- Department of Neurosurgery, Tanta University, Tanta, Gharbia, Egypt.
DOI:10.25259/SNI_610_2020
Copyright: © 2020 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Ahmed Abdelaziz Elsharkawy, Hytham Elatrozy. Endoscopic antegrade aqueductoplasty and stenting with panventricular catheter in management of trapped fourth ventricle in patients with inadequately functioning supratentorial shunt. 18-Nov-2020;11:393
How to cite this URL: Ahmed Abdelaziz Elsharkawy, Hytham Elatrozy. Endoscopic antegrade aqueductoplasty and stenting with panventricular catheter in management of trapped fourth ventricle in patients with inadequately functioning supratentorial shunt. 18-Nov-2020;11:393. Available from: https://surgicalneurologyint.com/?post_type=surgicalint_articles&p=10396
Abstract
Background: Trapped fourth ventricle (TFV) usually develops as a complication of supratentorial ventricular CSF shunting, especially when hydrocephalus is caused by intraventricular hemorrhage and/or infection. This study aimed to assess the feasibility of endoscopic aqueduct stenting using a single refashioned shunt tube to treat cases presenting with both TFV and shunt malfunction.
Methods: We retrospectively collected and analyzed data from patients presenting with TFV and supratentorial shunt malfunction who underwent endoscopic aqueduct stenting using a refashioned shunt tube. All cases were treated at our institution between January 2010 and July 2019. The surgical technique is described.
Results: Eighteen patients were enrolled in our study. There were ten males and eight females. The mean age was 11.2 years (range = 1–33 years). Headache, nausea, and vomiting were the most common clinical presentations. The mean duration of follow-up was 22.1 months (range = 6–60 months). All cases showed clinical and radiological improvement after surgery.
Conclusion: Endoscopic antegrade aqueductoplasty and stenting with the refashioned panventricular shunt catheter are an adequate treatment option for both TFV and supratentorial shunt malfuncion.
Keywords: Aqueductal stenting, Endoscopic aqueductoplasty, Refashioned shunt tube, Shunt malfunction, Trapped fourth ventricle
INTRODUCTION
Trapped fourth ventricle (TFV) defines a pathological condition observed when the fourth ventricle balloons with CSF after obstruction of its inlet (the aqueduct) and exit (foramina of Magendie and Luschka). The condition usually develops as a complication of supratentorial ventricular CSF shunting, especially when hydrocephalus is caused by intraventricular hemorrhage and/or infection. Ependymal inflammation develops and ultimately leads to scarring and obstruction of tight CSF routes and isolation of ventricular systems. The dilated fourth ventricle acts as a posterior fossa mass lesion compressing on the brainstem and cerebellum. Failure of adequate management of TFV can lead to severe neurological morbidity and/or mortality.[
The therapeutic approaches for TFV are multiple and include CSF diversion (transcerebellar fourth ventricular shunting), open surgical fenestration of the fourth ventricles, and endoscopic procedures (aqueductoplasty and/or stenting). TFV patients with complete clinical and radiological stability can be managed conservatively.[
MATERIALS AND METHODS
This is a retrospective study comprising 18 consecutive patients, who underwent endoscopic surgery, at our institute between January 2010 and July 2019, for management of symptomatic TFV and supratentorial shunt malfunction. A review of radiographic studies showed that preoperative good quality MRI had been required for all cases before the procedure. This aimed to include only patients with MRI evidence of short segment aqueductal obstruction and some clear anatomic landmarks in the posterior third ventricle to help endoscopic navigation as pineal recess or posterior commissure. All cases underwent endoscopic aqueductoplasty and stenting with the refashioned shunt tube. No cases were excluded due to procedural failure. Demographic, clinical, and radiological data were gathered from the hospital database. The ethical committee at our institute approved this study.
Before surgery, CSF samples were obtained through the shunt reservoir to exclude any active shunt hardware infection. This was observed in only one patient (Case 12) in whom externalization of the shunt was done until infection was cleared out, followed by surgery using antibiotic impregnated shunt catheter as a stent. The required length of the ventricular catheter and the location of the extra side ports were estimated through preoperative sagittal MRI views. The ventricular catheter was refashioned accordingly, during surgery, to reach down to the most distal part of the cavity of the fourth ventricle, and to have extra side ports inside the third and lateral ventricles as well.
Surgical procedure
Under general anesthesia, the patient was placed in a supine position. The head was placed in horseshoe-shaped headrest to allow changing head tilt and rotation during different stages of surgery. One precoronal burr hole was placed, behind the hairline and anterior to the coronal suture, based on the inclination of the aqueduct obtained from preoperative sagittal MRI views. After removal of the old shunt hardware a freehand insertion of the endoscope is done, aiming the floor of the third ventricle to perform endoscopic third ventriculostomy (ETV). The trajectory for ETV was patent in seven patients, including wide foramen of Monro and cavity of the third ventricle. In the other 11 patients, ETV was technically undoable. The endoscope was then moved backward to access the aqueduct. A 3-French Fogarty Balloon catheter was then passed through the aqueduct with very little inflation to violate any covering membrane and perform aqueductoplasty.
The new refashioned ventricular catheter, with stylet inside, is passed alongside the endoscope sheath to reach the aqueduct. After few millimeters’ advancement of the catheter through the aqueduct, the stylet is moved incrementally upward letting the catheter be guided in further advancement by the lumen of the aqueduct and dilated cavity of the fourth ventricle. When the calculated length of the catheter is reached, the endoscope is removed, and proximal shunt catheter is then connected to a reservoir, and new distal abdominal catheter is tunneled down to the peritoneal cavity which was ligated in the seven patients in whom ETV was performed aiming at later on the removal of the distal catheters if ETV had succeeded. Skin is meticulously closed in layers.
The outcome was assessed clinically and radiologically. Follow-up CT scans were scheduled at day 1 and day 30 after surgery. Follow-up MRI was planned at 3 and 6 months postoperatively. Other follow-up items included documenting the need for any shunt revision or any neurological complication. Illustrative cases are shown in [
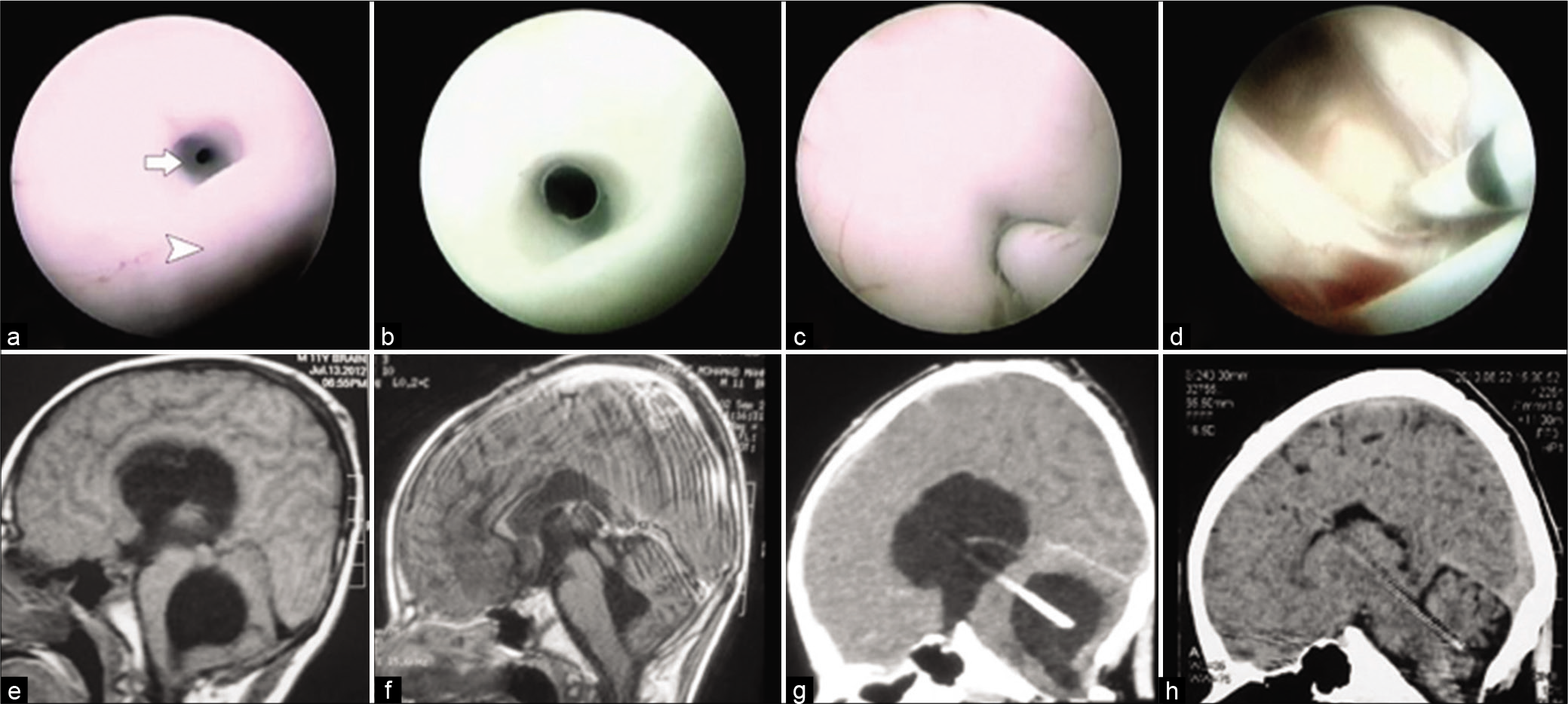
Figure 1:
(a) Endoscopic view within the posterior third ventricle demonstrating the posterior commissure (arrow head) and membrane covering the aqueduct with tiny opening (arrow). (b) Endoscopic view within the posterior third ventricle after balloon aqueductoplasty. (c) Aqueduct stenting using shunt tube. (d) Endoscopic view through the lateral ventricle before withdrawal of the endoscope. (e) Pre-operative sagittal T1 MRI showing ballooning of ventricular system. (f) Post-operative sagittal T1 MR image showing marked decline in all ventricular system size after stenting. (g) Early post-operative sagittal reconstructed CT image. (h) Late post-operative sagittal reconstructed CT image showing full resolution of all ventriculomegaly.
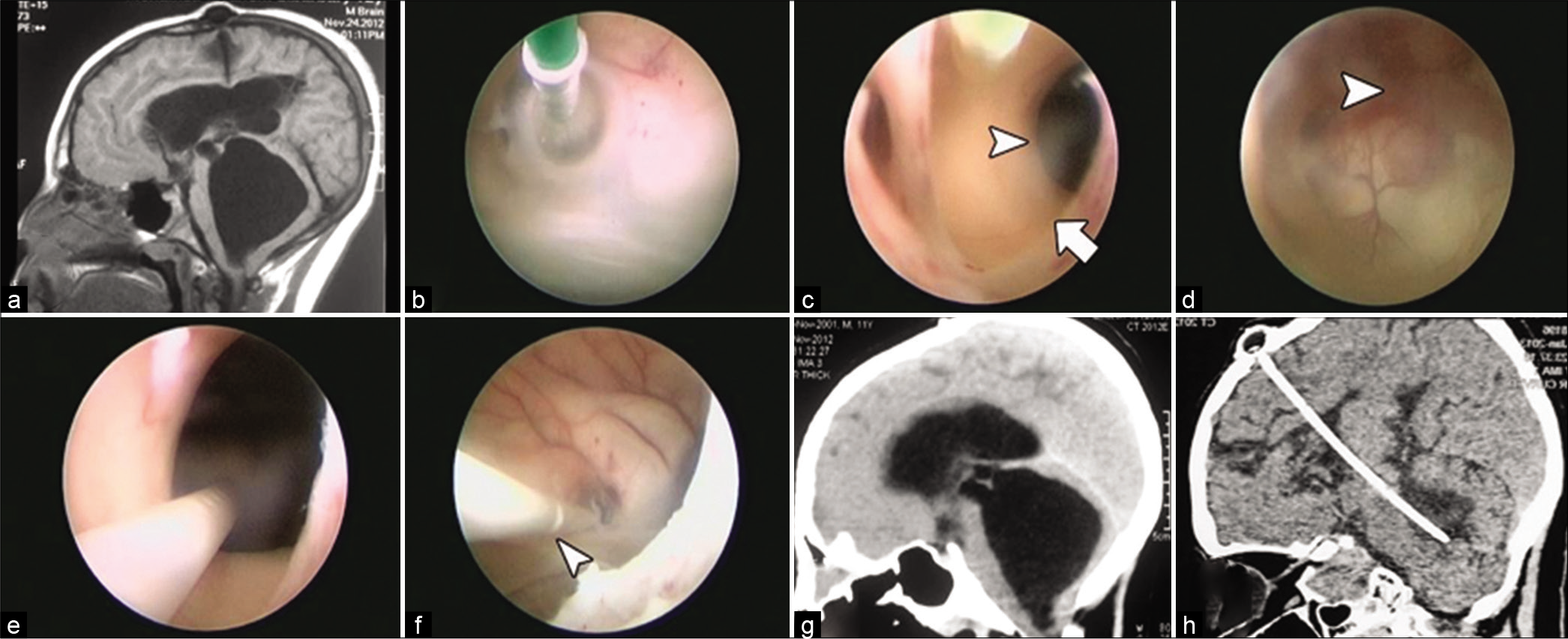
Figure 2:
(a) Preoperative sagittal T1 MR image showing markedly dilated TFV with shunt tube inserted in the lateral ventricle and intraventricular adhesions circumventing a dilated aqueduct opening and occluding the cavity of the third ventricle. (b) Endoscopic view showing balloon foraminoplasty. (c) Endoscopic view through restored foramen of Monro (arrow) showing the dilated opening of the aqueduct (arrow head). (d) Endoscopic view through the dilated aqueduct showing the fourth ventricular cavity with its choroid plexus (arrow head). (e) Endoscopic view showing queduct stenting using shunt tube. (f) Endoscopic view through the lateral ventricle before withdrawal of the endoscope with refashioned shunt tube and extra-ports (one of them is pointed out by arrow head). (g) Pre-operative sagittal reconstructed CT image showing ballooning of third and fourth ventricles. (h) Late post-operative sagittal reconstructed CT image showing full resolution of all ventriculomegaly.
RESULTS
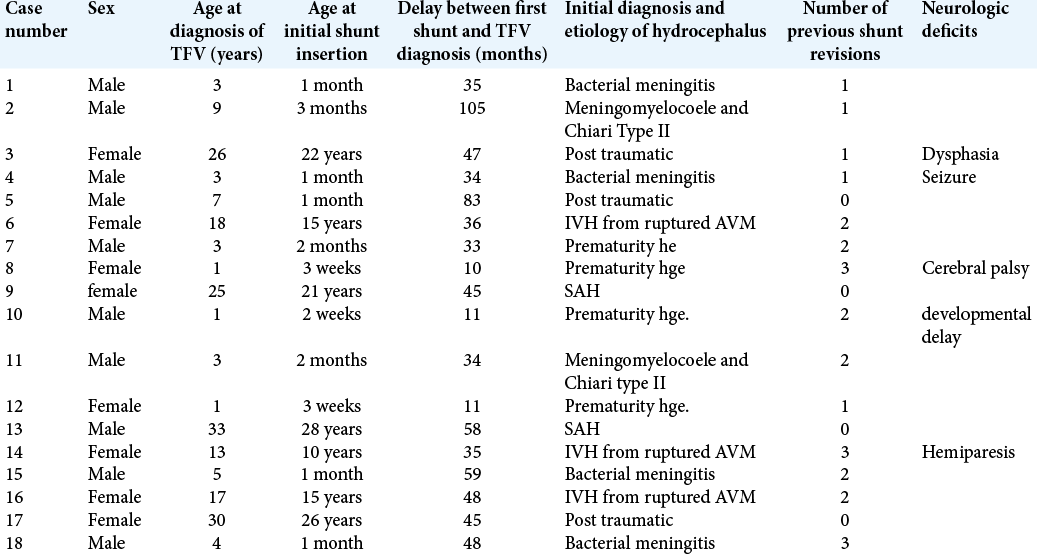
Eighteen patients of symptomatic TFV and supratentorial VP shunt malfunction were included in this study. There were ten males and eight females. The mean age of the patients at the time of surgery was 11.2 years (range = 1–33 years). The mean duration of follow-up was 22.1 months (range = 6–60 months). Most patients (14/18) had undergone previous shunt revisions before the diagnosis of TFV. The average delay between initial lateral ventricle shunt placement and the diagnosis of TFV was 43.2 months (range = 10–105 months) [
Preoperative clinical manifestations included headache (15/18), nausea and vomiting (15/18), gait ataxia (10/18), truncal ataxia (3/18), cranial nerve palsy (4/18), diplopia (4/18), visual obscuration (3/18), and bulbar palsy (4/18), including dysphagia, dysphonia, and choking on oral fluids. Aqueduct catheter placement was successful in all cases. ETV failed in all the seven cases where shunt ligation had to be removed within 48 h after surgery. Hydrocephalus and TFV were controlled by a single shunt in all cases [
After surgery, there was radiological evidence of the diminution of the size of the TFV and lateral ventricles in all cases. Rapid resolution of clinical signs and symptoms of raised intracranial pressure and gradual amelioration of signs and symptoms of posterior fossa mass lesions occurred in all cases after surgery. Two patients (Cases seven and 18) developed transient oculomotor palsy which spontaneously resolved in 4 weeks following surgery. One patient (Case 4) developed asymptomatic posterior fossa subdural hygroma. Ordinary shunt tubes were used in seventeen patients, and an antibiotic impregnated shunt tube was used in one patient (Case 12).
DISCUSSION
TFV pathogenesis entails obstruction of the CSF pathway at the level of the foramina of Magendie and Luschka and the cerebral aqueduct. It is almost exclusively seen in lateral ventricle shunted patients, especially with hydrocephalus following infection and/or hemorrhage.[
In the year 1978, Zimmerman et al.[
The transcerebellar fourth ventricular shunting then became the most frequently used maneuver to treat TFV. However, it was associated with a high rate of obstruction and the need for repeated revisions.[
In 1920, Walter Dandy performed the first retrograde aqueductoplasty by freehand insertion of a rubber catheter curved up and guided by metal stylet into the aqueduct through a suboccipital open naked eye approach retracting the cerebellar lobes utilizing a nasal speculum.[
An ideal management strategy for multilocular hydrocephalus should equalize pressure in different ventricular compartments and drain all the ventricular system. Different endoscopic techniques have been described to assist frontal insertion of a refashioned panventricular catheter traversing the lateral and third ventricles then passing into the fourth ventricle through the aqueduct after aqueductoplasty. Refashioning the ventricular catheter by adding extra holes allows the connection and drainage of all the ventricular compartments.[
In this report, we present our experience with the management of 18 patients presenting with symptomatic TFV and lateral ventricle shunt malfunction. Our procedure entails the endoscope-assisted insertion of a refashioned panventricular shunt catheter starting from a frontal entry point, passing into the lateral ventricle then through the foramen of Monro into the third ventricle, and ending into the fourth ventricle through the aqueduct of Sylvius. We used Lotta® ventriculo-scope system with Hopkins® wide angle 6-degree telescope, an outer diameter 6.1 mm, length 18 cm, and working channel diameter 2.9 mm to guide intraventricular navigation, Fogarty catheter (3-French) for aqueductoplasty and a burr hole type reservoir to fix the panventricular catheter at the frontal insertion site.
The development of symptoms and signs of cerebellar and/or brain stem malfunction in our patients, was mostly insidious, while symptoms and signs of raised intracranial pressure due to shunt malfunction developed more acutely and were more frequently the drive to seek medical advice. The etiology of shunt malfunction was mostly (78%) proximal catheter failure due to bugging of the tiny holes. The delay between initial shunt insertion and the diagnosis of TFV was variable (mean= 43 months, range = 10–105 months). After surgery, the resolution of symptoms and signs of raised intracranial pressure was more rapid and complete, while symptoms of cerebellar and/or brainstem compression resolved more gradually and usually incompletely. All cases showed radiological evidence of diminution of supratentorial ventriculomegaly and the size of the fourth ventricle. During the follow-up period (mean= 22 months, range= 6–60 months) of our patients, no case developed shunt malfunction or infection.
Comparable endoscopic maneuvers to manage TFV have been described in earlier publications with some variations in technical steps.[
Our technique to perform the extra holes is to advance the stylet inside the tube till the point desired for making an extra hole, then the tube is bent across the stylet tip. Micro scissors are used to make a small cut at the flexion knee against the stylet tip (micro scissors blades are kept parallel to the long access of the tube so that any unintended extension of the hole occurs along the tube and not across it to avoid weakening the tube). To avoid the ingrowth of choroid plexus, measurements were taken to place the extra-holes along the segment of the tube in the frontal horn of the lateral ventricle away from the foramen of Monro and along the tube segment in the lower third ventricle close to the aqueduct.
The two main factors which rendered ETV undoable in eleven of our eighteen patients were obscuration of anatomic landmarks at the floor of the anterior third ventricle as a sequelae of inflammatory and/or hemorrhagic pathology and the narrow space in front of the basilar artery due to anterior brain stem shifting by the large ballooned TFV. Moreover, the location of our single burr hole was in favor of working in the posterior third ventricle rather than the anterior third ventricle.
Our provisional plan of management, if proximal shunt revision is needed, is to perform an endoscopic inspection of the panventricular tube. We believe that the extraction of this panventricular tube should be done under endoscopic visual control after releasing any adhesions using bipolar cautery and/or scissors.
There are some limitations to this study, including retrospective nature, small number of patients (n = 18), and single-institution experience. However, our results may be of interest. This is the first publication of a series of patients presenting with supratentorial shunt malfunction and TFV successfully managed with endoscopic-assisted antegrade aqueductoplasty and stenting with panventricular refashioned shunt catheter. Failure of shunt independence in all seven cases in whom ETV was done, which may indicate coincidence of subarachnoid scarring with ependymal scarring in these patients. Long-term patency of shunt system with no need for revision may be related to refashioning of the catheter with extra wider pores, exclusion of active CSF infection before surgery, and changing whole shunt system hardware. Lack of postoperative complications in the one patient (Case 12) treated with the antibiotic impregnated shunt tube suggests the safety of chemically treated catheter although being in intimate contact with parts of the brainstem.
CONCLUSION
Endoscopic antegrade aqueductoplasty and stenting with the refashioned panventricular shunt catheter are a feasible minimally invasive treatment option for the management of TFV in patients with inadequately functioning supratentorial shunts using one shunt tube.
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Ang BT, Steinbok P, Cochrane DD. Etiological differences between the isolated lateral ventricle and the isolated fourth ventricle. Childs Nerv Syst. 2006. 22: 1080-5
2. Cinalli G, Maixner WJ, Sainte-Rose C.editors. Pediatric Hydrocephalus. Milan: Springer; 2012. p.
3. Cinalli G, Spennato P, Savarese L, Ruggiero C, Aliberti F, Cuomo L. Endoscopic aqueductoplasty and placement of a stent in the cerebral aqueduct in the management of isolated fourth ventricle in children. J Neurosurg. 2006. 104: 21-7
4. DeFeo D, Foltz EL, Hamilton AE. Double compartment hydrocephalus in a patient with cysticercosis meningitis. Surg Neurol. 1975. 4: 247-51
5. Dezena RA.editors. Endoscopic Third Ventriculostomy: Classic Concepts and a State-of-the-Art Guide. Berlin: Springer International Publishing; 2019. p.
6. Eder HG, Leber KA, Gruber W. Complications after shunting isolated IV ventricles. Childs Nerv Syst. 1997. 13: 13-6
7. Ersahin Y. Endoscopic aqueductoplasty. Childs Nerv Syst. 2007. 23: 143-50
8. Fritsch MJ, Kienke S, Manwaring KH, Mehdorn HM. Endoscopic aqueductoplasty and interventriculostomy for the treatment of isolated fourth ventricle in children. Neurosurgery. 2004. 55: 372-7
9. Fritsch MJ, Kienke S, Mehdorn HM. Endoscopic aqueductoplasty: Stent or not to stent?. Childs Nerv Syst. 2004. 20: 137-42
10. Fritsch MJ, Schroeder HW. Endoscopic aqueductoplasty and stenting. World Neurosurg. 2013. 79: S20e15-28
11. Gallo P, Szathmari A, Simon E, Ricci-Franchi AC, Rousselle C, Hermier M. The endoscopic trans-fourth ventricle aqueductoplasty and stent placement for the treatment of trapped fourth ventricle: Long-term results in a series of 18 consecutive patients. Neurol India. 2012. 60: 271-7
12. Harter DH. Management strategies for treatment of the trapped fourth ventricle. Childs Nerv Syst. 2004. 20: 710-6
13. Hawkins JC, Hoffman HJ, Humphreys RP. Isolated fourth ventricle as a complication of ventricular shunting, Report of three cases. J Neurosurg. 1978. 49: 910-3
14. Lee M, Leahu D, Weiner HL, Abbott R, Wisoff JH, Epstein FJ. Complications of fourth-ventricular shunts. Pediatr Neurosurg. 1995. 22: 309-13
15. Longatti P, Marton E, Magrini S. The marionette technique for treatment of isolated fourth ventricle: Technical note. J Neurosurg Pediatr. 2013. 12: 339-43
16. Miki T, Nakajima N, Wada J, Haraoka J. Indications for neuroendoscopic aqueductoplasty without stenting for obstructive hydrocephalus due to aqueductal stenosis. Minim Invasive Neurosurg. 2005. 48: 136-41
17. Oi S, Matsumoto S. Pathophysiology of aqueductal obstruction in isolated IV ventricle after shunting. Childs Nerv Syst. 1986. 2: 282-6
18. Pomeraniec IJ, Ksendzovsky A, Ellis S, Roberts SE, Jane JA Jr. Frequency and long-term follow-up of trapped fourth ventricle following neonatal posthemorrhagic hydrocephalus. J Neurosurg Pediatr. 2016. 17: 552-7
19. Rajshekhar V. Endoscopic management of trapped fourth ventricle using the posterior fossa route. Neurol India. 2012. 60: 269-70
20. Sagan LM, Kojder I, Poncyljusz W. Endoscopic aqueductal stent placement for the treatment of a trapped fourth ventricle. J Neurosurg. 2006. 105: 275-80
21. Shin M, Morita A, Asano S, Ueki K, Kirino T. Neuroendoscopic aqueductal stent placement procedure for isolated fourth ventricle after ventricular shunt placement. Case report. J Neurosurg. 2000. 92: 1036-9
22. Spennato P, O’Brien DF, Fraher JP, Mallucci CL. Bilateral abducent and facial nerve palsies following fourth ventricle shunting: Two case reports. Childs Nerv Syst. 2005. 21: 309-16
23. Torres-Corzo J, Vecchia RR, Rangel-Castilla L. Trapped fourth ventricle treated with shunt placement in the fourth ventricle by direct visualization with flexible neuroendoscope. Minim Invasive Neurosurg. 2004. 47: 86-9
24. Tyagi G, Singh P, Bhat DI, Shukla D, Pruthi N, Devi BI. Trapped fourth ventricle-treatment options and the role of open posterior fenestration in the surgical management. Acta Neurochir (Wien). 2020. 162: 2441-9
25. Udayakumaran S, Biyani N, Rosenbaum DP, Ben-Sira L, Constantini S, Beni-Adani L. Posterior fossa craniotomy for trapped fourth ventricle in shunt-treated hydrocephalic children: Long-term outcome. J Neurosurg Pediatr. 2011. 7: 52-63
26. Upchurch K, Raifu M, Bergsneider M. Endoscope-assisted placement of a multiperforated shunt catheter into the fourth ventricle via a frontal transventricular approach. Neurosurg Focus. 2007. 22: E8
27. Yadav YR, Parihar V. The endoscopic trans-fourth ventricle aqueductoplasty and stent placement for the treatment of trapped fourth ventricle; stent blockage complications under estimated?. Neurol India. 2012. 60: 455
28. Zimmerman RA, Bilaniuk LT, Gallo E. Computed tomography of the trapped fourth ventricle. AJR Am J Roentgenol. 1978. 130: 503-6







James Ausman
Posted November 23, 2020, 8:01 am
Very nice case series for a challenging problem providing an excellent guide to a solution.