- Department of Neurosurgery, National Institute of Mental Health and Neurosciences (NIMHANS), Bengaluru, Karnataka, India
Correspondence Address:
Manish Beniwal
Department of Neurosurgery, National Institute of Mental Health and Neurosciences (NIMHANS), Bengaluru, Karnataka, India
DOI:10.4103/sni.sni_445_17
Copyright: © 2018 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Harsh Deora, Manish Beniwal, Shilpa Rao, K. V. L. N. Rao, V. Vikas, Sampath Somanna. Wolf in Sheep's clothing: Intracranial actinomycosis masquerading as en-plaque meningioma. 14-Feb-2018;9:39
How to cite this URL: Harsh Deora, Manish Beniwal, Shilpa Rao, K. V. L. N. Rao, V. Vikas, Sampath Somanna. Wolf in Sheep's clothing: Intracranial actinomycosis masquerading as en-plaque meningioma. 14-Feb-2018;9:39. Available from: http://surgicalneurologyint.com/surgicalint-articles/wolf-in-sheeps-clothing-intracranial-actinomycosis-masquerading-as-en%e2%80%91plaque-meningioma/
Abstract
Background:Actinomycosis israeli is a gram-positive non-acid-fast bacterium and is the most common causative agent of actinomycosis. It usually presents as a brain abscess via a secondary spread from the cervicofacial region or lungs. However, presentation as an en-plaque lesion with bone involvement out of proportion to brain parenchyma is rare and needs to be acknowledged.
Case Description:We report a case of intracranial en-plaque lesion in a 47-year-old female with restriction of mouth opening and right third nerve palsy for 1 year. Imaging showed hyperostosis of the maxilla, temporal base, sphenoid, and zygoma with dural thickening suggestive of a meningioma. Surgical decompression yielded a granulomatous lesion along with empirical antitubercular therapy. However, patient succumbed to the infection and final histopathology revealed actinomycosis infection.
Conclusion:Adequate knowledge along with clinical suspicion are the pillars to treating this unfortunate disease. Cheek swelling with bone involvement should be actively sought as risk factors. This case presents a unique opportunity to identify the fox among the sheep by elucidating the characteristic imaging findings and intraoperative view of this rare entity.
Keywords: Abscess, actinomycosis, cranial infection, en-plaque, pachymeningitis
INTRODUCTION
“En-plaque” was a term coined by Cushing and Eisenhardt to describe a particular growing pattern in meningiomas consisting of a diffuse sheet-like appearance closely following the inner table of the skull.[
Actinomycosis is a rare, chronic, and slowly progressing granulomatous disease caused by filamentous Gram-positive bacteria from the genus Actinomyces. The bacteria spread from the oral cavity, lung, or abdominal cavity and usually present as localized infection in immunocompetent hosts or as a disseminated disease in immunocompromised individuals.[
CASE REPORT
A 47-year-old female presented to our outpatient department with complaints of drooping of the right eyelid and painless swelling of the right cheek for 1 year. She also complained of difficulty in mouth opening for the past 6 months. The swelling had been progressively increasing and showed no postural variation or pulsatility. The mouth opening was restricted to less than 1 finger, and the patient could drink only liquids using a straw. At the time of presentation, she did not give any history of fever or past medical history of diabetes, high-risk behavior, heart problems, or any other conditions that would predispose her to an intracranial infection.
On examination, the swelling was nontender, hard diffuse without any clear margins or defined edges. It was arising from the maxilla and was not independently mobile off the same. The right eye proptosis was axial and nonpulsatile with no extraocular movement restriction. The vision was 6/24 in both eyes on Snellen's chart. Rinne's test showed that the right ear had no air or bone conduction while on the left side air conduction was more than bone conduction. Weber test was lateralized to the left side. Rest of the examination was unremarkable. Laboratory investigations were normal.
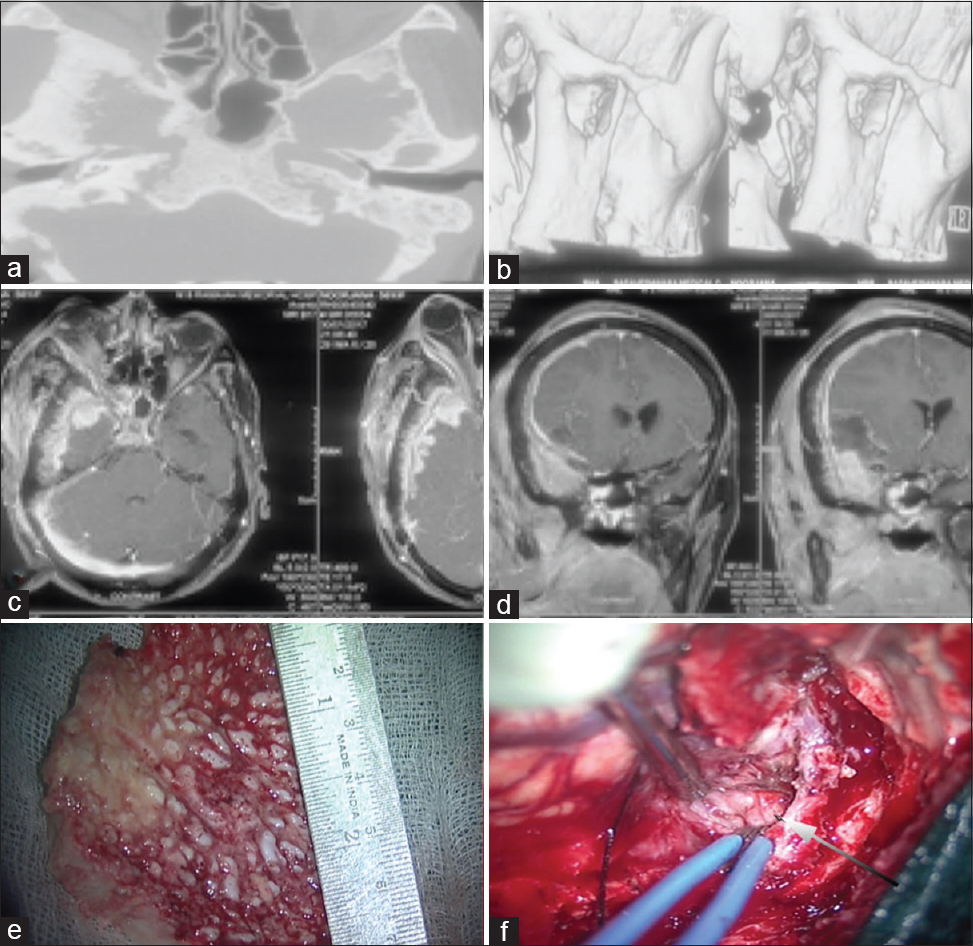
After clinical evaluation, a large lesion involving the maxilla, orbit, and temporal base with extension to the middle ear was suspected of the possibility of slow-growing neoplasm being a possibility. She underwent a computed tomography (CT) scan, which showed diffuse hyperostosis of the right frontal, squamous temporal, sphenoid and zygomatic bones, and an en-plaque ground glass lesion, involving the basitemporal bone [Figure
Figure 1
(a and b) Cranial CT scan with bony reconstruction showing diffuse hyperostosis of the temporal bone, zygomatic bone with condylar involvement. (c and d) MRI T1 weighted images with gadolinium contrast enhancement axial and coronal sections showing diffuse enhancement in the temporal and infratemporal region. (e) Intraoperative resected specimen of the inner surface temporal bone showing hyperostosis and irregular surface, (f) Intraoperative image showing intradural granulomatous lesion as marked by the arrow
Diagnosis of an en-plaque meningioma was suspected and she underwent a right frontotemporal craniotomy and complete excision of the intracranial lesion and partial decompression of involved areas of bone [
Due to the endemic nature of tuberculosis in India and squash report of pathology, diagnosis of tubercular granulomatous inflammation was made and the patient was started on antitubercular drug therapy and discharged on antitubercular therapy with a hospital stay of 5 days. She was also put on antiepileptic drugs (Phenytoin) for prevention of postoperative seizures and steroids to counteract tubercular arteritis. Human immunodeficiency virus (HIV) was negative on repeated testing from two different centers. However, she reported back to the emergency 3 days after discharge in an unconscious state. According to the relatives, patient was complaining of mild headache since the time of discharge, and on the 3rd day after discharge, in the morning patient was found unconscious in her bed. At the time of presentation, patient vitals were not recordable, her GCS was 3/15, with bilateral pupils nonreactive and fixed. She was immediately given cardiopulmonary resuscitation, but could not be revived despite aggressive resuscitative measures. The exact cause of death could not be ascertained, but could be flare up of the primary disease as later seen in brain autopsy.
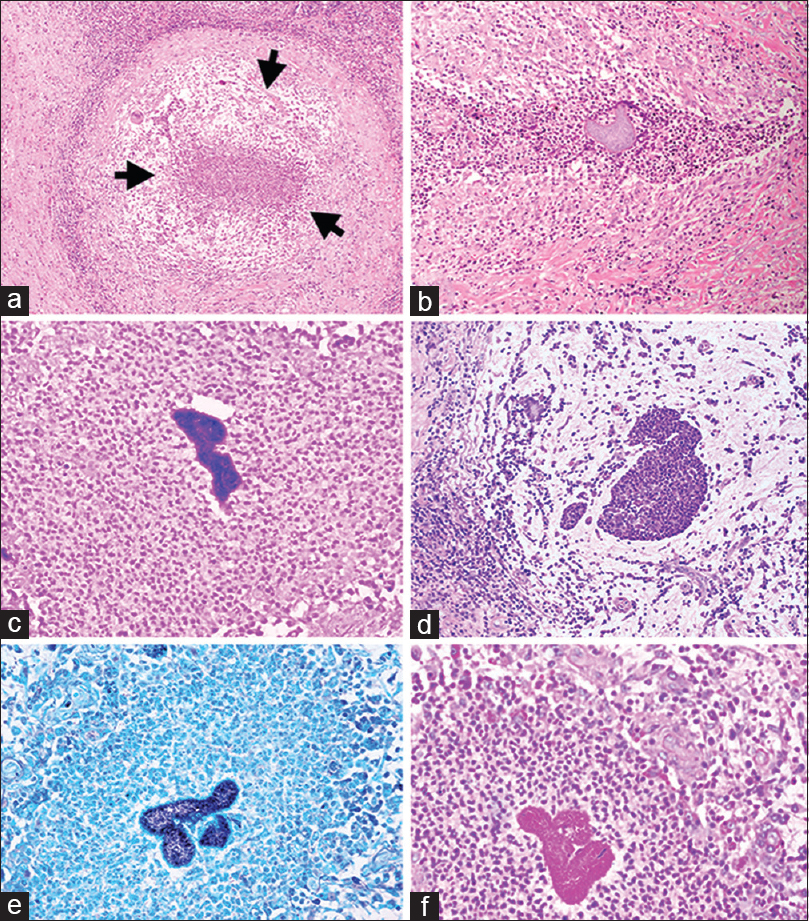
Subsequent histopathological analysis revealed a suppurative granulomatous inflammatory pathology with dense fibrosis of the stroma. The thickened fibrotic dura showed multiple discrete and confluent suppurative microabscess rimmed by histiocytes and granulomatous inflammatory cells along with multinucleate giant cells (foreign body type). Many of the microabscesses showed colonies and collections of slender filamentous bacteria with evidence of neutrophilic reaction. Rest of the stroma showed dense fibrosis with scattered lymphoplasmacytic infiltrate and reactive blood vessels. The adjacent brain parenchyma included showed cerebritis. The colonies were highlighted on Grams stain and Gomori methenamine silver stain. Staining for acid-fast bacilli was negative for tubercular and Nocardia species. These findings on histopathological examination resulted in the final diagnosis of a chronic necrotizing granulomatous inflammation with Actinomyces-like organisms [
Figure 2
Shows a large well-formed granuloma within the brain parenchyma walled off by dense lymphomononuclear cells (a), and presence of neutrophilic abscesses surrounding an actinomycotic colony (b). These colonies are highlighted on Gram's (c and d), GMS (Gomori Methenamine-Silver) (e) and PAS (Periodic acid–Schiff) (f) stains
Partial autopsy limited to the removal of brain revealed presence of bacterial colonies with a suppurative granulomatous inflammation, as seen in the biopsied tissue. There were no other lesions on the external examination at autopsy.
DISCUSSION
Actinomycotic infection can have varied involvement ranging from orocervical, cardiothoracic, skeletal, soft tissue, abdomen, and pelvis or cranial.[
The infection is commonly caused by Actinomyces israeli, which is a Gram-positive, nonacid-fast branching filamentous bacteria with anaerobic or microaerophilic requirements. Actinomycosis is a nonsuppurative infection with typically a long delay between infection and presentation.[
Imaging generally shows a singular cerebral abscess which may be uni or multilocular and is usually encapsulated with the predilection for frontal and temporal lobes.[
There is no linear method of management that has proven to be superior to others.[
On repeated asking for history of any procedure, patient's relatives gave a history of tooth extraction 3 years back on the right side, which may have been the source of infection. There was no history of taking any immunosuppressant drugs. Xia et al. reported in 2003 that endodontic infections with Actinomyces are much higher in frequency than previously thought. Actinomyces destroys local tissue and replaces it with a chronic inflammatory tissue.[
In this case we missed several key indicators, which can be used as learning points for future such cases. We did not pay heed to her clinical complaint of restricted mouth opening and radiological imaging, which showed diffuse involvement which is uncommon in meningioma. Past history of tooth extraction was also noted postmortem. Thus, improper history can lead to disastrous consequences.
For a pathologist also, presence of a suppurative granulomatous inflammation should have alerted them to meticulously search for actinomycotic colonies, apart from tuberculosis. Young neurosurgeons should especially be aware of this clinical differential. The severity of infection and misdiagnosis led to the death of our patient. Mortality and morbidity remain high and figures of 28% and 54% have been cited.[
CONCLUSION
Several insights are gained from our unfortunate case. First, although a chronic indolent infection, Actinomycosis can be fulminant and fatal. Misdiagnosis is the norm unless risk factors are actively sought. Prolonged and high-dose antibiotics along with aggressive debridement are currently the best management options. Presentation may be in the form of a chronic granulomatous inflammation involving the cervical, maxillofacial region, especially in an immunocompetent patient without risk factors. On histopathology, the presence of a suppurative granulomatous inflammation should alert one to meticulously search for infective colonies, instead of always thinking of tuberculosis.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form the patient(s) has/have given his/her/their consent for his/her/their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Akhaddar A, Elouennass M, Baallal H, Boucetta M. Focal intracranial infections due to Actinomyces species in immunocompetent patients: Diagnostic and therapeutic challenges. World Neurosurg. 2010. 74: 346-50
2. Bonnefond S, Catroux M, Melenotte C, Karkowski L, Rolland L, Trouillier S. Clinical features of actinomycosis: A retrospective, multicenter study of 28 cases of miscellaneous presentations. Medicine. 2016. 95: e3923-
3. Chopra S, Christie M, Felimban H. Intracranial actinomycosis mimicking meningioma. Ann Saudi Med. 1995. 15: 515-8
4. Cushing H EL.editorsThe Meningiomas: Their Classification, Regional Behavior, Life History, and Surgical End Results. Springfield: Charles C Thomas; 1938. p.
5. Gazzano E, Chanteret C, Duvillard C, Folia M, Romanet P. A case of actinomycosis of the middle ear and a review of the literature. Int J Pediatr Otorhinolaryngol. 2010. 5: 70-3
6. Khosla VK, Banerjee AK, Chopra JS. Intracranial actinomycoma with osteomyelitis simulating meningioma. Case report. J Neurosurg. 1984. 60: 204-7
7. Mohindra S, Savardekar A, Rane S. Intracranial actinomycosis: Varied clinical and radiologic presentations in two cases. Neurol India. 2012. 60: 325-7
8. Narayan SK, Swaroop A, Jayanthi S. Chronic epidural intracranial actinomycosis: A rare case. Ann Indian Acad Neurol. 2009. 12: 195-6
9. Smego RA, Jr . Actinomycosis of the central nervous system. Rev Infect Dis. 1987. 9: 855-65
10. Sundaram C, Purohit AK, Prasad VS, Meena AK, Reddy JJ, Murthy JM. Cranial and intracranial actinomycosis. Clin Neuropathol. 2004. 23: 173-7
11. Wang S, Wolf RL, Woo JH, Wang J, O’Rourke DM, Roy S. Actinomycotic brain infection: Registered diffusion, perfusion MR imaging and MR spectroscopy. Neuroradiology. 2006. 48: 346-50
12. Wong VK, Turmezei TD, Weston VC. Actinomycosis. BMJ. 2011. 343: d6099-
13. Xia T, Baumgartner JC. Occurrence of Actinomyces in infections of endodontic origin. J Endod. 2003. 29: 549-52