- Department of Neurosurgery, Faculty of Life Sciences, Kumamoto University School of Medicine, Kumamoto, Japan
Correspondence Address:
Kenji Fujimoto
Department of Neurosurgery, Faculty of Life Sciences, Kumamoto University School of Medicine, Kumamoto, Japan
DOI:10.4103/sni.sni_189_17
Copyright: © 2017 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Kenji Fujimoto, Shigetoshi Yano, Naoki Shinojima, Takuichiro Hide, Jun-ichi Kuratsu. Endoscopic endonasal transsphenoidal surgery for patients aged over 80 years with pituitary adenomas: Surgical and follow-up results. 06-Sep-2017;8:213
How to cite this URL: Kenji Fujimoto, Shigetoshi Yano, Naoki Shinojima, Takuichiro Hide, Jun-ichi Kuratsu. Endoscopic endonasal transsphenoidal surgery for patients aged over 80 years with pituitary adenomas: Surgical and follow-up results. 06-Sep-2017;8:213. Available from: http://surgicalneurologyint.com/surgicalint-articles/endoscopic-endonasal-transsphenoidal-surgery-for-patients-aged-over-80-years-with-pituitary-adenomas-surgical-and-follow%e2%80%91up-results/
Abstract
Background:With the rapid aging of the general population, the number of pituitary adenoma (PA) diagnosed in elderly patients is increasing. The aim of this study was to evaluate the efficacy of endoscopic endonasal transsphenoidal surgery (ETSS) for PA in patients aged ≥80 years.
Methods:We retrospectively reviewed the medical records of all patients aged ≥80 years who underwent ETSS for PA at our hospital from January 2001 through December 2014. Treatment results were assessed by the extent of surgical removal, symptom improvement, postoperative complications, and Karnofsky performance status (KPS). The results were also compared with the surgical result of PA patients aged
Results:Twelve patients aged ≥80 years underwent ETSS for PA. Recovery of visual function was observed in 11 patients (91.7%). Postoperative cerebrospinal fluid (CSF) leakage was observed in 3 patients. New hormonal replacement therapy was required in 2 patients. These complications had not affected patient prognosis. During the follow-up periods, deterioration of KPS was observed in 2 patients due to pneumonia or cerebral infarction. In total, 150 PA patients aged P = 0.016). Visual improvement was observed in 93.2% of the younger patient group, which was almost equal to that in the older one.
Conclusion:ETSS is a safe and effective surgical technique in PA patients aged ≥80 years.
Keywords: Eighty years old or over, endoscopic endonasal transsphenoidal surgery, pituitary adenoma
INTRODUCTION
In a rapidly aging society such as Japan, the population at least 75 years of age is currently increasing at the fastest pace ever and is expected to increase to 2.179 million (18.1%) by 2025.[
Pituitary adenoma (PA) is the third most common intracranial tumor, accounting for 15–20% of all primary intracranial tumors.[
In our hospital, we have performed a total of 3880 neurosurgical operation from January 2001 through December 2014. Patients were aged 80 years or older in 173 cases (4.5%). PA accounted for 365 cases in total, among which the patients were aged 80 years or older in 12 cases (3.3%). This ratio must not be viewed as unimportant.
We usually performed pure endoscopic endonasal transsphenoidal surgery (ETSS) with a bilateral nostril approach for PA. ETSS allows safe and sufficient resection of sellar lesion tumors and is reported to be effective even for elderly patients aged 65–70 years or older.[
In the present study, we retrospectively analyzed the results of pure ETSS for PA in patients aged 80 years or older. Characteristics of this group were compared with the characteristics of patients younger than 80 years treated in our hospital for PA.
MATERIALS AND METHODS
Patients
This study included all patients aged 80 years or older operated for PA by ETSS at our hospital from January 2001 through December 2014. Multiple surgeries on the same patient were divided into multiple cases because the purpose of this investigation was to evaluate the effectiveness of pure ETSS for PA in patients aged 80 years or older. Surgical indications were presence of neurological signs related to the tumor.
Preoperative assessment
All patients underwent preoperative magnetic resonance imaging (MRI) using a 1.5- or 3.0-T unit with T1- and T2-weighted imaging with or without Gd-based contrast medium. The degree of lateral expansion of the tumor on its prominent side was evaluated by the Knosp grading system.[
Endocrinological tests were routinely done of the basal level of growth hormone (GH), insulin-like growth factor–I, prolactin, thyroid-stimulating hormone (TSH), free thyroxin (FT), adrenocorticotropic hormone (ACTH), free cortisol, luteinizing hormone, follicle-stimulating hormone, estradiol, and testosterone. A hormone loading test was not routinely performed considering the risk of pituitary stroke induced by that test.
The preoperative physical status was evaluated according to the American Society of Anesthesiologists Physical Status (ASA-PS) grading.[
Postoperative assessment
As a postoperative evaluation, we assessed the extent of surgical removal, symptom improvement, postoperative complication, length of postoperative hospital stay, and the Karnofsky performance status (KPS). We classified the extent of surgical removal as total (no residual tumor), subtotal (presence of a residual part but more than 95% removed), and partial (less than 95% removed) on the basis of a postoperative MRI obtained in the immediate postoperative period. KPS was assessed at discharge and every 6 months. The latest KPS was obtained by telephone.
Comparison with patients younger than 80 years old
To investigate the difference in effectiveness of pure ETSS between patients aged 80 years or older and younger than 80 years old, we collected the medical records of all consecutive patients younger than 80 operated for PA by pure ETSS. Patient data collected included age, tumor size, surgical indication, preoperative and postoperative complications, symptom improvement, and length of postoperative hospital stay.
Statistical analysis
Statistical analyses were performed using SPSS software (version 19, IBM Corp.). Results are presented as means ± standard deviations. Clinical variables were compared between patients aged 80 years or older and those younger than 80 by the Fisher exact or Mann–Whitney U tests. The Fisher exact test was used for categorical variables and Mann–Whitney U test for continuous variables. Differences for which the probability values were less than 0.05 were considered statistically significant.
RESULTS
Preoperative clinical characteristics
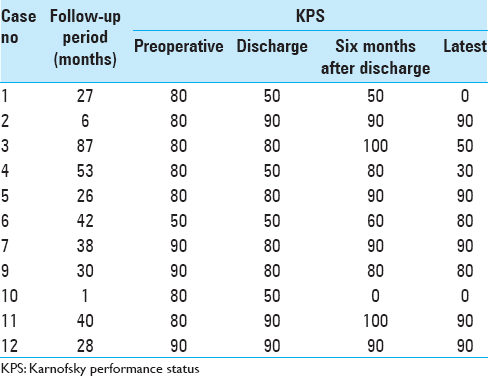
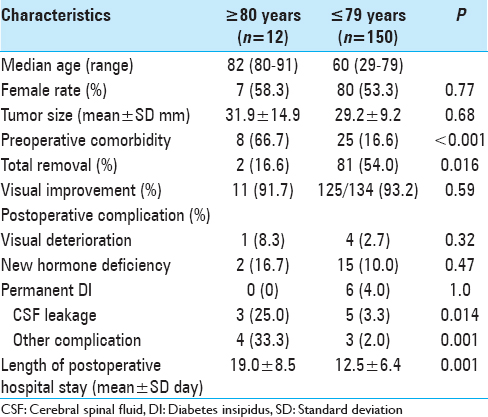
From January 2001 through December 2014, we performed ETSS on 11 PA patients aged 80 years or older. Among them, 1 patient was re-operated due to regrowth with a visual field defect, so the number of operations was 12. All patients’ characteristics are summarized in
Patients presented with a high incidence of preoperative comorbidities, including hypertension in 5 patients, cardiac disease in 3 patients, endocrinological disease in 4 patients, respiratory disease in 3 patients, diabetes mellitus in 3 patients, hyperlipidemia in 2 patients, cerebral infarction in 1 patient, dementia in 1 patient, seizure in 1 patient, and joint trouble in 2 patients. There were 2 patients taking antiplatelet agent (cases 7, 11). According to ASA-PS grading, preoperative patients’ physical status was categorized into grade 1 (no systemic disease) in 3 patients, grade 2 (mild systemic disease and normal daily life) in 7 patients, and grade 3 (severe systemic disease and possible daily life with support) in 2 patients. Preoperative KPS was 50 in 1 patient (Case 6) due to heart failure and 80 or more in the other patients.
Surgery and results
Results of the surgery are summarized in
In case a large amount of CSF leakage would occur during surgery, we intended to prepare a pedicled mucosal flap, and there was one such case (case 8).
Bony septum was not used for the construction of skull base in subtotal or partial removal cases considering the risk for postoperative hemorrhage compressing the optic nerve.
Among the total of 12 procedures, total removal was done in 2 patients [
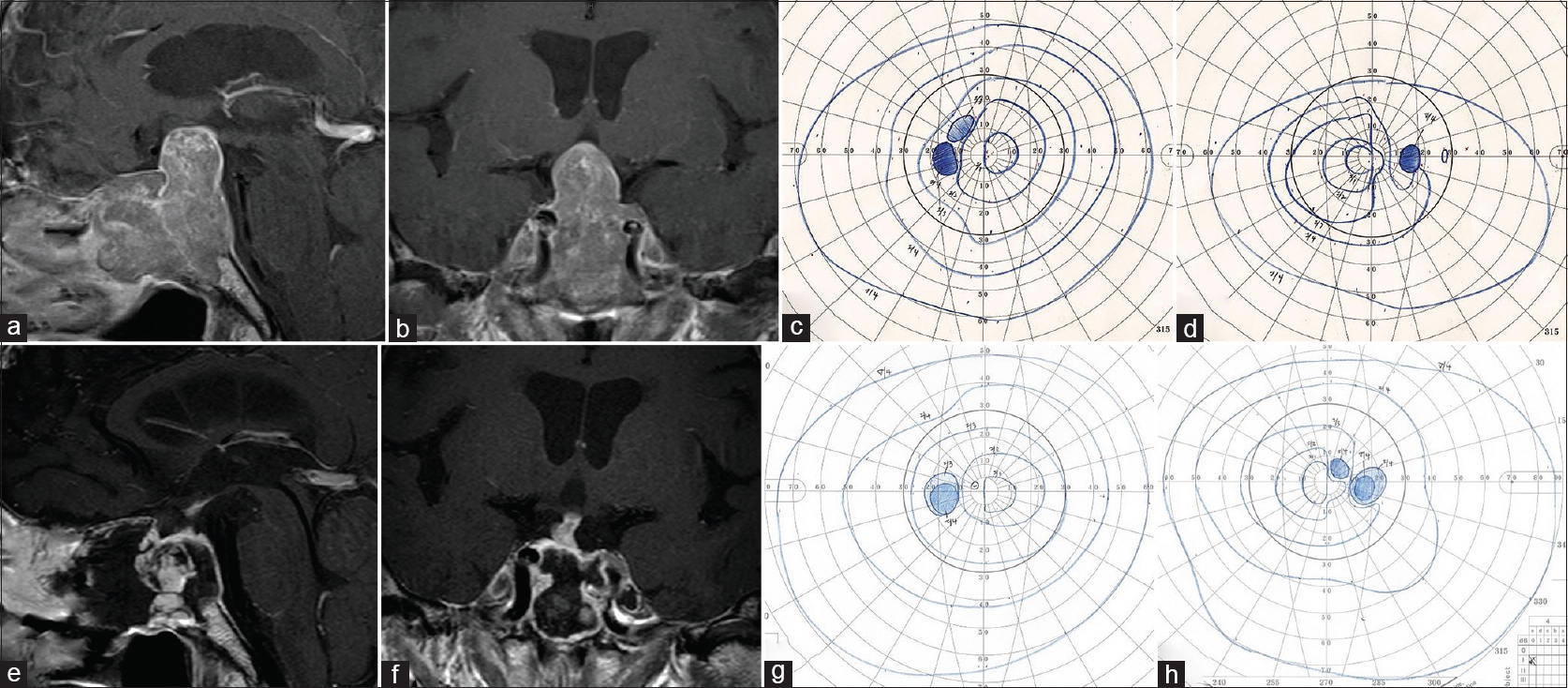
Figure 1
In Case 11, preoperative sagittal (a) and coronal (b) views of contrast-enhanced magnetic resonance imaging (MRI) demonstrated a tumor extending the suprasellar area and compressing the optic chiasm upward. Bitemporal hemianopia was detected preoperatively using the Goldmann perimeter (c, left; d, right). Postoperative sagittal (e) and coronal (f) views of contrast-enhanced MRI revealed total removal of the tumor. Postoperatively, the bitemporal hemianopia improved (g, left; h, right)
Neurological and endocrinological outcome
Recovery of visual function was accomplished in 11 patients, but deterioration was observed in 1 patient. One patient was complicated by incomplete paralysis of the oculomotor nerve that lasted for 1 month. The cause was thought to be compression of the oculomotor nerve by a hematoma formed in the left cavernous sinus at the time of removal of the tumor.
Regarding the endocrinological functions, transient diabetes insipidus (DI) was observed in 2 patients during the hospital stay, but there was no patient in whom desmopressin was needed after discharge. Additional pituitary hormone impairment was observed in 2 patients. Case 6 demonstrated decreased TSH level postoperatively, although FT3 and FT4 levels were normal. Case 10 revealed preoperative impairment of GH and TSH and additional ACTH impairment postoperatively.
Postoperative complication
Postoperative hospital stay ranged from 12 to 41 days with a mean of 19.0 ± 8.5 days. Intraoperative CSF leakage observed in 4 patients (cases 3, 4, 6, 7) was minor, whereas that in 2 patients (cases 7, 8) was moderate. Postoperative CSF leakage was observed in 3 patients (cases 4, 10, 11). In case 4, we only positioned fat graft inside the residual cavity without tight packing because intraoperative CSF leakage was minor and a residual tumor was present. However, postoperative CSF leakage lasted; therefore, we underwent multilayer reconstruction using a nasoseptal pedicled mucosal flap. In cases 10 and 11, we reconstructed the skull base with fat graft and polyglactin acid sheet owing to the absence of intraoperative CSF leakage. Four days after the operation, CSF leakage was suspected but showed improvement after 2 days of rest.
Three patients experienced postoperative general complications (cases 1, 6, 10). Case 1 was complicated by aspiration pneumonia and cystitis. This induced an adrenal crisis due to cortisol deficiency. Hydrocortisone administration improved the adrenal crisis, but pulmonary edema and renal failure were prolonged. In case 6, heart failure, from which patient had suffered before, transiently deteriorated postoperatively and required medical treatment. In case 10 [
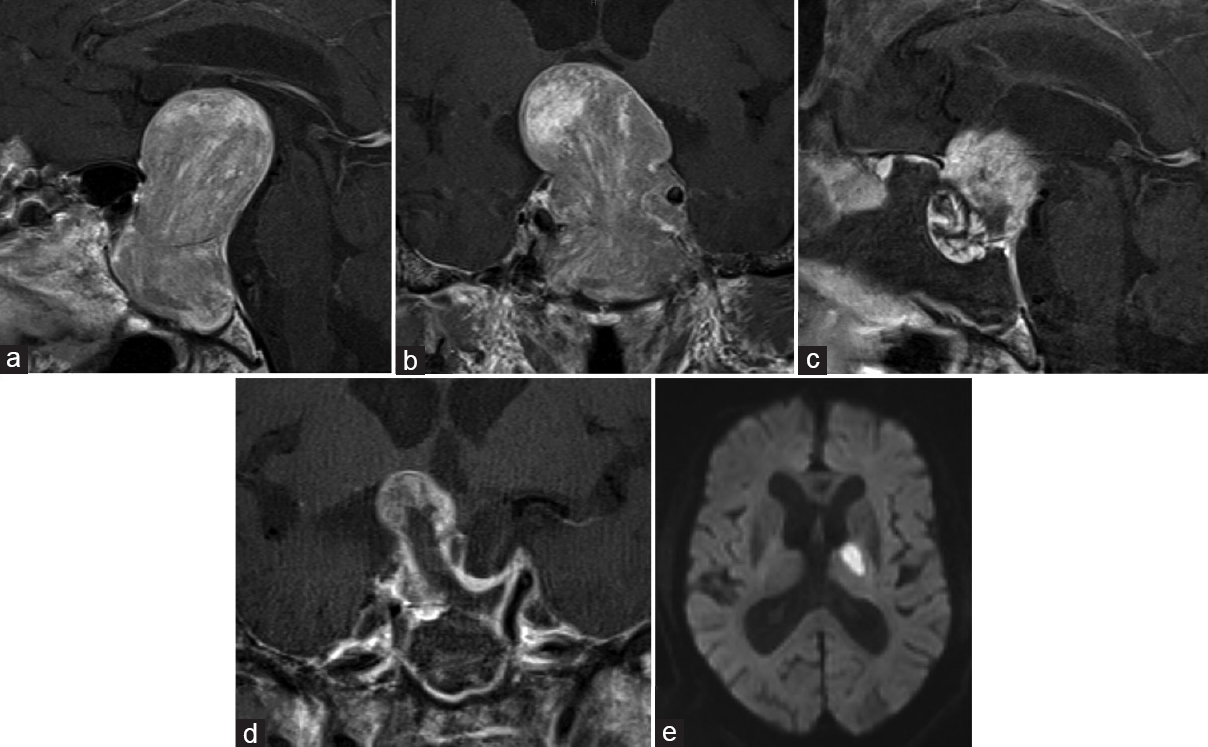
Figure 2
In Case 10, preoperative sagittal (a) and coronal (b) views of contrast-enhanced magnetic resonance imaging (MRI) showed a giant (60 mm) tumor presenting a major suprasellar extension and compression of the third ventricle. Postoperatively, sagittal (c) and coronal (d) views contrast-enhanced MRI revealed decompression of the tumor. Postoperative diffusion-weighted image (e) showed a left thalamic infarction
Follow-up results
Follow-up periods ranged from 1 to 87 months with a mean of 34.4 ± 24.1 months. One patient (case 8) suffered from regrowth of a residual tumor and underwent re-operation. The other patients did not suffer from deterioration of their visual function after surgery. None of the patients underwent gamma knife radiosurgery in follow-up periods. The time course of KPS from preoperative to final survey time is shown in
Comparison with patients younger than 80 years old
In the present study, all PAs in the patients aged 80 years or older were nonfunctioning, therefore, we compared these patients with nonfunctioning PA patients younger than 80. From January 2001 through December 2014, we performed ETSS for consecutive 150 nonfunctioning PA patients younger than 80, with a mean age of 60.0 ± 12.4 years.
Length of postoperative hospital stay of these patients was longer than that of the patients younger than 80 (19.0 ± 8.5 days vs 12.5 ± 6.4 days, respectively; P = 0.001).
DISCUSSION
In a rapidly aging society such as Japan, the population of people aged 75 years or older is expected to rise to 2.179 million (18.1%) by 2025.[
Surgical outcome
The present study shows that ETSS for PA in patients aged 80 years or older is effective enough to improve compression symptoms caused by the tumor. Though we did not dare to remove the entire tumor to decompress the optic nerve first, visual improvement was observed in 91.7%, which was almost the same as the result in patients younger than 80 years of age (93.2%). One patient underwent re-operation for symptom recurrence, but the other patients did not require re-operation or gamma knife radiosurgery during the follow-up period.
Yunoue et al. reported the good outcome of endoscopy-assisted microscopic transsphenoidal surgery in 10 patients aged 80 years or older.[
Thus, our series are not necessarily typical cases of PA patients aged 80 years or older, but our series together with Yunoues’ series indicate that transsphenoidal surgery is effective for various types of PA patients aged 80 years or older.
Treatment outcomes of pure ETSS for PA have been reported in patients aged 65 or 70 years or older.[
The present study is the first study revealing the effectivity of pure ETSS for PA patients aged 80 years or older.
Considering the slow growing nature of PA, especially in the elderly and their limited life expectancy, our policy of decompression as much as possible within the safe range, in which total removal is not always sought, may have validity.[
Surgical complications
Postoperative complications were observed more frequently in the elderly patients studied in the present report. In particular, CSF leakage was significantly more frequent (25%).
It has been previously reported that more postoperative complications occur in elderly people than that in young people.[
Previously published rates of postoperative CSF leakage after surgery for PA vary from 0.5% to 6.0% and in the elderly population studies they vary from 4.9% to 15.6%.[
Although we have removed tumors to prevent CSF leakage, in cases of recurrent or giant PA, a pedicled mucosal flap to reconstruct the skull base should be prepared regardless of CSF leakage during surgery.
Previous reports have indicated that postoperative new anterior hypopituitarism occurred in 1.4–19.8% of the patients.[
Although postoperative hormone replacement was required in 8 patients, none of the patients required additional treatment with symptoms of hormone deficiency in the follow-up period after discharge. Hormone deficiency had been well controlled with oral medicine at discharge. Zhan et al. reported that there was no difference between patients aged 65 years or older and 40–55 years old in postoperative new anterior hypopituitarism occurrence, but there is no report on patients aged 80 years or older.[
In the present study, postoperative hospital stay was longer in patients aged 80 years or older due to the therapy for postoperative complications. In the follow-up period, poor prognosis was observed in 2 patients (cases 1, 10). In case 10, with a giant PA, left thalamic infarction was thought to occur by stretching or kinking of the thalamotuberal arteries caused by tumor reduction. The surgery-related left thalamic infarction affected prognosis. Giant PA has been reported to have more postoperative complications than nongiant PA, therefore the poor prognosis in case 10 was likely to be due to the characteristics of the tumor itself rather than the influence of old age.[
Study limitations
Our study has several limitations. To begin, this study is a retrospective study of data that were acquired previously, and it has the inherent limitations of this kind of studies. Second, the number of cases is small. Third, the follow-up period of some patients may have been short. Considering that recurrence of PA can occur many years after surgery, a longer follow-up is required.
CONCLUSION
Pure endoscopic endonasal surgery is an effective and safe surgical technique in terms of visual function recovery even in PA patients aged 80 years or older. Considering the slow-growing nature of PA in the elderly and the limited life expectancy of the elderly, it is not always necessary to aggressively perform total removal of the tumor; this is in contrast to the situation in young people. Subtotal and partial removal provides sufficient decompression of the optic nerve and a prolonged progression-free status without postoperative complications influencing the long-term prognosis. Elderly age would not preclude such patients from undergoing vision-saving surgery as needed when their general condition is well preserved.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Berker M, Hazer DB, Yucel T, Gurlek A, Cila A, Aldur M. Complications of endoscopic surgery of the pituitary adenomas: Analysis of 570 patients and review of the literature. Pituitary. 2012. 15: 288-300
2. Chabot JD, Chakraborty S, Imbarrato G, Dehdashti AR. Evaluation of Outcomes After Endoscopic Endonasal Surgery for Large and Giant Pituitary Macroadenoma: A Retrospective Review of 39 Consecutive Patients. World Neurosurg. 2015. 84: 978-88
3. Dallapiazza RF, Grober Y, Starke RM, Laws ER, Jane JA. Long-term results of endonasal endoscopic transsphenoidal resection of nonfunctioning pituitary macroadenomas. Neurosurgery. 2015. 76: 42-
4. Ezzat S, Asa SL, Couldwell WT, Barr CE, Dodge WE, Vance ML. The prevalence of pituitary adenomas: A systematic review. Cancer. 2004. 101: 613-9
5. Ferrante L, Trillo G, Ramundo E, Celli P, Jaffrain-Rea ML, Salvati M. Surgical treatment of pituitary tumors in the elderly: Clinical outcome and long-term follow-up. J Neurooncol. 2002. 60: 185-91
6. Gondim JA, Almeida JP, Albuquerque LA, Gomes EF, Schops M. Giant pituitary adenomas: Surgical outcomes of 50 cases operated on by the endonasal endoscopic approach. World Neurosurg. 2014. 82: e281-90
7. Gondim JA, Almeida JP, de Albuquerque LA, Gomes E, Schops M, Mota JI. Endoscopic endonasal transsphenoidal surgery in elderly patients with pituitary adenomas. J Neurosurg. 2015. 123: 31-8
8. Grossman R, Mukherjee D, Chaichana KL, Salvatori R, Wand G, Brem H. Complications and death among elderly patients undergoing pituitary tumour surgery. Clin Endocrinol (Oxf). 2010. 73: 361-8
9. Halvorsen H, Ramm-Pettersen J, Josefsen R, Ronning P, Reinlie S, Meling T. Surgical complications after transsphenoidal microscopic and endoscopic surgery for pituitary adenoma: A consecutive series of 506 procedures. Acta Neurochir (Wien). 2014. 156: 441-9
10. Hong J, Ding X, Lu Y. Clinical analysis of 103 elderly patients with pituitary adenomas: Transsphenoidal surgery and follow-up. J Clin Neurosci. 2008. 15: 1091-5
11. Juraschka K, Khan OH, Godoy BL, Monsalves E, Kilian A, Krischek B. Endoscopic endonasal transsphenoidal approach to large and giant pituitary adenomas: Institutional experience and predictors of extent of resection. J Neurosurg. 2014. 121: 75-83
12. Knosp E, Steiner E, Kitz K, Matula C. Pituitary adenomas with invasion of the cavernous sinus space: A magnetic resonance imaging classification compared with surgical findings. Neurosurgery. 1993. 33: 610-
13. Kovacs K, Ryan N, Horvath E, Singer W, Ezrin C. Pituitary adenomas in old age. J Gerontol. 1980. 35: 16-22
14. Kurosaki M, Ludecke DK, Flitsch J, Saeger W. Surgical treatment of clinically nonsecreting pituitary adenomas in elderly patients. Neurosurgery. 2000. 47: 843-
15. Kurosaki M, Saeger W, Ludecke DK. Pituitary tumors in the elderly. Pathol Res Pract. 2001. 197: 493-7
16. Locatelli M, Bertani G, Carrabba G, Rampini P, Zavanone M, Caroli M. The trans-sphenoidal resection of pituitary adenomas in elderly patients and surgical risk. Pituitary. 2013. 16: 146-51
17. Losa M, Franzin A, Mangili F, Terreni MR, Barzaghi R, Veglia F. Proliferation index of nonfunctioning pituitary adenomas: Correlations with clinical characteristics and long-term follow-up results. Neurosurgery. 2000. 47: 1313-
18. Mamelak AN, Carmichael J, Bonert VH, Cooper O, Melmed S. Single-surgeon fully endoscopic endonasal transsphenoidal surgery: Outcomes in three-hundred consecutive cases. Pituitary. 2013. 16: 393-401
19. Minniti G, Esposito V, Piccirilli M, Fratticci A, Santoro A, Jaffrain-Rea ML. Diagnosis and management of pituitary tumours in the elderly: A review based on personal experience and evidence of literature. Eur J Endocrinol. 2005. 153: 723-35
20. cited 18 February 2017. Available from: http://www.ipss.go.jp/syoushika/tohkei/Popular/P_Detail2016.asp?fname=T02-09.htm.
21. Ostrom QT, Gittleman H, Fulop J, Liu M, Blanda R, Kromer C. CBTRUS Statistical Report: Primary Brain and Central Nervous System Tumors Diagnosed in the United States in 2008-2012. Neuro Oncol. 2015. 17: iv1-iv62
22. Owens WD, Felts JA, Spitznagel EL. ASA physical status classifications: A study of consistency of ratings. Anesthesiology. 1978. 49: 239-43
23. Robenshtok E, Benbassat CA, Hirsch D, Tzvetov G, Cohen ZR, Iraqi HM. Clinical course and outcome of nonfunctioning pituitary adenomas in the elderly compared with younger age groups. Endocr Pract. 2014. 20: 159-64
24. Sheehan JM, Douds GL, Hill K, Farace E. Transsphenoidal surgery for pituitary adenoma in elderly patients. Acta Neurochir (Wien). 2008. 150: 571-4
25. Tabaee A, Anand VK, Barron Y, Hiltzik DH, Brown SM, Kacker A. Endoscopic pituitary surgery: A systematic review and meta-analysis. J Neurosurg. 2009. 111: 545-54
26. Tanaka Y, Hongo K, Tada T, Sakai K, Kakizawa Y, Kobayashi S. Growth pattern and rate in residual nonfunctioning pituitary adenomas: Correlations among tumor volume doubling time, patient age, and MIB-1 index. J Neurosurg. 2003. 98: 359-65
27. Teramoto A, Hirakawa K, Sanno N, Osamura Y. Incidental pituitary lesions in 1,000 unselected autopsy specimens. Radiology. 1994. 193: 161-4
28. Turner HE, Adams CB, Wass JA. Pituitary tumours in the elderly: A 20 year experience. Eur J Endocrinol. 1999. 140: 383-9
29. Wagenmakers MA, Netea-Maier RT, van Lindert EJ, Timmers HJ, Grotenhuis JA, Hermus AR. Repeated transsphenoidal pituitary surgery (TS) via the endoscopic technique: A good therapeutic option for recurrent or persistent Cushing's disease (CD). Clin Endocrinol (Oxf). 2009. 70: 274-80
30. Yamada S, Fukuhara N, Oyama K, Takeshita A, Takeuchi Y. Repeat transsphenoidal surgery for the treatment of remaining or recurring pituitary tumors in acromegaly. Neurosurgery. 2010. 67: 949-56
31. Yunoue S, Tokimura H, Tominaga A, Fujio S, Karki P, Usui S. Transsphenoidal surgical treatment of pituitary adenomas in patients aged 80 years or older. Neurosurg Rev. 2014. 37: 269-
32. Zhan R, Ma Z, Wang D, Li X. Pure Endoscopic Endonasal Transsphenoidal Approach for Nonfunctioning Pituitary Adenomas in the Elderly: Surgical Outcomes and Complications in 158 Patients. World Neurosurg. 2015. 84: 1572-8