stimulation frees the song: Case report. J Neurosurg. 2015. 123: 1401-4
7. Jensen RL, Caamano E, Jensen EM, Couldwell WT. Development of contrast enhancement after long-term observation of a dysembryoplastic neuroepithelial tumor. J Neurooncol. 2006. 78: 59-62
8. Leung SY, Gwi E, Ng HK, Fung CF, Yam KY. Dysembryoplastic neuroepithelial tumor. A tumor with small neuronal cells resembling oligodendroglioma. Am J Surg Pathol. 1994. 18: 604-14
9. Mano Y, Kumabe T, Shibahara I, Saito R, Sonoda Y, Watanabe M. Dynamic changes in magnetic resonance imaging appearance of dysembryoplastic neuroepithelial tumor with or without malignant transformation. J Neurosurg Pediatr. 2013. 11: 518-25
10. Moazzam AA, Wagle N, Shiroishi MS. Malignant transformation of DNETs: A case report and literature review. Neuroreport. 2014. 25: 894-9
11. Thom M, Toma A, An S, Martinian L, Hadjivassiliou G, Ratilal B. One hundred and one dysembryoplastic neuroepithelial tumors: An adult epilepsy series with immunohistochemical, molecular genetic, and clinical correlations and a review of the literature. J Neuropathol Exp Neurol. 2011. 70: 859-78
12. Yang AI, Khawaja AM, Ballester-Fuentes L, Pack SD, Abdullaev Z, Patronas NJ. Multifocal dysembryoplastic neuroepithelial tumours associated with refractory epilepsy. Epileptic Disord. 2014. 16: 328-32