- Department of Neurosurgery, Faculty of Medicine, University of Tsukuba, Tsukuba, Ibaraki, Japan
- Division of Stroke Prevention and Treatment, Faculty of Medicine, University of Tsukuba, Tsukuba, Ibaraki, Japan.
Correspondence Address:
Yoshiro Ito, Department of Neurosurgery, Faculty of Medicine, University of Tsukuba, Tsukuba, Ibaraki, Japan.
DOI:10.25259/SNI_1041_2021
Copyright: © 2022 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Kazuho Karasudani1, Yoshiro Ito1, Ai Muroi1, Masayuki Sato1, Aiki Marushima1, Mikito Hayakawa2, Yuji Matsumaru1, Eiichi Ishikawa1. Endoscopic third ventriculostomy for noncommunicating hydrocephalus by vertebrobasilar dolichoectasia: A case report. 22-Apr-2022;13:166
How to cite this URL: Kazuho Karasudani1, Yoshiro Ito1, Ai Muroi1, Masayuki Sato1, Aiki Marushima1, Mikito Hayakawa2, Yuji Matsumaru1, Eiichi Ishikawa1. Endoscopic third ventriculostomy for noncommunicating hydrocephalus by vertebrobasilar dolichoectasia: A case report. 22-Apr-2022;13:166. Available from: https://surgicalneurologyint.com/surgicalint-articles/11549/
Abstract
Background: Vertebrobasilar dolichoectasia (VBD) is a vasculopathy characterized by the elongation, widening, and tortuosity of a cerebral artery. Rarely, hydrocephalus results when the extended basilar artery impairs communication of the cerebral ventricle and cerebrospinal fluid dynamics. We experienced such a case when a patient underwent endoscopic third ventriculostomy (ETV) for noncommunicating hydrocephalus with VBD.
Case Description: A 54-year-old man presented with cognitive dysfunction and was diagnosed with VBD by magnetic resonance imaging (MRI). Seven years later, he exhibited subacute impaired consciousness due to acute noncommunicating hydrocephalus, undergoing external ventricular drainage (EVD) that improved consciousness. After EVD removal, the noncommunicating hydrocephalus did not recur; however, 7 months later, subacute consciousness impairment due to noncommunicating hydrocephalus was again observed. MRI showed a significant dilation of both lateral ventricles and ballooning of the third ventricle while the right posterior cerebral artery shifted slightly posteriorly. The patient underwent ETV and clinical symptoms improved. One year after the treatment, MRI observed a patent ETV fenestration and no deleterious changes in clinical symptoms were observed.
Conclusion: ETV can be an effective treatment for the noncommunicating hydrocephalus with VBD when performed with preoperative assessment of vascular anatomy and attention to vascular injury.
Keywords: Countercurrent pulsation, Endoscopic third ventriculostomy, Non-communicating hydrocephalus, Vertebrobasilar dolichoectasia
INTRODUCTION
Dolichoectasia is a cerebral vasculopathy with deformation, mainly in the vertebral and basilar arteries (vertebrobasilar dolichoectasia; VBD), with prevalence estimates ranging between 0.2% and 4.4%. However, among patients presenting with stroke, the prevalence ranges between 2.6% and 17.1%.[
CASE PRESENTATION
A 54-year-old man presented with cognitive dysfunction and was diagnosed with VBD at another hospital. Magnetic resonance imaging (MRI) showed that the basilar artery was elongated, protruding upward, and compressing the third ventricle floor [
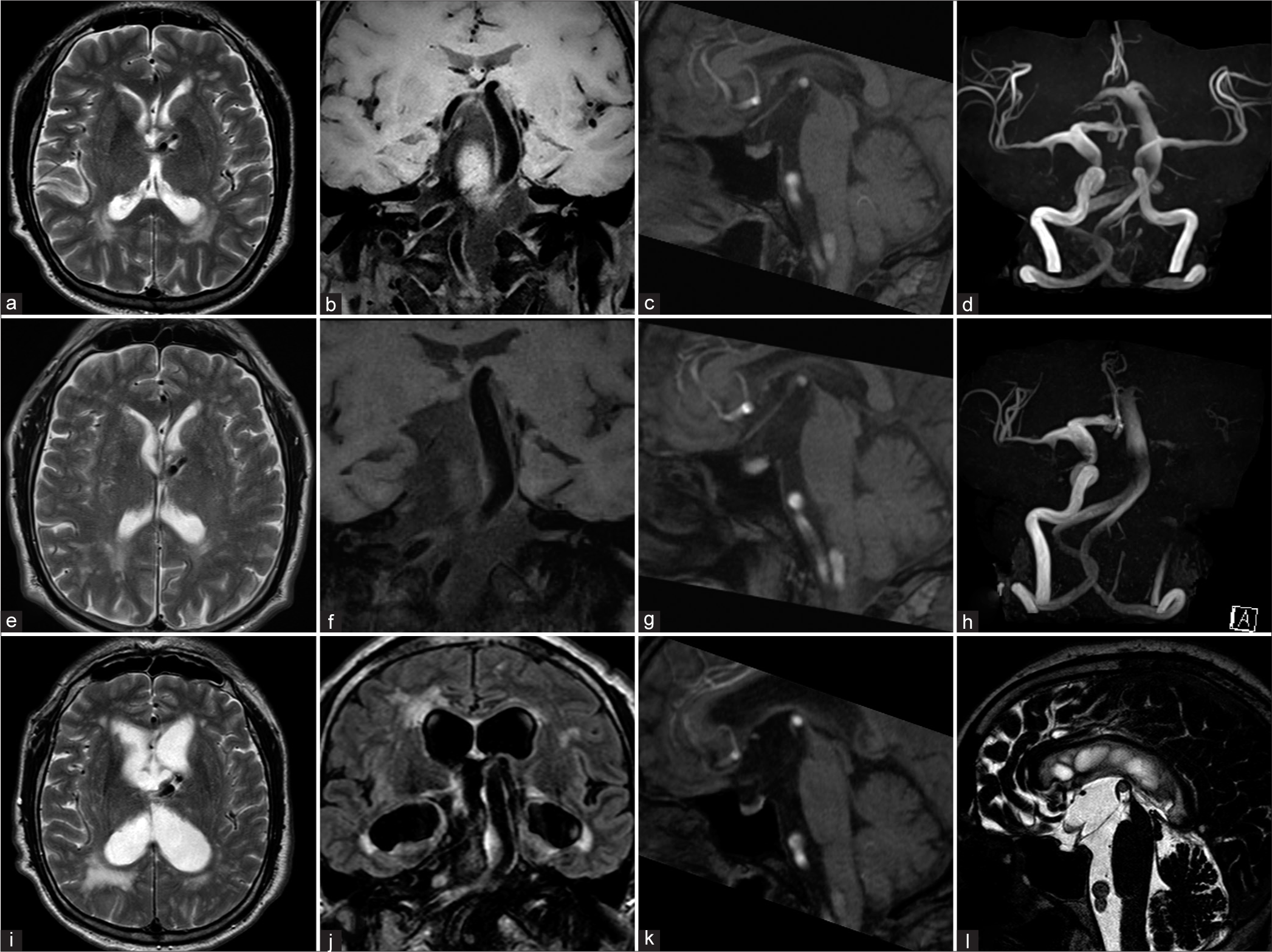
Figure 1:
(a-d) Initial MRI (a: T2-weighted image, b: T1-weighted volume isotropic turbo spin-echo acquisition, c: sagittal reconstruction image of MRA, and d: MRA). The basilar artery was dilated and the top of the artery extended upward (b and d). The right posterior cerebral artery extended the floor of the third ventricle upward but did not touch the fornix (c). The enlargement of the ventricles was not observed (a). (e-h) MRI 5 years later (the second examination); (e) T2-weighted image, (f) T1-weighted volume isotropic turbo spin-echo acquisition, (g: sagittal reconstruction image of MRA, h: MRA). The left internal carotid artery was asymptomatically occluded (h). The basilar artery and right posterior cerebral artery extended further upward and the floor of the third ventricle touched the fornix (f and g). The enlargement of the ventricles was not observed (e). (i-l) MRI 1 year and 7 months later (the third examination, just before ETV); (i) T2-weighted image, (j) fluid-attenuated inversion recovery image, (k) sagittal reconstruction image of MRA, and (l) T2 drive image. The bilateral ventricle and the third ventricle were enlarged (I and j). The right posterior cerebral artery extended further upward and touched the fornix while deformities of the midbrain and pons were also observed (k). The floor of the third ventricle was observed to be ballooning (l).
MRI showed a significant dilation of both lateral ventricles, periventricular hyperintensity, and ballooning of the third ventricle [
An insertion port was placed on the right frontal lobe toward the anterior horn of the right ventricle and a flexible endoscope was inserted [
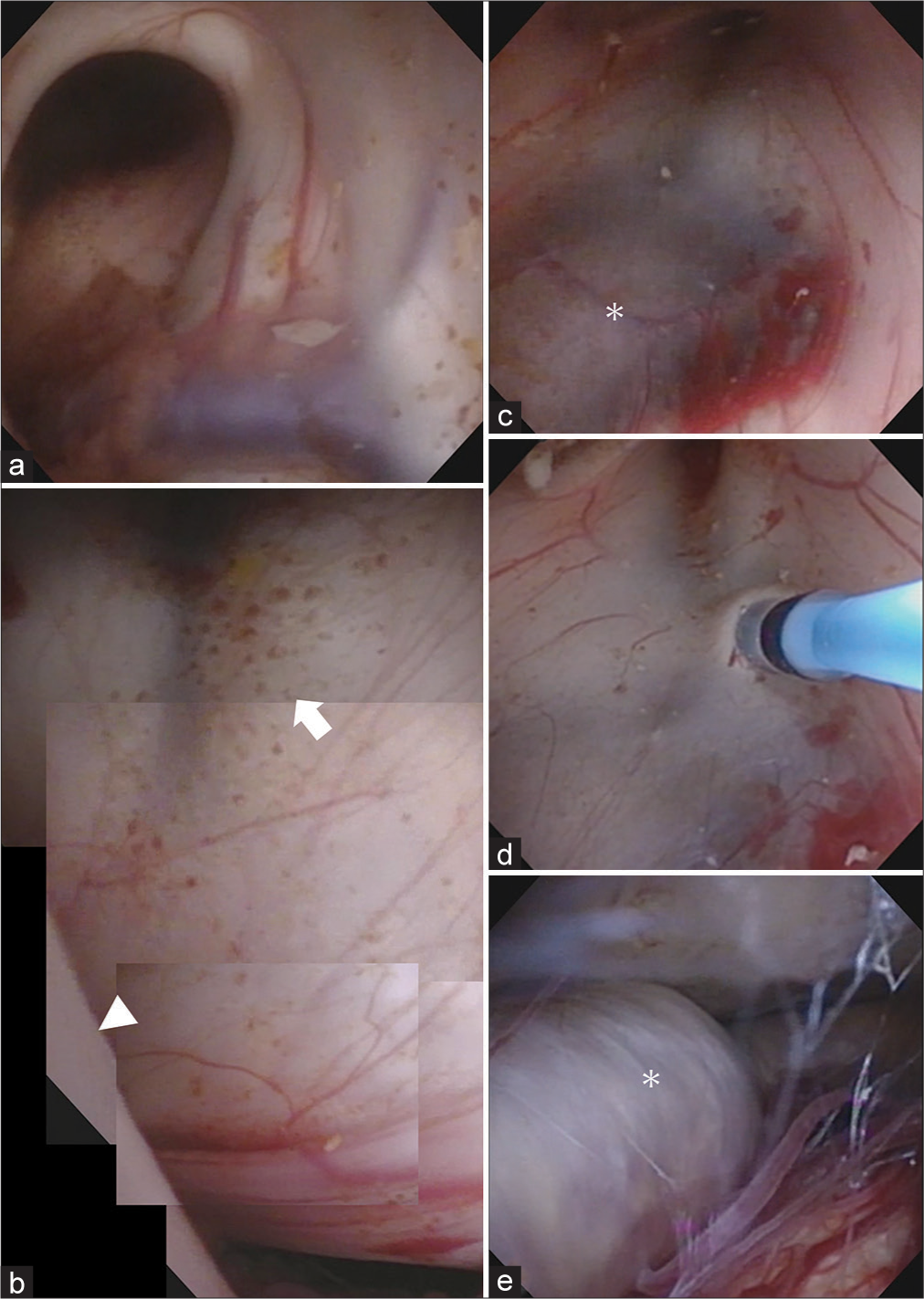
Figure 2:
Images of the endoscopic third ventricle ventriculostomy. (a) The foramen of Monro was observed from the right ventricle, which was enlarged. (b) Consolidated images of endoscopic findings in the third ventricle. The floor of the third ventricle extended to the fornix due to the upward extension of the right posterior cerebral artery (white arrowhead). The mamillary body was stretched (white arrow). (c) The basilar artery (asterisk) was confirmed through the floor of the third ventricle. (d) The floor of the third ventricle was fenestrated to avoid injuring the basilar artery. (e) The vascular wall of the basilar artery (asterisk) had white sclerotic changes.
On postoperative day 7, MRI showed a decrease in dilatation of both lateral ventricles and the periventricular hyperintensity had also improved [
DISCUSSION
Pathologically, dolichoectasia is a result of internal elastic lamina disruption from matrix metalloproteinase dysfunction and the migration of smooth muscle cells that induce breakdown and remodeling of the tunica media. As a result, affected arterial walls are prone to dilatation.[
We experienced a case of noncommunicating hydrocephalus with VBD and performed ETV with a good long-term outcome. The patient initially had stenosis in the third ventricle due to PCA compression caused by the upward extension of the BA. Moreover, the temporary increase in CSF pulse pressure caused by VBD may have dilated the third ventricle and deviated the PCA posteriorly, resulting in third ventricle obstruction. EVD reduced the size of the third ventricle, released the PCA deviation, and restored CSF flow, which may have temporarily improved the hydrocephalus thought to be caused by the impairment of CSF communication due to countercurrent pulsation rather than simple CSF flow obstruction. We chose ETV in the present case for the following reasons: first, ETV is the first choice for noncommunicating hydrocephalus. Second, considering the pathogenesis of hydrocephalus, we thought that ETV with spontaneous CSF drainage would have fewer complications than VP shunt placement with forced CSF drainage.[
We assert that hemodynamic change is a key trigger of the dolichoectatic process since the basilar artery began to extend upward after the ICA was asymptomatically occluded, although annual MRI images after the diagnosis of VBD showed no changes for 5 years. The increase in hemodynamic stress on the posterior circulation due to ICA occlusion was considered to have caused the upward extension of the basilar artery.[
CONCLUSION
We treated a case of noncommunicating hydrocephalus with VBD by ETV. ETV for noncommunicating hydrocephalus with VBD can be an effective treatment when performed with a preoperative assessment of vascular anatomy and attention to vascular injury.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Acknowledgments
The authors would like to thank Dr. Alexander Zaboronok of the University of Tsukuba, Faculty of Medicine, Department of Neurosurgery for professional and language revision, and Dr. Bryan J. Mathis of the University of Tsukuba Hospital International Medical Center for language revision.
References
1. Ebrahimzadeh K, Bakhtevari MH, Shafizad M, Rezaei O. Hydrocephalus as a rare compilation of vertebrobasilar dolichoectasia: A case report and review of the literature. Surg Neurol Int. 2017. 8: 256
2. Gutierrez J, Sacco RL, Wright CB. Dolichoectasia-an evolving arterial disease. Nat Rev Neurol. 2011. 7: 41-50
3. Jagetia A, Patel K, Sachdeva D, Rathore L. Dolichoectatic internal carotid artery presenting as a sellar-suprasellar mass with symptomatic hydrocephalus. Neurol India. 2017. 65: 681-2
4. Jiang L, Gao G, Zhou Y. Endoscopic third ventriculostomy and ventriculoperitoneal shunt for patients with non-communicating hydrocephalus: A PRISMA-compliant meta-analysis. Medicine. 2018. 97: e12139
5. Labidi M, Lavoie P, Lapointe G, Obaid S, Weil AG, Bojanowski MW. Predicting success of endoscopic third ventriculostomy: Validation of the ETV success score in a mixed population of adult and pediatric patients. J Neurosurg. 2015. 123: 1447-55
6. Lee JM, Park JS, Koh EJ. Severe vertebrobasilar dolichoectasia as a cause of obstructive hydrocephalus: A case report. Medicine. 2019. 98: e15752
7. Lou M, Caplan LR. Vertebrobasilar dilatative arteriopathy (dolichoectasia). Ann N Y Acad Sci. 2010. 1184: 121-33
8. Lu L, Chen H, Weng S, Xu Y. Endoscopic third ventriculostomy versus ventriculoperitoneal shunt in patients with obstructive hydrocephalus: Meta-analysis of randomized controlled trials. World Neurosurg. 2019. 129: 334-40
9. Mohammed K, Iqbal J, Kamel H, Mathew J, Al-Sulaiti G. Obstructive hydrocephalus and facial nerve palsy secondary to vertebrobasilar dolichoectasia: Case report. Surg Neurol Int. 2018. 9: 60
10. Oishi Y, Ueda R, Kuranari Y, Imanishi T. Vertebrobasilar dolichoectasia causing symptomatic hydrocephalus due to third ventricular compression. J Stroke Cerebrovasc Dis. 2018. 27: 3503-4
11. Pico F, Labreuche J, Amarenco P. Pathophysiology, presentation, prognosis, and management of intracranial arterial dolichoectasia. Lancet Neurol. 2015. 14: 833-45
12. Samim M, Goldstein A, Schindler J, Johnson MH. Multimodality imaging of vertebrobasilar dolichoectasia: Clinical presentations and imaging spectrum. Radiographics. 2016. 36: 1129-46
13. Smoker WR, Corbett JJ, Gentry LR, Keyes WD, Price MJ, McKusker S. High-resolution computed tomography of the basilar artery: 2. Vertebrobasilar dolichoectasia: Clinical-pathologic correlation and review. AJNR Am J Neuroradiol. 1986. 7: 61-72
14. Wolters FJ, Rinkel GJ, Vergouwen MD. Clinical course and treatment of vertebrobasilar dolichoectasia: A Systematic review of the literature. Neurol Res. 2013. 35: 131-7
15. Yuh SJ, Alkherayf F, Lesiuk H. Dolichoectasia of the vertebral basilar and internal carotid arteries: A case report and literature review. Surg Neurol Int. 2013. 4: 153
16. Zhang DP, Peng YF, Ma QK, Zhao M, Zhang HL, Yin S. Why does my patient’s basilar artery continue to grow? A four-year case study of a patient with symptoms of vertebrobasilar dolichoectasia. BMC Neurol. 2018. 18: 45