- Department of Neurosurgery, University Hospital Río Hortega, Valladolid, Spain.
- Department of Radiology, University Hospital Río Hortega, Valladolid, Spain.
Correspondence Address:
Santiago Cepeda
Department of Neurosurgery, University Hospital Río Hortega, Valladolid, Spain.
DOI:10.25259/SNI_862_2020
Copyright: © 2020 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Santiago Cepeda1, Sergio García-García1, Ignacio Arrese1, María Velasco-Casares2, Rosario Sarabia1. Acute changes in diffusion tensor-derived metrics and its correlation with the motor outcome in gliomas adjacent to the corticospinal tract. 10-Feb-2021;12:51
How to cite this URL: Santiago Cepeda1, Sergio García-García1, Ignacio Arrese1, María Velasco-Casares2, Rosario Sarabia1. Acute changes in diffusion tensor-derived metrics and its correlation with the motor outcome in gliomas adjacent to the corticospinal tract. 10-Feb-2021;12:51. Available from: https://surgicalneurologyint.com/?post_type=surgicalint_articles&p=10579
Abstract
Background: This study involves analysis of the relationship between variables obtained using diffusion tensor imaging (DTI) and motor outcome in gliomas adjacent to the corticospinal tract (CST).
Methods: Histologically confirmed glioma patients who were to undergo surgery between January 2018 and December 2019 were prospectively enrolled. All patients had a preoperative magnetic resonance imaging (MRI) study that included DTI, a tumor 2 cm or less from the CST, and postsurgical control within 48 h. Patients with MRI that was performed at other center, tumors with primary and premotor cortex invasion, postsurgical complications directly affecting motor outcome and tumor progression
Results: Eleven patients were analyzed, and six corresponded to high-grade gliomas and five to low-grade gliomas. Four patients had previous motor impairment and seven patients had postsurgical motor deficits (four transient and three permanent). An FA ratio of 0.8 between peritumoral CST regions and the contralateral hemisphere was found to be the cutoff, and lower values were obtained in patients with permanent motor deficits.
Conclusion: Quantitative analysis of DTI that was performed in the immediate postsurgery period can provide valuable information about the motor prognosis after surgery for gliomas near the CST.
Keywords: Corticospinal tract, Diffusion tensor imaging, Fiber tracking, Glioma, Motor outcome
INTRODUCTION
Gliomas are neoplasms that are characterized by their ability to extend through white matter (WM) tracts compromising structures that are involved in movement, language, and cognition. Gliomas located near the corticospinal tract (CST) are a surgical challenge because of the proximity between the borders of the resection and important structures that may lead to neurosurgical deficits. Surgery on these tumors has reached a high level of technological complexity, using tools such as intraoperative fluorescence, neuronavigation, and neurophysiological monitoring.[
The motor cortex is divided into the primary motor cortex, premotor cortex, cingulate motor area, and supplementary motor area. The CST is the main pathway that is involved in voluntary movement control. Pyramidal neurons of the primary motor cortex form the CST, which also receives fibers from the accessory motor areas, and from the parietal/ somatosensory cortex.[
Diffusion tensor imaging (DTI) is a non-invasive magnetic resonance imaging (MRI) technique that measures the anisotropic diffusion of water molecules. WM tractography, which is also known as fiber tracking, allows a three-dimensional visualization of WM tracts. In addition, the architecture and integrity of WM fascicles can be assessed using a mathematical model that is composed of eigenvalues (λ) and eigenvectors (ε) by which the displacement of the water molecule signal within a voxel is characterized.[
Several publications have studied the acute diffusivity changes in WM and selected regions of the CST in patients early after a stroke. A relationship between decreased anisotropic diffusivity and worse motor outcome has been described.[
For brain tumors, there are few studies that use DTI as a prognostic tool for motor function.[
MATERIALS AND METHODS
We prospectively enrolled histologically confirmed glioma patients who were to undergo surgery in our department between January 2018 and December 2019. All patients had a preoperative MRI study that included DTI. We included patients in whom the tumor was located 2 cm or less from the CST and who had postsurgical MRI control within 48 h. The surgeries were performed in a standardized manner using microsurgical techniques, neuronavigation, neurophysiological monitoring, and intraoperative ultrasound.
We excluded patients with MRI that was performed at other centers, tumors that invaded the primary motor cortex, supplementary motor area, and premotor cortex, and patients who had postsurgical complications with a direct impact on motor outcome (hematomas or ischemia). Stereotactic biopsies and patients with tumor progression for <6 months that were demonstrated in control MRI studies were also excluded from the study.
The clinical and radiological variables are summarized in [
Image acquisition protocol
All subjects were scanned using a 1.5 T MRI scanner (Signa HDxt; GE Healthcare, Milwaukee, WI, USA). DTI data were acquired using a single shot echo-planar imaging sequence (TE, 96 ms; TR, 13675 ms; field of view, 256 × 256 mm; matrix size, 128 × 128; voxel size, 1.015 × 1.015 × 3 mm). The diffusion-weighting gradient was applied in 25 isotropically distributed directions using a b value of 1000 s/mm2. Fifty gapless slices were obtained to cover the whole brain, with a thickness of 3 mm. The total DTI acquisition time was approximately 4 min. The b-table was checked by an automatic quality control routine to ensure its accuracy.[
Imaging analysis and fiber tracking
Fiber tracking was performed with a deterministic algorithm using DSI Studio (dsi-studio.labsolver.org).[
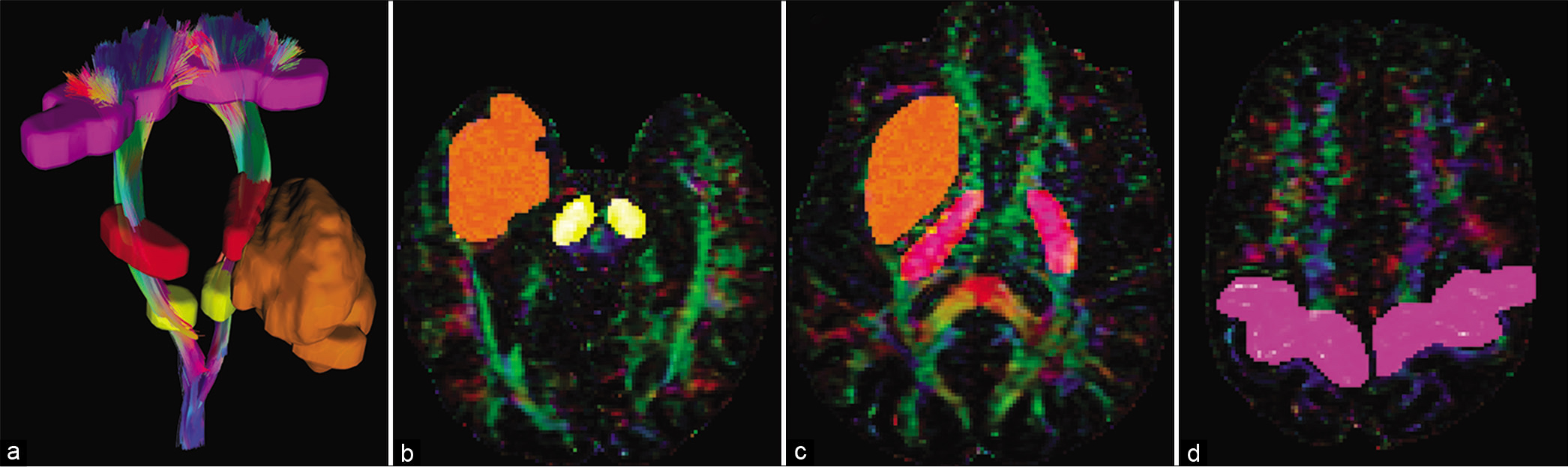
Fig 1:
Example of the methodology used for fiber tracking. (a) Three-dimensional visualization of CST tracts (ipsitumoral and healthy) along with the tumor and regions of interest (ROIs) that were used. Axial slices of diffusion tensor imaging-based color map showing ROI for the midbrain (b), posterior limb of internal capsule (c), and primary motor cortex (d).
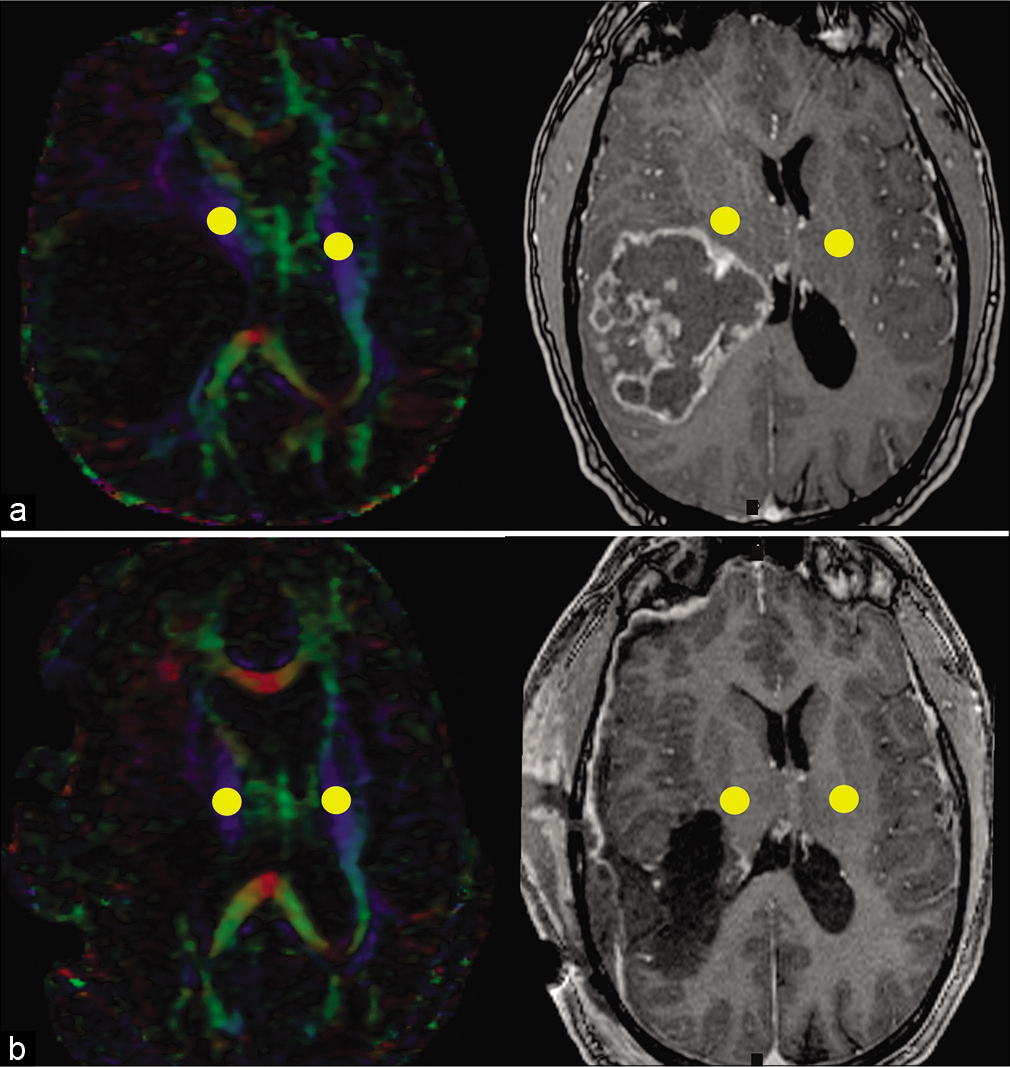
Fig 2:
Illustrative case showing the methodology that was used to determine the peritumoral corticospinal tract (CST) region. (a) Preoperative diffusion tensor imaging (DTI) color map (left) and T1-weighted postcontrast (right) axial images showing a high grade glioma adjacent to CST. ROI is placed over the area corresponding to the CST, as determined by the color fiber direction. A second ROI is placed in the contralateral hemisphere. (b) Postoperative DTI color map (left) and T1-weighted postcontrast (right) axial images. The ipsilateral ROI is placed in the CST region that is adjacent to the postsurgical cavity; a second ROI is placed in the contralateral CST region in the healthy hemisphere.
Statistical analysis
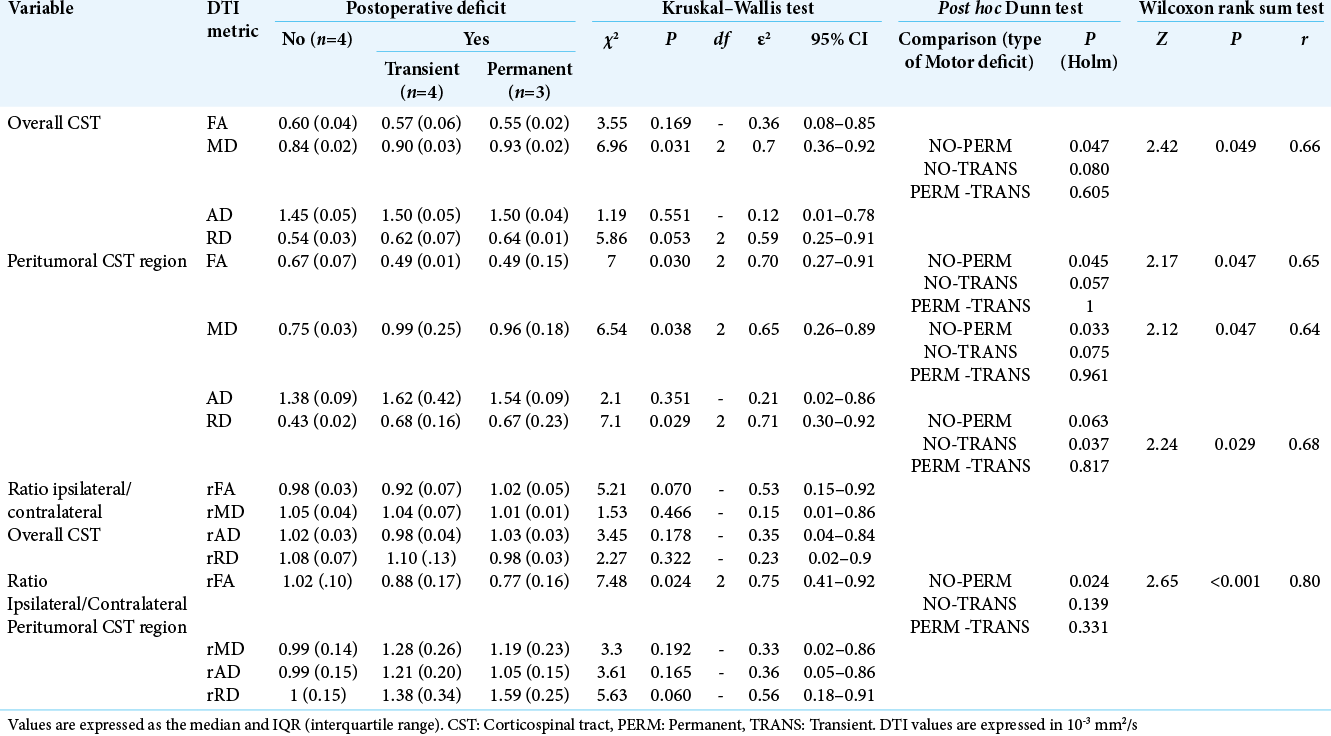
A normality test was applied to the sample using the Shapiro–Wilk test. Variables that were derived from the tensor had a non-normal distribution. The DTI values were compared with the presence of a preoperative deficit using the Wilcoxon–Mann–Whitney test, calculating the U statistic and the effect size with the r coefficient. The postsurgical motor status that was represented by the categories “no deficit,” “transient deficit,” and “permanent deficit” was compared with the tensor metrics using the Kruskal–Wallis test, using the epsilon squared (ε2) coefficient as a measure of the effect size. A post hoc study was also performed using the Holm correction and the Wilcoxon rank-sum test.
The medians of the tensor values were also analyzed according to the presence or absence of motor impairment, comparing the ipsilateral to the contralateral values in both pre- and post-operative MRI. Thus, we used the Wilcoxon signed-rank test. All statistical analyses were performed in R version 3.5.0 (R Foundation for Statistical Computing, Vienna, Austria).
RESULTS
During the study period, 53 patients with histologically confirmed glioma underwent surgical resection. Fourteen patients met the criteria of proximity to the CST. Three patients were excluded because of motor/premotor cortex invasion, postsurgical primary motor cortex infarction, or DTI performed at another center. Thus, 11 patients met the selection criteria and were included in the analysis.
Six (55.5%) patients were men and five (45.5%) patients were women. The mean age was 49.82 ± 14.34 years, the median presurgical Karnofsky performance scale (KPS) was 80 (20). Six were high-grade and five were low-grade gliomas. The mean initial volume was 43.32 ± 33.1 cm3. Four patients presented a presurgical motor deficit and the motor balance is summarized in [
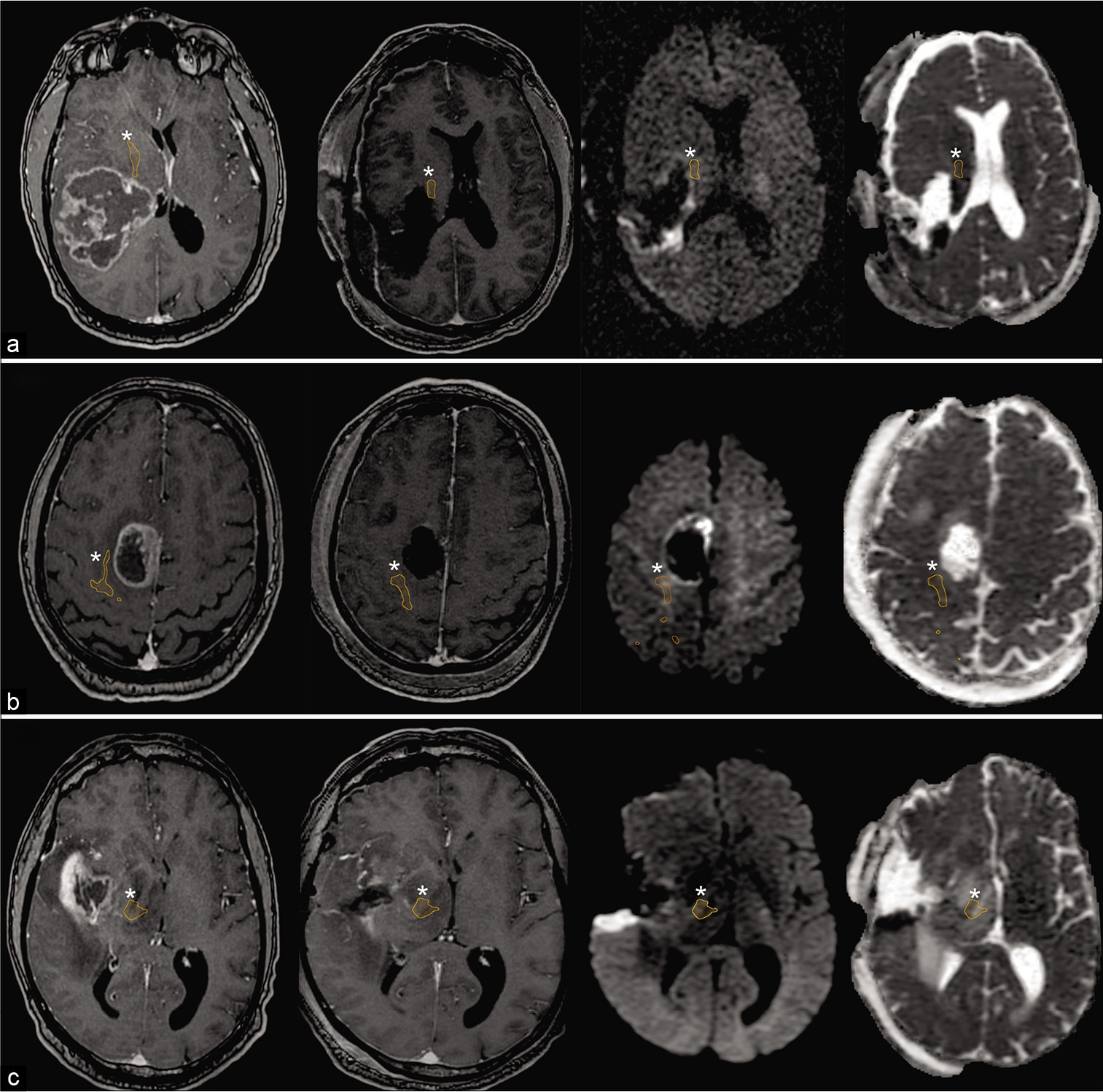
Figure 3:
Illustrative cases of permanent motor deficits. From left to right: preoperative T1-weighted postcontrast and postoperative images: T1-weighted postcontrast, diffusion-weighted imaging (DWI), and apparent diffusion coefficient (ADC) map. The location of the pyramidal tract that was determined by fiber tracking is also shown (white asterisk). (a) A 53-year-old man with a parieto-temporal glioblastoma; (b) a 65-year-old man with a right frontal glioblastoma; and (c) a 54-year-old man with a right temporo-insular glioblastoma.
The presence of a permanent motor deficit was not correlated with the clinical or radiological variables including initial tumor volume, extent of resection, and distance to the CST. Although the three patients with a permanent deficit corresponded to high-grade gliomas, this relationship did not reach statistical significance. [Supplementary
Quantitative analysis of the tensor metrics is summarized in [Supplementary
[Supplementary
The results of the postoperative DTI and motor outcome analyses are shown in [
Figure 4:
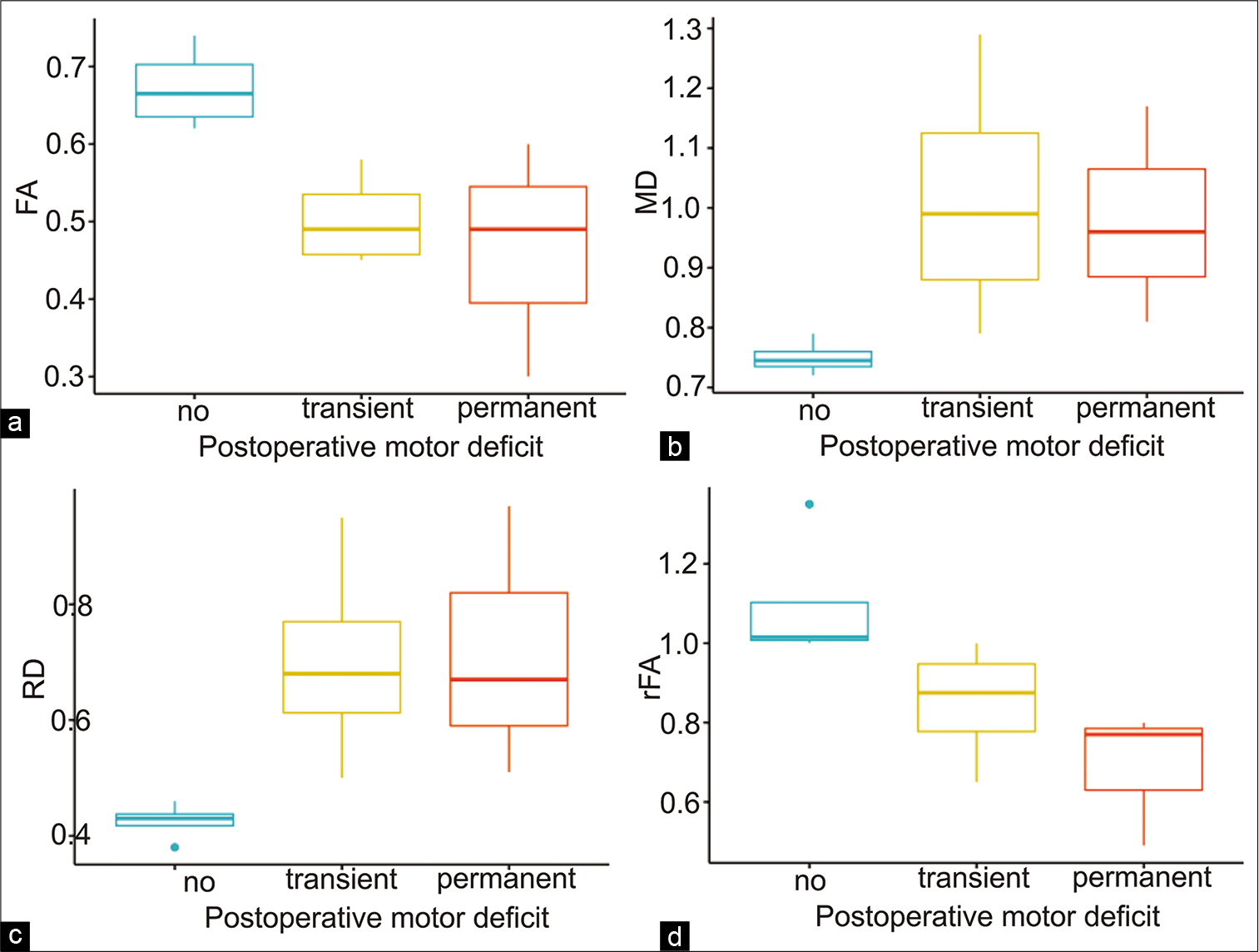
Box plots showing differences in the peritumoral diffusion tensor imaging-derived metrics according to the type of postoperative motor deficit. (a) Peritumoral fractional anisotropy (FA); (b) peritumoral mean diffusivity; (c) peritumoral radial diffusivity; and (d)p FA ratio (rFA; affected/unaffected).
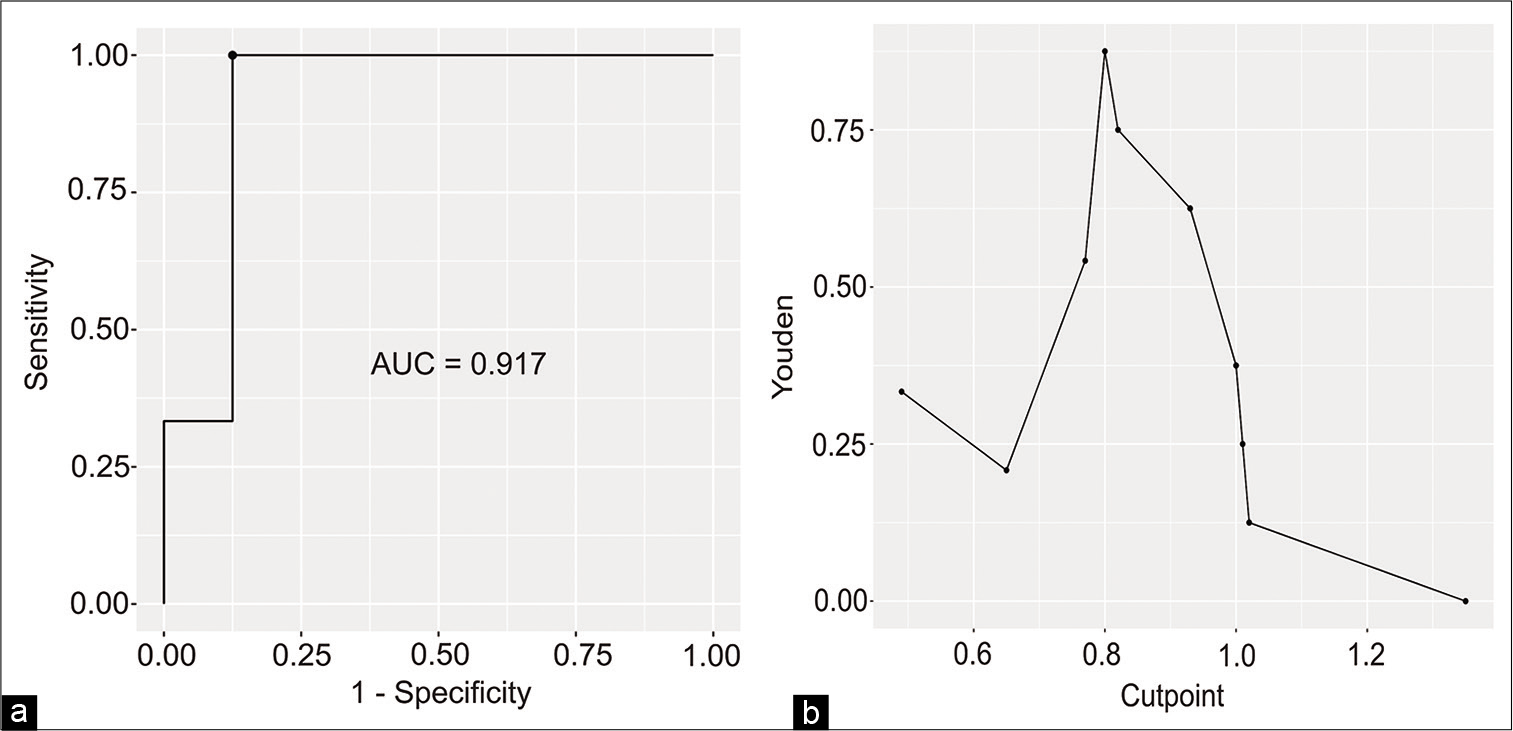
A cutoff based on the receiver operating characteristic curves was calculated, establishing a threshold value of 0.8 for the rFA, and thus, lower values are related to the presence of a permanent motor deficit in our series [Supplementary
DISCUSSION
In the present study, we implemented DTI quantitatively to elucidate the relationship between the clinical outcome and this radiological-derived information. Among the main findings of this investigation, patients who presented a postsurgical motor deficit had lower FA and higher MD and RD values compared to patients who did not show any deficit.
Among the strengths of our study, our results rely on DTI studies that were performed within the first 48 postoperative h. In addition, the analysis and image processing focus on the CST regions that are adjacent to the tumor and surgical cavity, and these areas are susceptible to postsurgical changes. Qualitative representation of the variable of interest (motor outcome) simplifies the analysis and its interpretation.
We established an objective definition of the tumor’s proximity to the CST based on previous publications.[
We emphasize the importance of standardizing MRI acquisition and postprocessing imaging, especially the time at which the postoperative control study is performed. Other studies that correlated MRI data with the motor outcome are based on preoperative studies[
The robustness of our methodology relies on the addition of a quantitative, comparative analysis based on data that were derived from pre and postoperative DTI. Most of previous studies had methodologies that often disregarded this important information. In a study by Laundre et al.,[
In a study by Gao et al.,[
In our series, the tensor metrics of the preoperative studies in which the entire CST is measured are similar for both the ipsilesional and contralateral healthy hemisphere. The values are similar to the series described by Cosottini et al.,[
The most important differences have been found when studying the CST regions that are adjacent to the tumor and the postsurgical cavity. We showed that in these areas, patients who presented with a postsurgical motor deficit had lower FA and higher values of MD and RD compared to patients who did not show any deficit. Doughty et al.[
For the ratio between peritumoral CST areas and the contralateral hemisphere, rFA was significantly lower in patients with permanent deficits. Through ROC curve analysis, the threshold we set in our series of 0.8 was related to a permanent postoperative motor deficit, and this threshold value was similar to that described in papers that assessed motor outcome in patients after a stroke,[
These regional variations of the CST can represent irreversible damage to the tract, which is also found in patients with transient deficits but with a different magnitude. Among the three patients with a permanent deficit in our series, all had a preoperative motor deficit, while the four patients with a new postoperative deficit presented peritumoral CST alterations. However, these values did not become significant for the group that did not present deficits. This suggests that alterations of the tensor metrics in this group may be related to surgical manipulation and displacement or decompression of the CST instead of structural damage, as would occur in patients with an irreversible deficit.
The study of early postsurgical DTI suggests that the presence of artifacts in the acquisition, the presence of blood, small areas of ischemia, or patient movements can make it difficult to reconstruct the pyramidal tract as well as the values that are derived from its analysis.
Our study has some limitations. These include a small study sample size; images acquired using a 1.5 T scanner, and the absence of long-term DTI controls that would allow us a longitudinal analysis of the tensor values and the motor outcomes.
Future studies should focus on validation of the present methodology and results with a larger prospective series. We believe that DTI can provide valuable prognostic information in glioma surgery near eloquent areas. Efforts should be directed toward the creation of predictive models based on DTI and clinical variables that allow identification of patients who have a high risk of permanent motor sequelae. However, if we can differentiate postsurgical alterations based on CST metrics, we can provide patients with transient deficits valuable information about its functional prognosis, potential recovery, and rehabilitation strategies. These image processing tools should evolve into user friendly software, and improvements in spatial resolution and tensor reconstruction techniques should help to overcome the current technical difficulties.
CONCLUSION
Quantitative DTI analysis that was performed in the immediate postsurgical period revealed changes in the CST area values near the tumor, which were consistently related to the motor outcome in our series. Our results showed that the peritumor-to-healthy FA ratio in the CST allows discrimination between patients who will and will not develop a permanent postoperative deficit.