- Department of Neurosurgery, Hospital Nacional Guillermo Almenara, Lima, Peru,
- Department of Ophthalmology, Hospital Nacional Guillermo Almenara, Lima, Peru,
- Department of Neurosurgery, School of Biomedical Sciences, Universidad Austral, Buenos Aires, Argentina,
- Department of Neurosurgery, Mater Dei Hospital, Bari, Italy
- Department of Neurosurgery, Azienda Ospedaliera Universitaria Pisana, University of Pisa, Pisa, Italy.
Correspondence Address:
Nicola Montemurro, Department of Neurosurgery, Azienda Ospedaliera Universitaria Pisana, University of Pisa, Pisa, Italy.
DOI:10.25259/SNI_571_2021
Copyright: © 2021 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Jafeth Lizana1, Carlos M. Dulanto Reinoso2, Nelida Aliaga3, Walter Marani4, Nicola Montemurro5. Bilateral central retinal artery occlusion: An exceptional complication after frontal parasagittal meningioma resection. 09-Aug-2021;12:397
How to cite this URL: Jafeth Lizana1, Carlos M. Dulanto Reinoso2, Nelida Aliaga3, Walter Marani4, Nicola Montemurro5. Bilateral central retinal artery occlusion: An exceptional complication after frontal parasagittal meningioma resection. 09-Aug-2021;12:397. Available from: https://surgicalneurologyint.com/surgicalint-articles/11031/
Abstract
Background: Central retinal artery occlusion (CRAO) is a rare acute disease associated with great morbidity. It is reported as a complication of surgical procedures, but rarely associated with brain surgery and no reports before due to parasagittal meningioma resection.
Case Description: We present the case of a 41-year-old female who underwent surgery for a parasagittal meningioma and developed a bilateral CRAO as an acute postoperative complication. Most common causes, such as cardiac embolism, carotid pathology and coagulation problems, were discussed and all clinical and neuroradiological exams performed were reported.
Conclusion: Bilateral CRAO as results of brain surgery is extremely rare; however, if it occurs, it should be early recognized and treated to minimize its high morbidity.
Keywords: Case report, Ophthalmic artery, Postoperative management, Retinal artery occlusion, Surgical complication
INTRODUCTION
Non-arteritic central retinal artery occlusion (CRAO) has an incidence of 0.85/100000/year. It has often been related to different surgical procedures; however, rarely to brain surgeries.[
CASE PRESENTATION
Here, we reported a 41-year-old female with a medical history of obesity and diabetes type 2, who underwent surgical resection of a frontal parasagittal with no intraoperative surgical and anesthetic complications and developed complete blindness on awakening as a complication. Postoperative ophthalmological examination revealed bilateral mydriasis and non-reactive to light in both eyes, although bilateral mydriasis can occur after general anesthesia.[
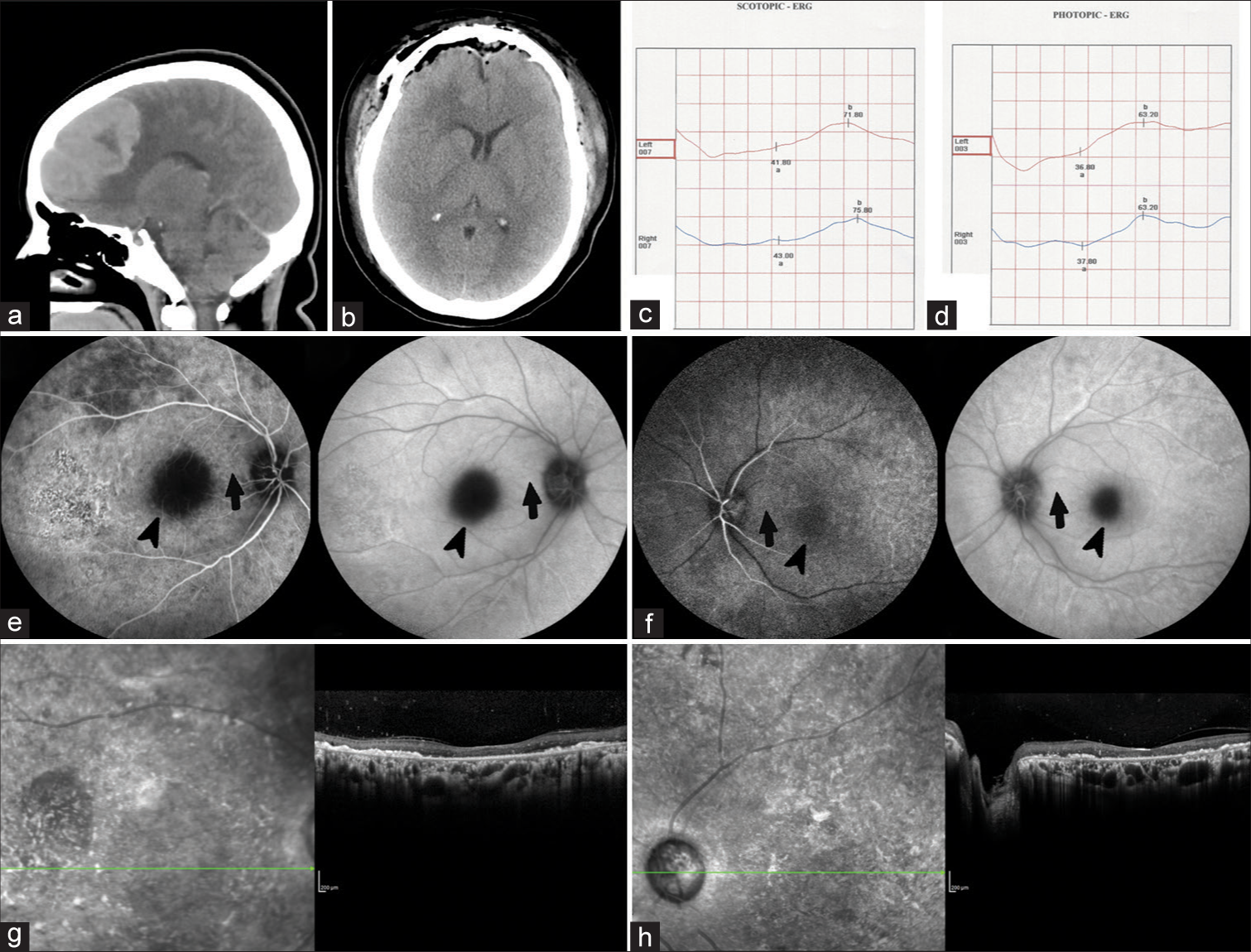
Figure 1:
Preoperative (a) and postoperative (b) CT head scans show the site of a parasagittal meningioma and its subsequent complete resection. Bilateral electroretinogram in scotopic (c) and phototopic (d) protocols, which show prolonged latencies, decreased amplitudes and distortion in a and b waves, as signs of moderate to severe diffuse retinopathy in both eyes. Right (e) and left (f) eyes retinal fluorescein biography shows delay in the arterial phase as well as in the optic nerve head hypofluorescence in late phases. The macula is marked with black arrows while the absence of cilioretinal arteries with black arrowheads. Spectral domain optical coherence tomography (at 3 months after surgery) of the right eye (g) and left eye (h) show chronic changes caused by CRAO.
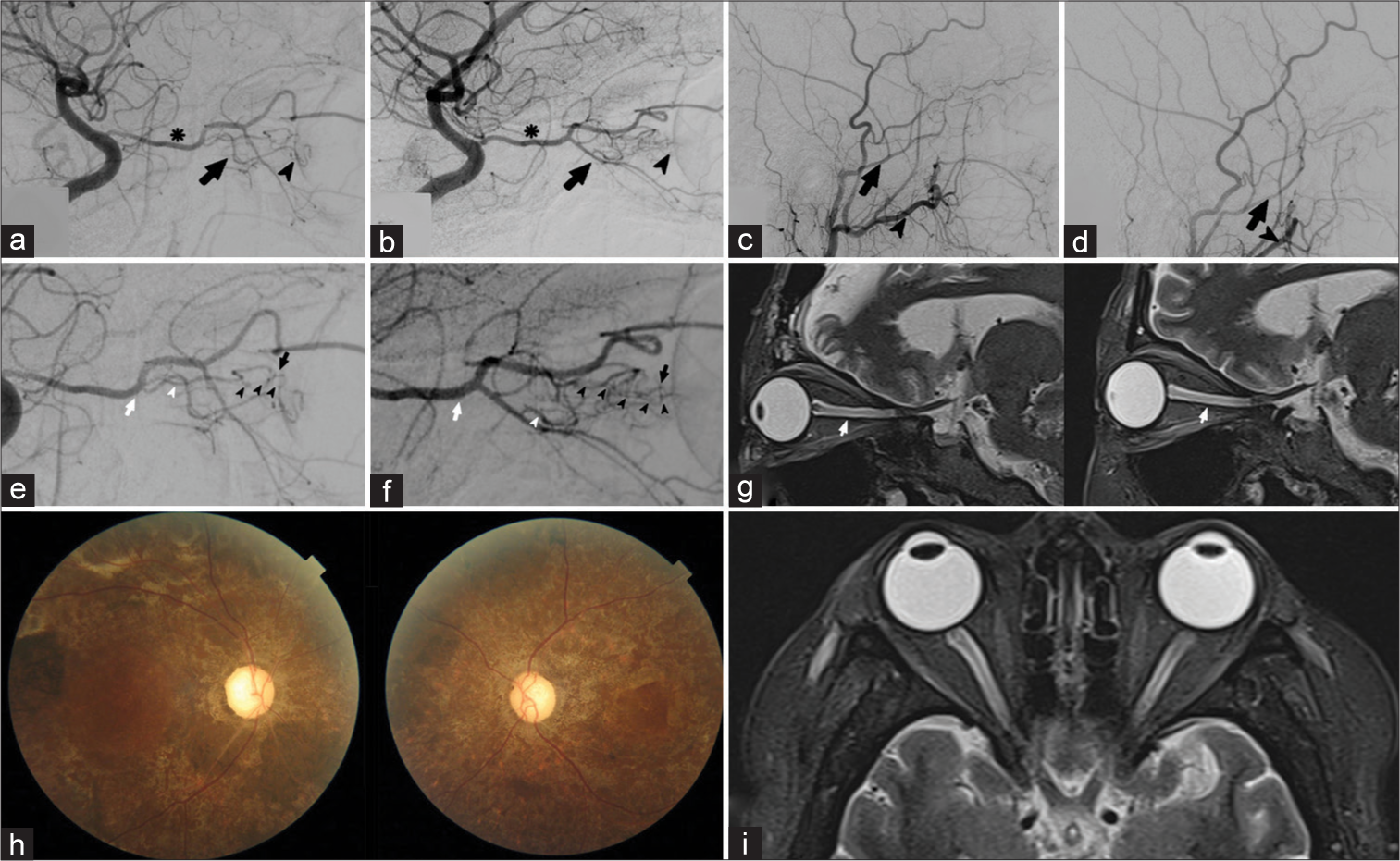
Figure 2:
Postoperative right (a) and left (b) internal carotid digital subtraction angiography (DSA) shows ophthalmic artery (black asterisks) on the same side, the ciliary arteries (black arrows) and the attenuated choroidal blush of the eye (black arrowheads). Right (c) and left (d) external carotids DSA shows no choroidal blush nor the presence of some dangerous anastomosis of the ophthalmic artery with the middle meningeal artery (black arrows) or with the internal maxillary (black arrowheads). Postoperative DSA (e) shows the right ophthalmic artery (white arrow), the origin of the central retinal artery (white arrowhead), and the delay in filling in phase of the distal portion of the central retinal artery (black arrowheads), which is partially supplied by the vascular ring of Zinn (black arrow). (f) shows left ophthalmic artery (white arrow), the posterior ciliary artery (white arrowhead), in addition to a delay in the filling of the central retinal artery (black arrow heads) and partial replacement through the vascular ring of Zinn (black arrow). Sagittal (g) T2-weighted brain MRI shows hyperintensity of both optic nerves in its intra-orbital segment (white arrowhead), suggestive of bilateral optic nerve infarction. Right and left fundi (h) at 3 months follow-up after surgery. Axial (i) T2-weighted brain MRI shows hyperintensity of both optic nerves.
DISCUSSION
CRAO is an ophthalmological emergency due to the short time retina tolerates ischemia (12–15 min) and this is due to the fact that the oxygen consumption rate (13 ml/100 g/min) of the retina is even greater than the brain (3.8 ml/100 g/min).[
The supply of the internal and external retina is the central retinal artery and the posterior short ciliary arteries, respectively.[
CONCLUSION
The present image report showed a rare case of bilateral CRAO as an acute complication of cranial surgery, which is extremely unusual according to the bibliography, reporting all clinical and neuroradiological exams performed to understand what the cause is, even if it remains unknown.
Declaration of patient consent
The author certifies that he has obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Berg KT, Harrison AR, Lee MS. Perioperative visual loss in ocular and nonocular surgery. Clin Ophthalmol. 2010. 4: 531-46
2. Canseco JA, Schroeder GD, Patel PD, Grasso G, Chang M, Kandziora F. Regional and experiential differences in surgeon preference for the treatment of cervical facet injuries: A case study survey with the AO spine cervical classification validation group. Eur Spine J. 2021. 30: 517-23
3. Cho SC, Jung C, Lee JY, Kim SJ, Park KH, Woo SJ. Retinal artery occlusion after intravascular procedures: Case series and literature review. Retina. 2019. 39: 766-78
4. Costello F, Gilberg S, Karsh J, Burns B, Leonard B. Bilateral simultaneous central retinal artery occlusions in Wegener granulomatosis. J Neuroophthalmol. 2005. 25: 29-32
5. Cumurcu T, Doganay S, Demirel S, Cankaya C. Traumatic optic neuropathy and central retinal artery occlusion following blunt ocular trauma. J Clin Med Res. 2011. 3: 55-7
6. Goodwin PL, Vaphiades MS, Johnson AP, Stroud CE. Bilateral central retinal artery occlusion associated with moyamoya syndrome in a sickle cell disease patient. Neuroophthalmology. 2008. 32: 21-6
7. Hayreh SS, Podhajsky PA, Zimmerman MB. Retinal artery occlusion: Associated systemic and ophthalmic abnormalities. Ophthalmology. 2009. 116: 1928-36
8. Hayreh SS, Zimmerman MB, Kimura A, Sanon A. Central retinal artery occlusion. Retinal survival time. Exp Eye Res. 2004. 78: 723-36
9. Hua L, Patel K, Corbett JJ. Bilateral central retinal artery occlusion in a patient with systemic lupus erythematosus. J Stroke Cerebrovasc Dis. 2015. 24: e139-41
10. Kitthaweesin K, Moontawee K, Thanathanee O. Sudden visual loss and total ophthalmoplegia after brain surgery. Neuroophthalmology. 2009. 33: 59-61
11. Michalinos A, Zogana S, Kotsiomitis E, Mazarakis A, Troupis T. Anatomy of the ophthalmic artery: A review concerning its modern surgical and clinical applications. Anat Res Int. 2015. 2015: 591961
12. Montemurro N, Perrini P, Rapone B. Clinical risk and overall survival in patients with diabetes mellitus, hyperglycemia and glioblastoma multiforme. A review of the current literature. Int J Environ Res Public Health. 2020. 17: 8501
13. Montemurro N, Santoro G, Marani W, Petrella G. Posttraumatic synchronous double acute epidural hematomas: Two craniotomies, single skin incision. Surg Neurol Int. 2020. 11: 435
14. Montemurro N. Intracranial hemorrhage and COVID-19, but please do not forget “old diseases” and elective surgery. Brain Behav Immun. 2021. 92: 207-8
15. Murthy RK, Perez L, Priluck JC, Grover S, Chalam KV. Acute, bilateral, concurrent central retinal artery occlusion in sickle cell disease after use of tadalafil (Cialis). JAMA Ophthalmol. 2013. 131: 1471-3
16. Myers MA, Hamilton SR, Bogosian AJ, Smith CH, Wagner TA. Visual loss as a complication of spine surgery: A review of 37 cases. Spine. 1997. 22: 1325-9
17. Park KH, Kim YK, Woo SJ, Kang SW, Lee WK, Choi KS. Iatrogenic occlusion of the ophthalmic artery after cosmetic facial filler injections: A national survey by the Korean retina society. JAMA Ophthalmol. 2014. 132: 714-23
18. Perrini P, Pieri F, Montemurro N, Tiezzi G, Parenti GF. Thoracic extradural haematoma after epidural anaesthesia. Neurol Sci. 2010. 31: 87-8
19. Ripart J, Desmonts JM. Loss of vision after anaesthesia for nonophthalmic surgery: A persistent problem. Eur J Anaesthesiol. 2009. 26: 524-6
20. Tobalem S, Schutz JS, Chronopoulos A. Central retinal artery occlusion-rethinking retinal survival time. BMC Ophthalmol. 2018. 18: 1-6
21. Vukkadala T, Balaji A, Azad SV, Kumar V. Bilateral central retinal artery occlusion in a patient with diagnosed Takayasu’s arteritis. Indian J Ophthalmol Case Rep. 2021. 1: 71
22. Weng W, Gao W, Zhao X, Mao Q, Gao G, Feng F. Bilateral mydriasis secondary to propofol administration in a patient with hypertensive cerebellar hemorrhage. J Neurosurg Anesthesiol. 2018. 30: 278-9
23. Xia T, Zarbin MA, Bhagat N. Retinal artery occlusions in young adults; a neuro-surgical complication. Invest Ophthalmol Vis Sci. 2018. 59: 4278-8