- Department of Neurology, Beth Israel Deaconess Medical Center, Boston, MA, USA
- Department of Neurosurgery, Beth Israel Deaconess Medical Center, Boston, MA, USA
Correspondence Address:
Ekkehard M. Kasper
Department of Neurosurgery, Beth Israel Deaconess Medical Center, Boston, MA, USA
DOI:10.4103/2152-7806.189441
Copyright: © 2016 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Brown MW, Tülin Serap Yilmaz, Kasper EM. Iatrogenic spinal hematoma as a complication of lumbar puncture: What is the risk and best management plan?. Surg Neurol Int 29-Aug-2016;7:
How to cite this URL: Brown MW, Tülin Serap Yilmaz, Kasper EM. Iatrogenic spinal hematoma as a complication of lumbar puncture: What is the risk and best management plan?. Surg Neurol Int 29-Aug-2016;7:. Available from: http://surgicalneurologyint.com/surgicalint_articles/iatrogenic-spinal-hematoma-complication-lumbar-puncture-risk-best-management-plan/
Abstract
Background:Lumbar puncture (LP) rarely results in complications such as spinal hematomas. However, it remains unclear if certain variables increase likelihood of these events, or if surgical intervention improves outcome.
Methods:In addition to two clinical vignettes, we evaluated the post-1974 literature for cases of spinal hematoma and subsequent intervention. Based on our compilation of data, we evaluated outcome relative to numerous distinct variables.
Results:Based on 35 LP-related spinal hematoma cases in the post-1974 literature and our encounters, we found 28.6% of patients presenting with preexisting coagulopathy had poor outcomes regardless of intervention, relative to 14.3% of patients without coagulopathy; a highly significant difference (P = 0.02). Once diagnosed, 21 patients were treated surgically and 14 nonsurgically. Of the 60% surgical patients, 57.1% had good outcomes, and 42.9% had poor outcomes within 12 months. Of 40% nonsurgical patients, 57.1% had good outcomes and 42.9% had poor outcomes. Results in these groups were not statistically different.
Conclusions:We found a significant correlation between preexisting coagulopathy and poor neurological outcome irrespective of intervention. However, outcomes for these patients may be confounded by comorbidities including underlying conditions contributing to their coagulopathy. No significant correlation between type of surgical intervention and good outcome was found, possibly attributable to the paucity of details in existing case reports and the difficulty defining the degree of spinal cord compromise from a given lesion. Despite our findings, emergent neurosurgical intervention may be beneficial for the management of complications such as cauda equina syndrome secondary to intrathecal spinal hematoma.
Keywords: Cauda equina, epidural hematoma, lumbar puncture, mass effect, spinal hematoma, spinal hemorrhage, subdural hematoma
INTRODUCTION
Patients with neurologic dysfunction often require a lumbar puncture (LP) for diagnosis of the underlying etiology, which includes infectious, autoimmune, or mechanical causes. Beyond this, LP may prove therapeutic for patients with increased intracranial pressure, such as those with idiopathic intracranial hypertension or cryptococcal meningitis, where LP offers symptomatic relief from decreasing cerebrospinal fluid (CSF) volume.
Complications range from the more common post-LP headache from reduced intracranial pressure to more rare cases of infection from nonsterile procedure, or trauma to local structures such as tissue, vessels, and nerve roots. Should the procedure result in any significant violation of vasculature, hematoma may occur resulting commonly in mild pain, skin discoloration (superficial “black-and-blue” mark), and ultimately, resolution without complication. If the vascular disruption occurs near or inside, the thecal sac, hematoma formation could exert mass effect resulting in compression of nerve roots, or the spinal cord proper. Severe complications, such as myelopathy or cauda equina syndrome (CES), require expeditious diagnosis and potential intervention to prevent irreversible paralysis or death.
Spinal (including epidural, subdural, and subarachnoid) hematomas were described in autopsies as early as 1682,[
In 2003, Kreppel et al., performed a large meta-analysis of the available literature, including 613 spinal hematoma patients reported between 1826 and 1996.[
CASE STUDIES
We present two cases from Beth Israel Deaconess Medical Center, Boston, Massachusetts, USA, in January and August 2014; one as a direct Emergency Department presentation, the second as a transfer from an outside facility for escalation of care. In each case, we elaborate on the presenting symptoms, neurosurgical evaluation, and intervention. Of note, neither patient had previously been administered anticoagulant medication, was subsequently found to have any clotting disorder or had prior surgical manipulation in the area of hematoma formation.
Case 1
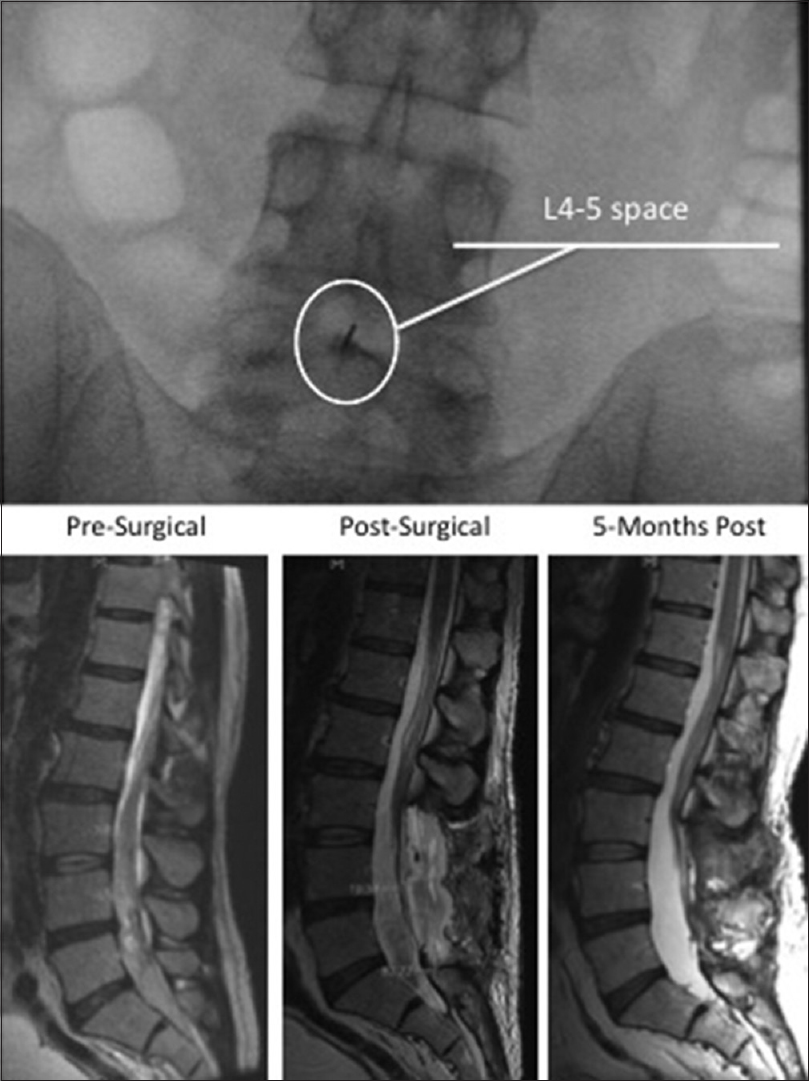
A 54-year-old man with a history of diverticulitis complicated by intra-abdominal abscess status postbowel resection presented with nuchal rigidity, proximal weakness from upper extremities to lower extremities, and associated numbness shortly after surgery. Admission LP revealed a leuko-pleocytosis (146 cells/μl) with 93% lymphocytes and elevated protein (162 mg/dL). Antibiotics were initiated with a repeated tap performed 4 days after initial LP. Concern for autoimmune disorder grew when antibiotic coverage failed to resolve the patient's bilateral inflammatory cervical polyradiculopathies. A course of intravenous immunoglobulin was administered with marginal improvement of his weakness. Repeat tap performed under fluoroscopic guidance 11 days postinitial LP demonstrated resolution of the leukocytosis; however, the tap was notably traumatic with red blood cells (RBCs) above 31,000/μl.
On postadmission day 19 and 8 days status post-LP under fluoroscopy, the patient reported severe lower back pain, worsening lower extremity weakness, saddle anesthesia, and urinary retention. Magnetic resonance imaging (MRI) of the lumbar spine with and without gadolinium contrast demonstrated an intrathecal subarachnoid and subdural hematoma extending from L3 through the sacral levels [
Urgent neurosurgery consultation resulted in decompressive surgery within 24-h of symptom onset. Laminectomies were performed between inferior L2 and L5, at which time a small ventrolateral epidural hematoma was identified compressing the spinal cord at the L4 level. Initially, the thecal sac appeared tightly stretched and bluish discolored from intradural hemorrhage; however, removal of the epidural hematoma and maneuvers to mobilize the dura and spinal cord dislodged the intradural collection as evidenced by adequate ventilation-synchronous respiratory pulsations of the CSF with each breath, as well as a slack and tension-free thecal sac. As a result, the dura was not opened for clot evacuation since decompression (the goal of surgery) had been achieved without perioperative complication.
Postoperatively, the patients’ urinary retention resolved and the patient began ambulating with wide-based stable gait. Follow-up appointments at 6 and 12 months revealed significant improvements in strength with near resolution of all proximal weakness and only residual dyspnea secondary to underlying pulmonary disease. No bowel/bladder dysfunction, lower extremity weakness, or anesthesia was present.
Case 2
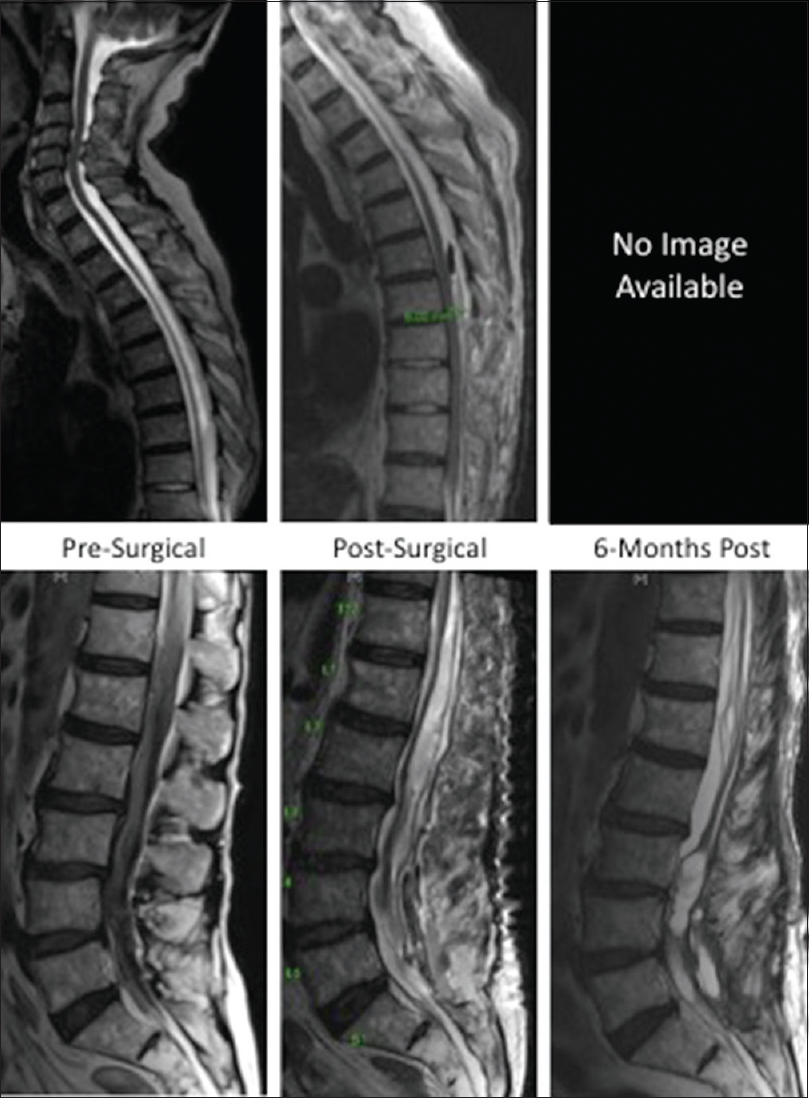
An 83-year-old man with a history of coronary artery disease status postquintuple coronary artery bypass grafts on no anticoagulation, or antiplatelet agent, initially presented to an outside hospital for evaluation of severe, sudden-onset headache with a subsequent diagnostic LP performed in the Emergency Department. Shortly after this intervention, the patient developed severe lower back pain and bilateral lower extremity weakness. Imaging with MR of the thoracic and lumbar spine demonstrated a thoracolumbar supposedly epidural hematoma for which the patient was admitted for nonsurgical management, which by report resulted in some improvement of lower extremity weakness. However, persistent motor deficits, and increasing difficulty ambulating with a rolling walker prompted a transfer to our tertiary care center for evaluation and potential intervention.
MRI performed at 3 weeks after symptom onset demonstrated a large subdural hematoma spanning from T7 to the sacrum, which exerted a considerable mass effect on the thoracic cord and cauda equina [
Postoperatively, the patient demonstrated resolution of the underlying hematoma without evident caudal nerve root compromise. After titration off pain management and steroids, he was discharged to an acute rehabilitation facility on hospital day 8. At 6-months, strength and sensation in the lower extremities recovered without any residual deficit. Repeat MRI demonstrated complete resolution of the intrathecal hematoma (not shown).
METHODS OF LITERATURE REVIEW
Through review of existing case studies and our own experience from two direct patient encounters, we evaluated all reported adult and pediatric cases of hematomas resulting from puncture (or attempted puncture) of the dura. A literature search was performed identifying articles between 1974 and 2014, which documented cases of spinal hematoma specifically associated with LP or spinal anesthesia. The articles were sourced through the use of a full-text PubMed (United States National Library of Medicine, Bethesda, Maryland, USA) search using various query strings including “spinal hematoma,” “lumbar puncture hematoma,” and “lumbar puncture complication.”
We restricted our search to full case reviews during this period containing all procedural details, excluding case data referred to by other articles, unless the referenced study was also available for review. The cases documented in this pooled cohort represent patients cared for in 11 countries on 3 continents. In each of the evaluated case studies, symptoms were considered a complication of LP as the tap immediately preceded symptom onset without other interventions, or prior trauma, which might have accounted for the hemorrhage.
We identified distinct variables thought to potentially correlate with spinal hematoma formation, such as needle gauge, traumatic tap, and existing coagulopathy. When documented, we collected time from LP to onset of neurologic symptoms exceeding isolated back pain, as well as time from onset of such symptoms to intervention. Finally, we documented the assessment of each patient's outcome at 6–12 months status postonset of symptoms.
Although some articles contained qualitative descriptors such as “good,” “fair,” and “poor” to categorize deficits, the meaning of these assessments varied too significantly between cases. We standardized categorization by compiling symptoms into one of three outcomes. “Good” refers to patients without deficit, or mild sensorimotor deficit without impairment of ambulation or bowel/bladder dysfunction. “Poor” refers to patients with significant impairment, inability to ambulate without support, or bowel/bladder dysfunction. “Expired” refers to patients who did not survive at 12 months postevent. For the purposes of evaluation, “poor” and “expired” are separately assessed but collectively analyzed as undesirable outcomes.
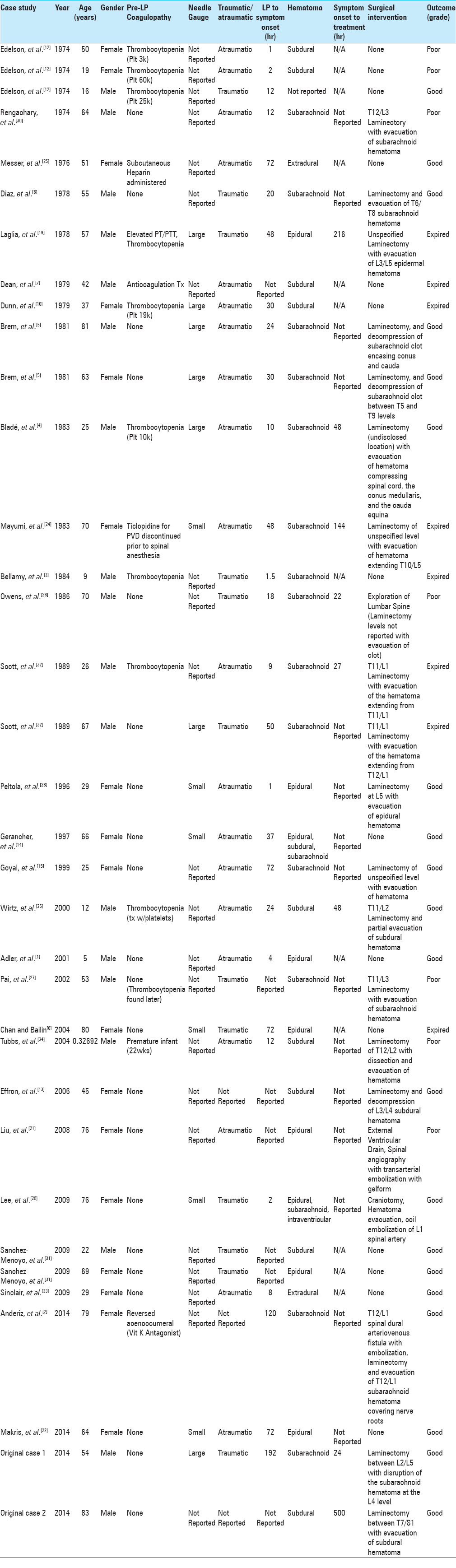
As LP-related spinal hematomas are extremely rare, only 33 case reports were found through our literature review from 1974 to 2014; these, along with the 2 cases we encountered, are detailed in
RESULTS OF LITERATURE EVALUATION
Through our analysis, we sought to determine whether a correlation could be made between inciting factors and the development of spinal hematoma, or between any intervention and the reported outcome.
With regards to general characteristics of our cohort, gender was evenly split between men (51%) and women (49%). Ages recorded in these cases ranged from 17 months to 83 years of age, with a median age of 53 years and a mean age of 48 years, suggesting a relatively symmetric distribution. Racial distribution was not reported; however, from a geographical perspective, the majority of cases (63%) originated in the United States.
When examining specific variables thought to contribute to the development of spinal hematoma, we identified 15 (42.9%) patients who had some underlying coagulopathy (or iatrogenic administration of anticoagulation before LP). For these 15 patients, 10 (28.6%) had poor outcome, including 6 (17.1%) who expired, relative to 5 (14.3%) with good outcome.
In the 20 (57.1%) noncoagulopathic patients, only 5 (14.3%) had poor outcomes, including 2 (5.7%) expired patients, whereas 15 (42.9%) patients showed good outcome at 12 months.
With regards to these outcomes stratified by preexisting coagulopathy, we found a statistically significant difference between coagulopathic patients suffering a poor outcome relative to their peers without coagulopathy (P = 0.0192; using Fisher's exact test).
We also evaluated the relationship between the gauge of the spinal needle used and the development of a hematoma; however, only 13 (37.1%) of the published patient case studies included in this analysis also reported the caliber of the needle. With regard to the outcome, no meaningful correlation was found between the 7 (20%) of patients reporting LP with larger caliber (gauge ≤ 20) and good outcome (P = 1.0000), or the 6 (17.1%) of patients reporting LP with smaller caliber (gauge > 20) and good outcome (P = 0.6730 using Fisher's exact test).
In 32 of 35 cases, the character or the tap (e.g., traumatic, high RBC) was reported. To categorize the results, we arbitrarily assigned a “traumatic” designation to taps, which were either designated “traumatic” in the original article, noted as “bloodstained” or with RBC count as >100 RBC/mm3. The “atraumatic” designation referred to those who did not have a successful tap (without reported bleeding), had clear CSF, or a cell count of <100 RBC/mm3. With this conservative categorization, 12/32 (34.4%) of publications reported a traumatic tap and 20/32 reported (62.5%) an atraumatic tap. When comparing the outcomes of the atraumatic patients, 11 had good outcome and 9 had poor outcome (with 5 patients expired by 12 months). The results of the traumatic group were split evenly with 6 experiencing good outcome, and 6 experiencing poor outcome (with 4 deaths by 12 months). However, no statistical difference could be seen between presence of traumatic tap and outcome (P = 1.0000 using Fisher's exact test).
With a relatively small and pooled cohort size as ours and only limited data reporting on the respective therapeutic intervention performed, it is not easily possible to derive at strong conclusions or recommendation that is based on specific clinical management decisions. However, we were interested in assessing a possible difference in outcome based on the decision of whether symptomatic patients were taken for surgical intervention.
Comparing patients undergoing some form of surgical intervention to patients managed nonsurgically (medically managed group), our analysis revealed 21 patients treated surgically and 14 patients managed nonsurgically. Of the 21 (60%) surgical patients, 12 (57.1%) had good outcome, defined as mild sensorimotor loss without any voiding dysfunction of either bowel or bladder or no reported deficit at all. Nine (42.9%) had poor outcomes (severe paraparesis, voiding dysfunction, or they expired [4 (19.0%)] within a 12 months period). With regards to the 14 (40%) nonsurgically managed patients, 8 (57.1%) had good outcome and 6 (42.9%) had poor outcome with 4 of these patients (28.6%) expiring within 12 months.
Using Fisher's exact test, a comparison of surgical versus nonsurgical intervention reveals exactly the same proportion of good to bad outcomes with any surgical intervention as without (P = 1.0000). Comparison of expiration at 12 months in surgical versus nonsurgical intervention demonstrates a difference in the percentage of death 19.0% surgical versus 28.6% nonsurgical; however, this difference was not found to be significant (P = 0.6853 using Fisher's exact test).
To determine whether time from symptom onset to decompression translated into a clinical outcome difference, we planned to review the time to surgical intervention, which was only noted in 8 of the 21 surgical cases. Unfortunately, as it turns out in many cases, such timing was not reported in detail. Thus, we abstained from any further analysis, since any inference of correlation to outcome would be of dubious significance.
DISCUSSION
Based on the differing interventions between our two LP-related spinal hematoma patients and case review, we sought to determine if a pertinent guideline can be established to advise the health care provider on suitable or even specific management. We also explored whether factors exist that correlate with better outcome.
Through evaluation of our cohort of 35 total LP-related spinal hematoma cases, we demonstrate that the presence of preexisting coagulopathy correlates with worse outcome at 6–12 months, relative to patients in whom spinal hematoma developed without coagulopathy. Clotting abnormalities included thrombocytopenia, preexisting use of anticoagulants, Vitamin K antagonists, antiplatelet medications, or in one pediatric case, extreme prematurity. However, such a correlation has significant confounders preventing a causal link to be made. The nature of underlying diseases (e.g., thrombocytopenia in leukemia), or disease (e.g., coronary artery disease) requiring the patient to be on long-term postoperative anticoagulation may in themselves contribute to the poor outcome through a multitude of factors rather than simple hypocoagulability. With regard to our primary aim to determine whether patients with spinal hematoma experienced better outcomes through surgical or medical (nonsurgical) intervention, we did not find a statistically significant difference between the reported outcomes of either group.
We also contrasted the “expired” subset of poor outcomes against good outcomes with regard to either surgical or medical intervention, but again did not find a statistically significant difference. However, several confounders may factor into our inability to demonstrate a correlation. Due to the rarity of these occurrences, and the even less frequently reported surgically managed cases, our overall sample size is relatively small giving our analysis limited power. Thus, we have constrained the ability to reject any assertion that medical management is correlated with identical or superior outcomes. The scenario is further complicated, as the intent and indication to treat may have been biased toward the chosen management (several patients suffering from spinal hematoma were deemed poor candidates for surgical intervention, or refused such intervention despite being good candidates). Furthermore, the variance in technical proficiency of the neurosurgical services between more than 30 reporting sites across eleven countries may confound the outcome.
Two other factors, time from LP to symptom onset and time from symptom onset to treatment, were considered relevant as they might significantly impact outcome; however, due to the underreporting of these variables across cases, we did not identify a valid recommendation. We noted the similarity of our findings to the ongoing highly controversial debate on time to treatment influencing outcome for all cases of CES. Some studies found a statistically significant improved outcome in symptomatic CES patients who receive the early surgical intervention,[
CONCLUSIONS
Important considerations for patients requiring LP or spinal anesthesia with the potential consequence of an iatrogenic spinal cord hematoma include the presence of any preexisting coagulopathy, for which we demonstrated correlation with worse outcomes. Whenever possible, we suggest aggressive, proactive management of coagulopathy before any intervention, as this may decrease the incidence of such potentially devastating injuries.
Although we were unable to demonstrate significant differences between distinct modalities of intervention, we did not ascertain inferior treatment. In the absence of available data, the indication and choice for treatment remain highly individualized. It is our opinion that surgical management remains an appropriate intervention for neurologic emergencies caused by spinal hematoma such as CES.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Adler MD, Comi AE, Walker AR. Acute hemorrhagic complication of diagnostic lumbar puncture. Pediatr Emerg Care. 2001. 17: 184-8
2. Andériz FO, Malpica AB, Sorribas EM, Clotet NC, Chasín JA. Paraplegia after lumbar puncture. Am J Med Sci. 2014. 347: 170-1
3. Bellamy EA, Perez DJ, Husband JE. CT demonstration of a spinal subarachnoid haematoma following lumbar puncture. J Comput Assist Tomogr. 1984. 8: 791-2
4. Bladé J, Gastón F, Montserrat E, Marín P, Grañena A, Bachs A. Spinal subarachnoid hematoma after lumbar puncture causing reversible paraplegia in acute leukemia. Case report. J Neurosurg. 1983. 58: 438-9
5. Brem SS, Hafler DA, Van Uitert RL, Ruff RL, Reichert WH. Spinal subarachnoid hematoma: A hazard of lumbar puncture resulting in reversible paraplegia. N Engl J Med. 1981. 304: 1020-1
6. Chan L, Bailin MT. Spinal epidural hematoma following central neuraxial blockade and subcutaneous enoxaparin: A case report. J Clin Anesth. 2004. 16: 382-5
7. Dean WM, Woodside JR. Spinal hematoma compressing cauda equina. Urology. 1979. 13: 575-7
8. Diaz FG, Yock DH, Rockswold GL. Spinal subarachnoid hematoma after lumbar puncture producing acute thoracic myelopathy: Case report. Neurosurgery. 1978. 3: 404-6
9. Dripps RD, Vandam LD. Hazards of lumbar puncture. J Am Med Assoc. 1951. 147: 1118-21
10. Dunn D, Dhopesh V, Mobini J. Spinal subdural hematoma: A possible hazard of lumbar puncture in an alcoholic. JAMA. 1979. 241: 1712-3
11. Duverney GJ. Spinal Asphyxia. The History of the Royal Academy of Sciences. 1682. 2: 28-
12. Edelson RN, Chernik NL, Posner JB. Spinal subdural hematomas complicating lumbar puncture: Occurrence in thrombocytopenic patients. Arch Neurol. 1974. 31: 134-7
13. Effron D, Schmidt KL, Sharma A. Subdural hematoma. J Emerg Med. 2006. 30: 93-4
14. Gerancher JC, Waterer R, Middleton J. Transient paraparesis after postdural puncture spinal hematoma in a patient receiving ketorolac. Anesthesiology. 1997. 86: 490-4
15. Goyal A, Dua R, Singh D, Kumar S. Spinal subarachnoid haematoma following lumbar puncture. Neurol India. 1999. 47: 339-40
16. Jackson R. Case of spinal apoplexy. Lancet. 1869. 2: 5-6
17. Kennedy JG, Soffe KE, McGrath A, Stephens MM, Walsh MG, McManus F. Predictors of outcome in cauda equina syndrome. Eur Spine J. 1999. 8: 317-22
18. Kreppel D, Antoniadis G, Seeling W. Spinal hematoma: A literature survey with meta-analysis of 613 patients. Neurosurg Rev. 2003. 26: 1-49
19. Laglia AG, Eisenberg RL, Weinstein PR, Mani RL. Spinal epidural hematoma after lumbar puncture in liver disease. Ann Intern Med. 1978. 88: 515-6
20. Lee SJ, Lin YY, Hsu CW, Chu SJ, Tsai SH. Intraventricular hematoma, subarachnoid hematoma and spinal epidural hematoma caused by lumbar puncture: An unusual complication. Am J Med Sci. 2009. 337: 143-5
21. Liu WH, Lin JH, Lin JC, Ma HI. Severe intracranial and intraspinal subarachnoid hemorrhage after lumbar puncture: A rare case report. Am J Emerg Med. 2008. 26: 633.e1-3
22. Makris A, Gkliatis E, Diakomi M, Karmaniolou I, Mela A. Delayed spinal epidural hematoma following spinal anesthesia, far from needle puncture site. Spinal Cord. 2014. 52: S14-6
23. Masdeu JC, Breuer AC, Schoene WC. Spinal subarachnoid hematomas: Clue to a source of bleeding in traumatic lumbar puncture. Neurology. 1979. 29: 872-6
24. Mayumi T, Dohi S. Spinal subarachnoid hematoma after lumbar puncture in a patient receiving antiplatelet therapy. Anesth Analg. 1983. 62: 777-9
25. Messer HD, Forshan VR, Brust JC, Hughes JE. Transient paraplegia from hematoma after lumbar puncture. A consequence of anticoagulant therapy. JAMA. 1976. 235: 529-30
26. Owens EL, Kasten GW, Hessel EA. Spinal subarachnoid hematoma after lumbar puncture and heparinization: A case report, review of the literature, and discussion of anesthetic implications. Anesth Analg. 1986. 65: 1201-7
27. Pai SB, Krishna KN, Chandrashekar S. Post lumbar puncture spinal subarachnoid hematoma causing paraplegia: A short report. Neurol India. 2002. 50: 367-9
28. Peltola J, Sumelahti ML, Kumpulainen T, Dastidar P, Helén P. Spinal epidural haematoma complicating diagnostic lumbar puncture. Lancet. 1996. 347: 131-
29. Qureshi A, Sell P. Cauda equina syndrome treated by surgical decompression: The influence of timing on surgical outcome. Eur Spine J. 2007. 16: 2143-51
30. Rengachary SS, Murphy D. Subarachnoid hematoma following lumbar puncture causing compression of the cauda equina. Case report. J Neurosurg. 1974. 41: 252-4
31. Sánchez-Menoyo JL, Ruiz-Ojeda J, Martínez-Arroyo A, García-Moncó JC, Aduna-De Paz M, Vicente-Olabarría I. Spinal cord hemorrhage complicating diagnostic lumbar puncture. Rev Neurol. 2009. 48: 418-20
32. Scott EW, Cazenave CR, Virapongse C. Spinal subarachnoid hematoma complicating lumbar puncture: Diagnosis and management. Neurosurgery. 1989. 25: 287-92
33. Sinclair AJ, Carroll C, Davies B. Cauda equina syndrome following a lumbar puncture. J Clin Neurosci. 2009. 16: 714-6
34. Tubbs RS, Smyth MD, Wellons JC, Oakes WJ. Intramedullary hemorrhage in a neonate after lumbar puncture resulting in paraplegia: A case report. Pediatrics. 2004. 113: 1403-5
35. Wirtz PW, Bloem BR, van der Meer FJ, Brouwer OF. Paraparesis after lumbar puncture in a male with leukemia. Pediatr Neurol. 2000. 23: 67-8