- Department of Neurosurgery College of Medical Sciences, Bharatpur, Chitwan, Nepal
- Department of Surgery, Beth Israel Deaconess Medical Center, Harvard Medical School, Boston, MA 02215, USA
- Section of Neurosurgery, Department of Experimental Biomedicine and Clinical Neurosciences (BIONEC), University of Palermo, Palermo, Italy
Correspondence Address:
Iype Cherian
Section of Neurosurgery, Department of Experimental Biomedicine and Clinical Neurosciences (BIONEC), University of Palermo, Palermo, Italy
DOI:10.4103/2152-7806.192486
Copyright: © 2016 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Cherian I, Beltran M, Kasper EM, Bhattarai B, Munokami S, Grasso G. Exploring the Virchow–Robin spaces function: A unified theory of brain diseases. Surg Neurol Int 07-Oct-2016;7:
How to cite this URL: Cherian I, Beltran M, Kasper EM, Bhattarai B, Munokami S, Grasso G. Exploring the Virchow–Robin spaces function: A unified theory of brain diseases. Surg Neurol Int 07-Oct-2016;7:. Available from: http://surgicalneurologyint.com/surgicalint_articles/exploring-virchow-robin-spaces-function-unified-theory-brain-diseases/
Abstract
Background:Cerebrospinal fluid (CSF) transport across the central nervous system (CNS) is no longer believed to be on the conventional lines. The Virchow–Robin space (VRS) that facilitates CSF transport from the basal cisterns into the brain interstitial fluid (ISF) has gained interest in a whole new array of studies. Moreover, new line of evidence suggests that VRS may be involved in different pathological mechanisms of brain diseases.
Methods:Here, we review emerging studies proving the feasible role of VRS in sleep, Alzheimer's disease, chronic traumatic encephalopathy, and traumatic brain injury (TBI).
Results:In this study, we have outlined the possible role of VRS in different pathological conditions.
Conclusion:The new insights into the physiology of the CSF circulation may have important clinical relevance for understanding the mechanisms underlying brain pathologies and their cure.
Keywords: Alzheimer disease, chronic traumatic encephalopathy, cisternostomy, paravascular pathway, Virchow–Robin spaces
INTRODUCTION
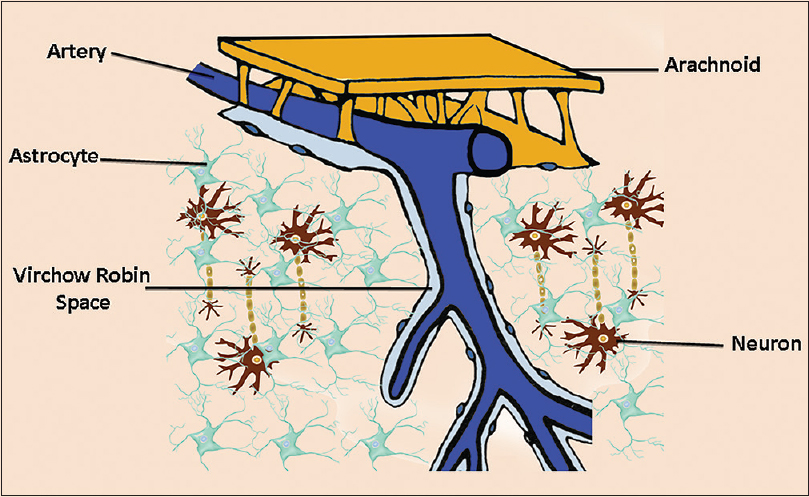
The classic model of cerebrospinal fluid (CSF) homeostasis is based on the circulation theory where the majority of CSF is considered to be produced by the choroid plexus and circulates from the ventricles into the subarachnoid spaces. Recently, however, this circulation model has been reconsidered because it is supposed that CSF can be produced and absorbed throughout the entire CSF system, and the pericapillary Virchow–Robin space (VRS) plays a critical role in the CSF system.[
The CSF communication from the basal cisterns to the brain interstitial fluid (ISF) through the VRS has put a lot of things into perspective. Accordingly, it has opened new avenues into the pathophysiological mechanisms of many diseases. The implications are far reaching from sleep and insomnia, Alzheimer disease, chronic traumatic encephalopathy (CTE), to traumatic brain injury (TBI), among others.
The VRS is seen around the vessels traversing from the cisterns into the brain. In 1851, Rudolph Virchow was the first to provide a detailed description of these microscopic spaces between the outer and inner/middle lamina of the brain vessels.[
Over time, the clinical significance of VRS has changed as the understanding of its function has evolved. Perivascular spaces are thought to play an important role in maintaining neural homeostasis in an environment with high metabolic activity in a system lacking a true lymphatic pathway.[
SLEEP
It is an unsolved question as to why humans and other animals are designed to sleep and what could be the biological function for such an essential function. Although many studies have suggested that sleeping is important for memory function,[
Furthermore, there are studies regarding the position of sleep and the direction of the head during sleep for maximum clearance. Interestingly, glymphatic transport seems to be most efficient in the recumbent, lateral position, especially on the right side.[
These observations strongly support studies analyzing the metabolic products in samples from the basal cisterns and simultaneously in the jugular vein during the day.
ALZHEIMER DISEASE
CSF flow through the VRS, which has long been known as “interstitial flow” and believed to play a role equivalent to the lymphatic system, has recently regained substantial attention due to its relation to beta-amyloid clearance.[
The neuropathological findings in Alzheimer advocate the deposition of amyloid-β (Aβ), tau protein, and neurofibrillary tangles.[
It is well-known that soluble Aβ can be removed from the brain by various clearance systems, such as cellular uptake following enzymatic activity, transport across the blood–brain barrier (BBB) and blood–cerebrospinal fluid barrier, and ISF bulk flow.[
Although in the past the majority of extracellular Aβ was believed to be cleared by the BBB,[
Because Aβ deposition can be increased in presymptomatic individuals years before the characteristic symptoms of Alzheimer disease clearly appear, a better understanding of the glymphatic system may provide new insight into the pathogenetic mechanisms with diagnostic and therapeutic value.
CHRONIC TRAUMATIC ENCEPHALOPATHY
CTE is a progressive neurodegenerative disorder caused by single or repetitive head trauma that does not result in observable neurological signs and symptoms of a concussion.[
Neuropathology findings include pigment-laden histiocytes in the VRS, which may be accompanied by sparse lymphocytic seeding.[
Further studies are warranted in order to detail the etiology of CTE and the role of the VRS.
TRAUMATIC BRAIN INJURY
Data from the literature show that TBI in the United States affects approximately 1.7 million people annually.[
Brain edema plays a main role in the pathophysiology of TBI and contributes to the mortality found in these patients.[
It has been suggested that the glymphatic removal of excess interstitial fluid decreases following injury,[
The fact that the cisterns are not seen in a scan of a patient with severe head injury along with a swollen brain may support such an occurrence. Fluid cannot be compressed and the amount of CSF (approximately 120 ml) contained in the cisterns cannot vanish anywhere in a short time. Furthermore, it cannot be shifted entirely into the spinal canal, as previously considered.
In this scenario, cisternostomy, a novel technique that incorporates the knowledge of skull base and microvascular surgery, has been recently proposed, and was found to decrease brain swelling, mortality, and morbidity.[
CONCLUSION
The understanding of VRS has been evolving and there is now a renewed interest in this field of research, especially after the glymphatic system has been described in humans.
In this study, we have outlined the possible role of VRS in different pathological conditions. Further studies are needed in order to clarify the role of the VRS in pathological conditions.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. . [Rudolf Ludwig Karl Virchow]. Prensa Med Mex. 1955. 20: 60-2
2. Alger SE, Chambers AM, Cunningham T, Payne JD. The role of sleep in human declarative memory consolidation. Curr Top Behav Neurosci. 2015. 25: 269-306
3. Ashworth A, Hill CM, Karmiloff-Smith A, Dimitriou D. Sleep enhances memory consolidation in children. J Sleep Res. 2014. 23: 302-8
4. Bushey D, Tononi G, Cirelli C. Sleep and synaptic homeostasis: Structural evidence in Drosophila. Science. 2011. 332: 1576-81
5. Buzsaki G. Memory consolidation during sleep: A neurophysiological perspective. J Sleep Res. 1998. 7: 17-23
6. Cherian I, Bernardo A, Grasso G. Cisternostomy for traumatic brain injury: Pathophysiological mechanisms and surgical technical notes. World Neurosurg. 2016. 29: 51-7
7. Cherian I, Yi G, Munakomi S. Cisternostomy: Replacing the age old decompressive hemicraniectomy?. Asian J Neurosurg. 2013. 8: 132-8
8. Cirelli C, Tononi G. Sleep and synaptic homeostasis. Sleep. 2015. 38: 161-2
9. Faul M, Sasser SM, Lairet J, Mould-Millman NK, Sugerman D. Trauma center staffing, infrastructure, and patient characteristics that influence trauma center need. West J Emerg Med. 2015. 16: 98-106
10. Gavett BE, Cantu RC, Shenton M, Lin AP, Nowinski CJ, McKee AC. Clinical appraisal of chronic traumatic encephalopathy: Current perspectives and future directions. Curr Opin Neurol. 2011. 24: 525-31
11. Gavett BE, Stern RA, McKee AC. Chronic traumatic encephalopathy: A potential late effect of sport-related concussive and subconcussive head trauma. Clin Sports Med. 2011. 30: 179-88
12. Grasso G. Surgical Treatment for Traumatic Brain Injury: Is It Time for Reappraisal?. World Neurosurg. 2015. 84: 594-
13. Guerreiro R, Hardy J. Genetics of Alzheimer's disease. Neurotherapeutics. 2014. 11: 732-7
14. Hay J, Johnson VE, Smith DH, Stewart W. Chronic Traumatic Encephalopathy: The Neuropathological Legacy of Traumatic Brain Injury. Annu Rev Pathol. 2016. 11: 21-45
15. Iliff JJ, Chen MJ, Plog BA, Zeppenfeld DM, Soltero M, Yang L. Impairment of glymphatic pathway function promotes tau pathology after traumatic brain injury. J Neurosci. 2014. 34: 16180-93
16. Iliff JJ, Wang M, Liao Y, Plogg BA, Peng W, Gundersen GA. A paravascular pathway facilitates CSF flow through the brain parenchyma and the clearance of interstitial solutes, including amyloid beta. Sci Transl Med. 2012. 4: 147ra111-
17. Kanaan NM, Cox K, Alvarez VE, Stein TD, Poncil S, McKee AC. Characterization of Early Pathological Tau Conformations and Phosphorylation in Chronic Traumatic Encephalopathy. J Neuropathol Exp Neurol. 2016. 75: 19-34
18. Karch CM, Cruchaga C, Goate AM. Alzheimer's disease genetics: From the bench to the clinic. Neuron. 2014. 83: 11-26
19. Kleindienst A, Dunbar JG, Glisson R, Marmarou A. The role of vasopressin V1A receptors in cytotoxic brain edema formation following brain injury. Acta Neurochir. 2013. 155: 151-64
20. Lee H, Xie L, Yu M, Kang H, Feng T, Deane R. The Effect of Body Posture on Brain Glymphatic Transport. J Neurosci. 2015. 35: 11034-44
21. Louveau A, Smirnov I, Keyes TJ, Eccles JD, Rouhani SJ, Peske JD. Structural and functional features of central nervous system lymphatic vessels. Nature. 2015. 523: 337-41
22. Marmarou A. A review of progress in understanding the pathophysiology and treatment of brain edema. Neurosurg Focus. 2007. 22: E1-
23. Mawuenyega KG, Sigurdson W, Ovod V, Munsell L, Kasten T, Morris JC. Decreased clearance of CNS beta-amyloid in Alzheimer's disease. Science. 2010. 330: 1774-
24. McKee AC, Cantu RC, Nowinski CJ, Hedley-Whyte ET, Gavett BE, Budson AE. Chronic traumatic encephalopathy in athletes: Progressive tauopathy after repetitive head injury. J Neuropathol Exp Neurol. 2009. 68: 709-35
25. McKee AC, Stein TD, Kiernan PT, Alvarez VE. The neuropathology of chronic traumatic encephalopathy. Brain Pathol. 2015. 25: 350-64
26. McKee AC, Stern RA, Nowinski CJ, Stein TD, Alvarez VE, Daneshvar DH. The spectrum of disease in chronic traumatic encephalopathy. Brain. 2013. 136: 43-64
27. Omalu B, Hammers JL, Bailes J, Hamilton RL, Kamboh MI, Webster G. Chronic traumatic encephalopathy in an Iraqi war veteran with posttraumatic stress disorder who committed suicide. Neurosurg Focus. 2011. 31: E3-
28. Omalu BI, Bailes J, Hammers JL, Fitzsimmons RP. Chronic traumatic encephalopathy, suicides and parasuicides in professional American athletes: The role of the forensic pathologist. Am J Forensic Med Pathol. 2010. 31: 130-2
29. Oreskovic D, Klarica M. The formation of cerebrospinal fluid: Nearly a hundred years of interpretations and misinterpretations. Brain Res Rev. 2010. 64: 241-62
30. Potter R, Patterson BW, Elbert DL, Ovod V, Kasten T, Sigurdson W. Increased in vivo amyloid-beta42 production, exchange, and loss in presenilin mutation carriers. Sci Transl Med. 2013. 5: 189ra177-
31. Reiman EM, Quiroz YT, Fleisher AS, Chen K, Velez-Pardo C, Jimenez-Del-Rio M. Brain imaging and fluid biomarker analysis in young adults at genetic risk for autosomal dominant Alzheimer's disease in the presenilin 1 E280A kindred: A case-control study. Lancet Neurol. 2012. 11: 1048-56
32. Schley D, Carare-Nnadi R, Please CP, Perry VH, Weller RO. Mechanisms to explain the reverse perivascular transport of solutes out of the brain. J Theor Biol. 2006. 238: 962-74
33. Shibata M, Yamada S, Kumar SR, Calero M, Bading J, Frangione B. Clearance of Alzheimer's amyloid-ss (1-40) peptide from brain by LDL receptor-related protein-1 at the blood-brain barrier. J Clin Invest. 2000. 106: 1489-99
34. Siegel JM. Clues to the functions of mammalian sleep. Nature. 2005. 437: 1264-71
35. Stern RA, Daneshvar DH, Baugh CM, Seichepine DR, Montenigro PH, Riley DO. Clinical presentation of chronic traumatic encephalopathy. Neurology. 2013. 81: 1122-9
36. Tarasoff-Conway JM, Carare RO, Osorio RS, Glodzik L, Butler T, Fieremans E. Clearance systems in the brain-implications for Alzheimer disease. Nat Rev Neurol. 2015. 11: 457-70
37. Weed LH. Studies on Cerebro-Spinal Fluid. No. II: The Theories of Drainage of Cerebro-Spinal Fluid with an Analysis of the Methods of Investigation. J Med Res. 1914. 31: 21-49
38. Weller RO, Djuanda E, Yow HY, Carare RO. Lymphatic drainage of the brain and the pathophysiology of neurological disease. Acta Neuropathol. 2009. 117: 1-14
39. Xie L, Kang H, Xu Q, Chen MJ, Liao Y, Thiyagarajan M. Sleep drives metabolite clearance from the adult brain. Science. 2013. 342: 373-7






Mary T Blazewicz
Posted July 24, 2022, 1:04 pm
I was diagnosed with Virchow-Robin. I was told I have a hole in my head that veins travel through. Doctor didn’t say anything about it. But reading this, I’m scared.