- Department of Pediatric Neurosurgery, Cincinnati Children’s Medical Center, Cincinnati, Ohio, United States.
- Department of Neurosurgery, University of Cincinnati College of Medicine, Cincinnati, Ohio, United States.
Correspondence Address:
Jesse Skoch, Division of Pediatric Neurosurgery, Cincinnati Children’s Medical Center, Cincinnati, Ohio, United States.
DOI:10.25259/SNI_302_2022
Copyright: © 2022 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Baher Hanna1, Michael W. Robinson2, Jesse Skoch1. Exclusively endoscopic management of complicated pineal cysts in young children: Definitive treatment through single burr-hole technique. 01-Jul-2022;13:281
How to cite this URL: Baher Hanna1, Michael W. Robinson2, Jesse Skoch1. Exclusively endoscopic management of complicated pineal cysts in young children: Definitive treatment through single burr-hole technique. 01-Jul-2022;13:281. Available from: https://surgicalneurologyint.com/?post_type=surgicalint_articles&p=11699
Abstract
Background: The management of complicated symptomatic pineal cysts in the pediatric population is challenging and variable. Surgical management may include treatment of hydrocephalus alone, or direct treatment of the cyst with or without direct hydrocephalus management. This is typically done through craniotomy-based microsurgical approaches to the pineal region or an endoscopic transventricular approach.
Methods: We present a stepwise minimally invasive technique to treat complicated pineal cysts in young children associated with an obstructive hydrocephalus in a single procedure through third ventriculostomy combined with an intraventricular marsupialization of the pineal cyst through a single burr-hole using stereotactic navigation.
Results: Two young patients with over 2 years of follow-up have done well without complication using this technique. Other literature reports for complex pineal cysts in pediatric patients are reviewed and this technique is not previously described for this population.
Conclusion: Endoscopic third ventriculostomy and cyst marsupialization using a single burr-hole and stereotactic navigation for symptomatic or enlarging pineal cysts in children allow for minimally invasive management, a rapid recovery, short hospital stay, and durable outcome owed to redundant CSF flow pathways.
Keywords: Complex pineal cyst, Endoscopic surgery, Endoscopic third ventriculostomy, Pineal apoplexy, Pineal cyst, Single burr-hole
INTRODUCTION
Pineal cysts are typically benign entities that are most commonly diagnosed in young women; the prevalence of pineal cysts is estimated at 2.4% (1.5% in males).[
Complicated pineal cysts (defined here as symptomatic beyond isolated headache, causing mass effect, hydrocephalus, or papilledema) typically warrant surgical management, but likely owed to their rarity, no standardized management exists. There are limited reports in the pediatric literature regarding treatment of complicated pineal cysts. Published examples include observation only, endoscopic third ventriculocisternostomy (ETV) alone, external ventricular drain, ETV then craniotomy for cyst resection, ventriculoperitoneal shunt then craniotomy, and craniotomy only for cyst excision through supracerebellarinfratentorial approach [
Neuroendoscopic intraventricular techniques offer the advantage of treating or preventing any associated hydrocephalus by a third ventricular floor cisternostomy and can be done simultaneously with fenestration and marsupialization of the anterior cyst wall.[
MATERIALS AND METHODS
Herein, we apply a stepwise surgical technique based on our experience with management of two recent cases of complicated pediatric pineal cysts, including a rare case of pineal apoplexy, successfully treated with endoscopic third ventriculostomy with combined endoscopic cyst fenestration to restore normal flow to the cerebral aqueduct and also achieve cyst wall biopsy [
Figure 1:
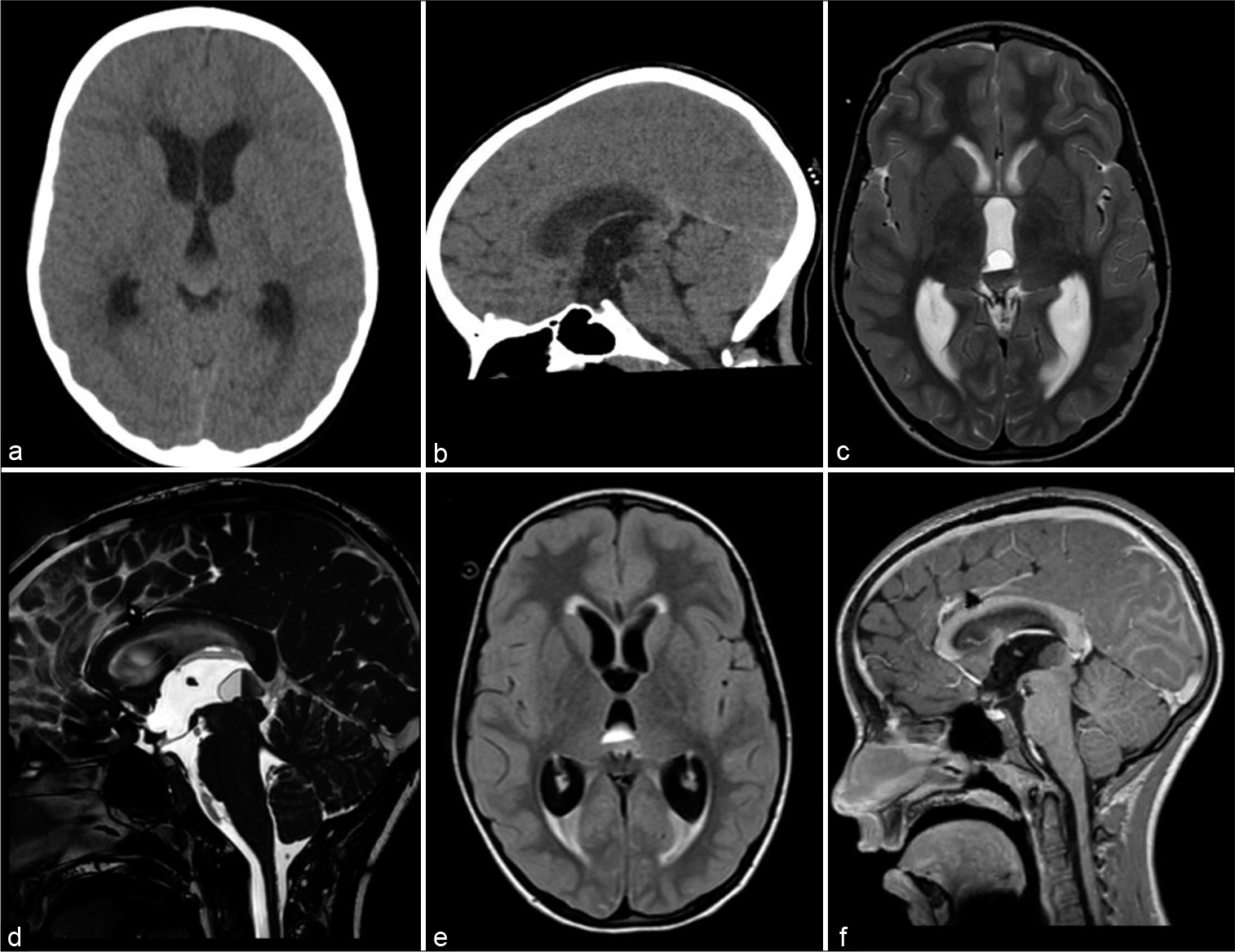
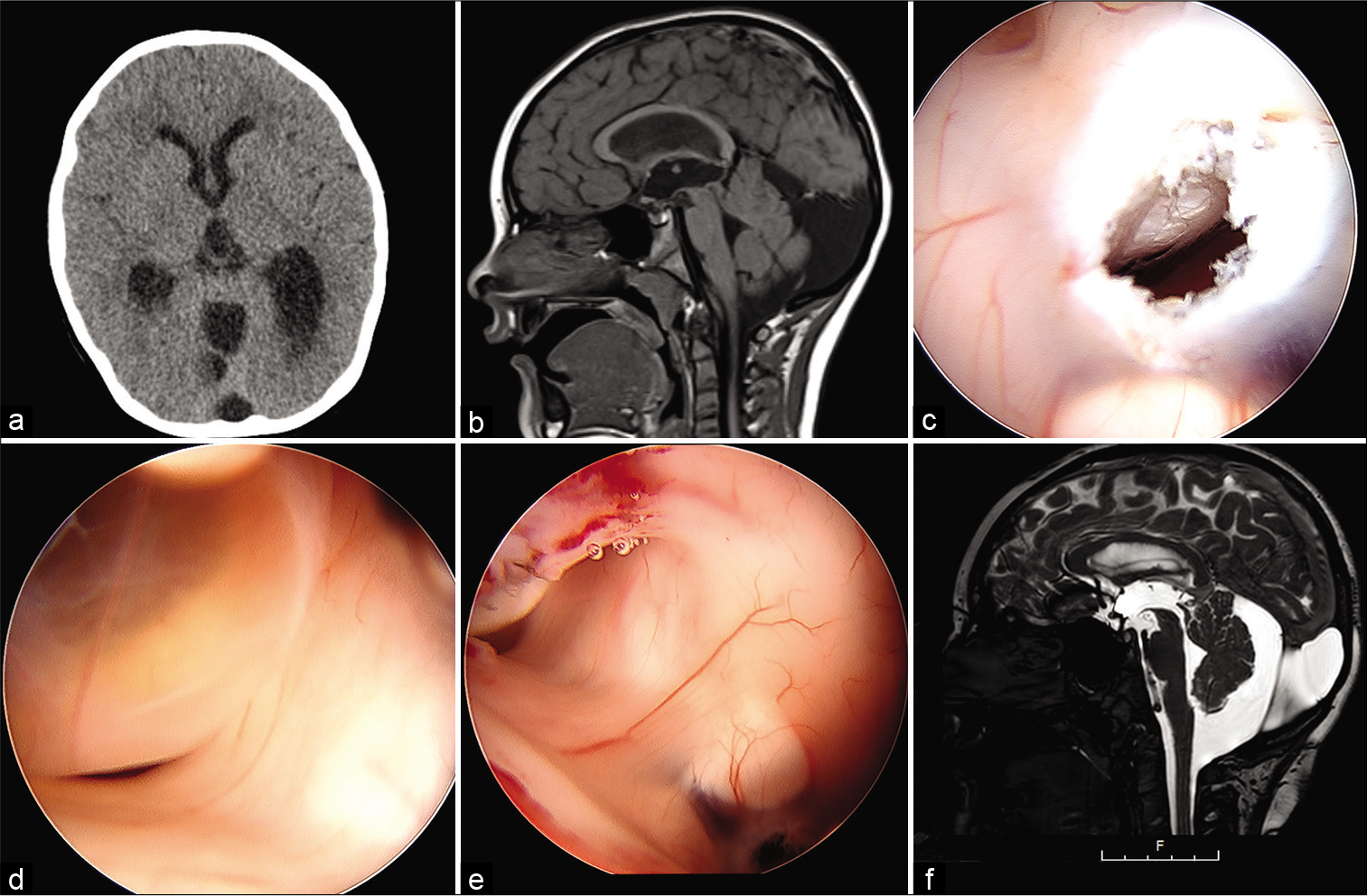
Case 1 preoperative radiographs. (a) Axial, (b) and sagittal noncontrasted CT head demonstrating lateral and third ventriculomegaly and a mixed density pineal region mass. (c) Axial T2, (d) sagittal T2, (e) axial FLAIR, and (f) sagittal T1 with contrast MRI of the brain revealing a nonenhancing 1.5 cm pineal region cyst with a dependent region of T1 hyperintense, T2 hypointense, with magnetic susceptibility, and consistent with hemorrhage. The ventral portion displays T1 hypointense, T2 hyperintense fluid collection, and consistent with blood components. The cerebral aqueduct is obstructed.
Figure 2:
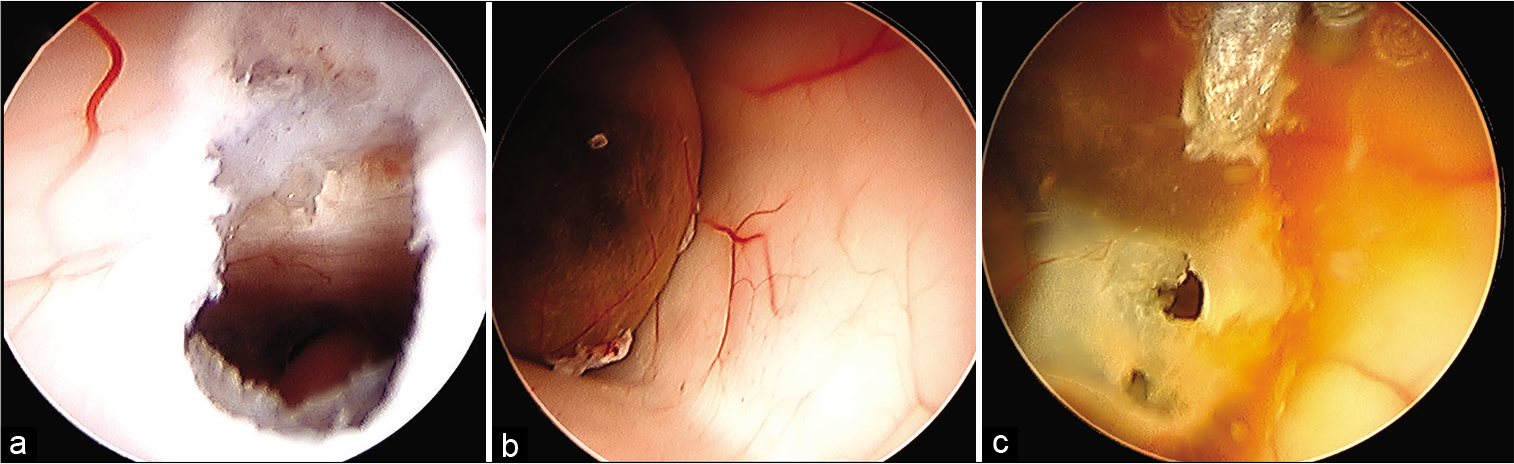
Case 1 operative endoscopic photographs. (a) Endoscopic view of third ventricle floor after ventriculostomy. The basilar artery is visible at the bottom of the field (posterior aspect of ventriculostomy), (b) Endoscopic view of the posterior third ventricle revealing a dark cystic mass that obstructs the cerebral aqueduct, (c) Coagulation of the cyst with copious debris, early cyst fenestration with cyst collapsing posteriorly, and revealing hemosiderin staining.
Figure 4:
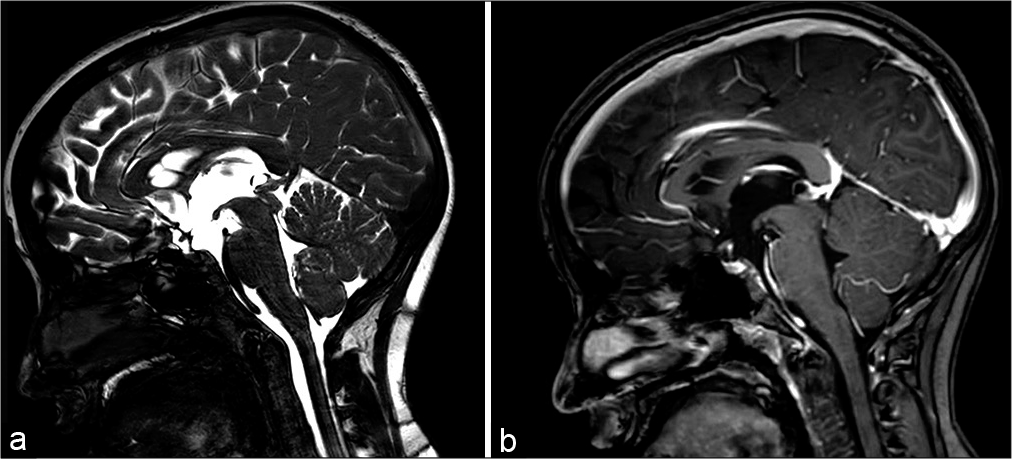
Case 2 (a) Axial noncontrast CT head showing the pineal region cyst is not discernible from the adjacent third ventricle CSF, ventriculomegaly, (b) Sagittal T1-weighted MRI image showing the pineal cyst and the superior portion of the tectum inferiorly displaced and partially occluding the superior cerebral aqueduct, (c) Endoscopic view of third ventricle floor after ventriculostomy. The basilar artery is visible, (d) Posterior third ventricle, revealing the pineal cyst that obstructs the cerebral aqueduct, (e) Fenestrated pineal cyst and the patent cerebral aqueduct, (f) 15 month postoperative sagittal T2-weighted image showing patent third ventriculostomy opening, decompressed pineal cyst, and patent cerebral aqueduct.
Supplemental Video 1
Annotations
00.32 – Using the endoscope, entering through Foramen of Monro, using the electrocautery, Fogarty balloon, and grasping forceps for the ETV.
00.42 – Exposure of the pineal cyst, using blunt and sharp electrocautery for cyst fenestration.
RESULTS
Case 1
Presentation
A 7-year-old girl with medical history of mild hereditary spherocytosis presented to the emergency department complaining of 2 days history of progressive headache associated with nausea, vomiting, confusion, and photophobia. Her initial head computed tomography (CT) [
Shortly thereafter, a right frontal ventriculostomy catheter was placed and kept open to drain at 20 cm H2O. Her mental status dramatically improved post procedurally. Both AFP and beta-HCG were undetectable in CSF, and CSF cytology was normal.
After temporary CSF diversion, we completed magnetic resonance imaging (MRI) of the entire neural axis. This revealed a mixed density 1.5 cm pineal region cyst, with dependent hemorrhagic components, and obstruction of the cerebral aqueduct [
Given the patient’s history of hereditary spherocytosis, she underwent a thorough workup led by our hematologic colleagues. Given a lack of anemia and asplenia, with only a mild reticulocytosis, and normal coagulation profile, her spherocytosis was deemed very mild and unlikely to be the cause of her pineal cyst hemorrhage.
Treatment
We considered various treatment options including craniotomy for pineal mass resection (especially with concern for possible hemorrhagic neoplastic tissue), observation of the cyst with ventriculoperitoneal shunt to manage her hydrocephalus, endoscopic third ventriculostomy, endoscopic cyst fenestration, or a combination thereof. We ultimately felt that the lesion was anatomically accessible through the Foramen of Monro for a frameless stereotactic endoscopic combined ventriculostomy and cyst fenestration with partial resection through a single entry point, a technique previously described in detail by Berhouma et al. in adults.[
Outcome
Postoperative day 1 MRI revealed a patent third ventriculostomy, a decompressed mixed-density pineal cyst, a patent cerebral aqueduct, and normalized ventricular system. The ventriculostomy catheter was removed on postoperative day 1. She discharged home on postoperative day 3. The gross and microscopic pathology analysis found cystic pineal gland with numerous hemosiderin-laden macrophages consistent with pineal cyst lining without any elements to suggest neoplasm.
MRI at 3 months follow-up revealed normalized pineal appearance with stable normal ventricular size and persistent patency of the cerebral aqueduct that was persistent at 42 months [
She remains asymptomatic, continues to do well after surgery, and demonstrates normal development more than 3 years out from surgery.
CASE 2
Presentation
A 5-year-old male with severe developmental delay, attention deficit hyperactivity disorder, and autism presented through outpatient neurosurgical clinic, becoming increasingly difficult to control with behavioral outbursts. A CT and subsequent MRI were ordered, and he was found to have ventriculomegaly compared [
Treatment
Given the patient’s young age and therefore greater potential for additional cystic expansion, CSF diversion alone might not address issues with pineal region mass-effect. We, therefore, considered strategies that would both eliminate cystic mass effect and ensure long-term normalization of CSF circulation without implants and again elected for the combined ETV and endoscopic cyst fenestration technique through a single frontal burr-hole entry point [
Outcome
The patient was discharged home on postoperative day 3. Three month and 1 year postoperative MRIs revealed a patent third ventriculostomy, a decompressed pineal cyst, a patent cerebral aqueduct, and normalized ventricular system [
Indications
The endoscopic transventricular approach to pineal cysts represent an excellent option in pediatric patients with hydrocephalus, as it allows both third ventriculostomy and marsupialization of the pineal cyst into the cavity of the third ventricle. Histological sampling is also feasible with minimal risks. Navigational planning software is helpful in determining the feasibility of a single burr-hole approach based on the individual patient’s anatomy. Considerations for use of flexible endoscopy are reasonable here as well.
Surgical technique
At the time of these procedures, we were not aware of the manuscript by Berhouma et al. that describe this approach in adult patients, but our technique was indeed similar for these pediatric patients.[
Our patients were placed supine on the operating room table. The cervical spine was placed in slight flexion and the head is secured in a Mayfield three pin head holder with pediatric pins. A soft scapular bump was used to avoid excessive flexion. Navigation is especially helpful for locating a single frontal entry point that can provide a feasible working trajectory to both the floor of the third ventricle and the anterior membrane of the obstructive cyst. It is important to consider that as soon as the cyst wall is punctured, the cyst will begin to deflate, and the anterior wall may retrude posteriorly. The ideal entry point was marked on the scalp and should be anterior to the coronal suture. The area was prepped and draped and a small linear incision was made. Consideration can be made for a burr-hole slightly larger than one would for a typical ETV; we used a 9 mm burr-hole to allow for anterior-posterior angulation to reach the two different target points during this procedure. A cruciate durotomy was performed and a gyral surface was identified and the pia coagulated.
If navigation is used already for trajectory planning, we recommend the relatively simple additional step of securing a navigation guide to the endoscope so that the tip of the endoscope can be tracked. This can be of utility if visualization is temporarily compromised at any point and helpful for precise tracking through the cortex and white matter to stay aligned with the optimal trajectory into the lateral ventricle. We first proceed with the ETV procedure. Since the entry point is slightly more anterior than for a typical ETV to optimize access to the posterior third ventricle, an angled scope may be preferable. We used a 30 degree rigid endoscope for both ETV and cyst fenestration in these cases. When ETV is satisfactorily completed, the angled scope is turned toward the posterior aspect of the ventricle and slid under the massa intermedia. While it was not necessary in either of the cases described here, partial sectioning of the massa intermedia with sharp midline dissection can provide a less obstructed view if necessary. If the cyst is not easily reachable or there is concern for tension on the Foramen of Monroe, consider use of flexible endoscopy if available or if a secondary more anteriorly located burr-hole might provide better access.
We opened the anterior border of these cysts with a combination of blunt and sharp electrocautery probes. We also applied cautery to the inferior aspect of the cyst wall, thus shrinking it posteriorly and superiorly away from the cerebral aqueduct [
The combination of a young, thin, and pediatric skull with a slightly larger (9 mm) burr-hole can increase the risk of pseudomeningocele formation. In addition to an on-lay dural allograft, we placed a titanium (case 1) and resorbable (case 2) burr-hole cover.
Technical considerations
The entry point in the calvarium is slightly anterior to the one used for classical third ventriculostomy to allow access to the posterior third ventricle without excessive tension on the Foramen of Monro. Stereotactic navigation is useful in cases, where single burr-hole is intended, especially for planning the most appropriate entry site and confirming that tension on the Foramen of Monro and fornix can be avoided. If line of site access does not appear feasible on stereotactic planning, flexible endoscopy may be an option. Performing the third ventriculostomy before fenestrating the pineal cyst is preferable to avoid any bloody pollution of the CSF that can settle easily in the region of the tuber cinereum and limit endoscopic visibility. A Fogarty balloon can be used to enlarge the ETV and/or the cyst fenestration site, especially if the cyst retrudes and can be difficult to reach with other instruments. Coagulation of the cyst wall is done with low intensity to avoid adhesion to the cyst wall and subsequent bleeding.
Potential complications
Intraventricular hemorrhage may require temporary external ventricular drainage, meningitis, cranial nerve palsy, seizure, forniceal injury, diabetes insipidus, recurrence, additional hemorrhage, or regrowth of the cyst.
DISCUSSION
There is a surprisingly small body of the literature guiding management of complicated pineal cysts in young children. Existing literature that describe management of these lesions utilize various methods from observation, craniotomy for resection or fenestration, and endoscopic techniques [
Microsurgical total excision through an supracerebellarinfratentorial approach has been described as a curative method.[
A report of four patients (one pediatric) with endoscopically treated pineal apoplexy and associated occlusive hydrocephalus due to aqueductal occlusion, without intervention for hydrocephalus, showed good results. In these cases, the cyst walls were removed as much as possible using endoscopy. The postoperative images showed persistent cyst remnants, but flow through the cerebral aqueduct was normal.[
Ventriculocisternostomy is an additional option for occlusive hydrocephalus secondary to a pineal cyst.[
In this study, we present a single burr-hole technique for ETV and cyst marsupialization using stereotactic navigation for the management of complicated pineal cysts in young children. This technique offers redundancy for hydrocephalus treatment in the growing child, permits nonaggressive management of the vascular cyst wall to prevent issues with mass effect and restore aqueductal patency, allows for tissue biopsy, minimizes the likelihood of future shunt dependence, and utilizes a skillset familiar to pediatric neurosurgeons.
CONCLUSION
Pineal cysts are typically benign incidental findings; however, complicated cysts that cause mass effect, hydrocephalus, and papilledema can be found in pediatric patients and warrant neurosurgical intervention. Pineal apoplexy with subsequent obstructive hydrocephalus is the rarest and most dangerous form with resultant acute neurological deterioration which can lead to sudden death. Most reported cases of surgically treated complicated pediatric pineal cysts discovered in this review involved apoplectic etiologies. To date, the pediatric literature on management of these cysts has been variable, but the majority of authors advocate for either total cyst excision through craniotomy or a combination of cyst surgery and hydrocephalus management. The transventricular endoscopic approach with stereotactic navigation allows simultaneous third ventriculostomy and cyst marsupialization to restore patency of the cerebral aqueduct and provides an opportunity to histologically sample the cyst wall and coagulate cyst edges to prevent further hemorrhage and minimizes the likelihood for future surgery. The patient selection to include complex cysts that project into the third ventricle and stereotactic planning to assess single burr-hole access feasibility is key.
Declaration of patient consent
Patients’ consent not required as patients’ identities were not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Alberti O, Riegel T, Hellwig D, Bertalanffy H. Frameless navigation and endoscopy. J Neurosurg. 2001. 95: 541-3
2. Al-Holou WN, Garton HJ, Muraszko KM, Ibrahim M, Maher CO. Prevalence of pineal cysts in children and young adults. Clinical article. J Neurosurg Pediatr. 2009. 4: 230-6
3. Avery GJ, Lind CR, Bok AP. Successful conservative operative management of pineal apoplexy. J Clin Neurosci. 2004. 11: 667-9
4. Berhouma M, Ni H, Vallee B. The endoscopic intraventricular management of pineal cysts: A minimally invasive modus operandi. Acta Neurochir (Wien). 2013. 155: 1901-5
5. Cooper ER. The human pineal gland and pineal cysts. J Anat. 1932. 67: 28-46
6. Davidson L. Endoscopic management of pineal cyst-associated aqueductal stenosis. Acta Neurochir (Wien). 2020. 162: 2975-82
7. Di Chirico A, Di Rocco F, Velardi F. Spontaneous regression of a symptomatic pineal cyst after endoscopic third-ventriculostomy. Childs Nerv Syst. 2001. 17: 42-6
8. Durnford AJ, Kirkham FJ, Mathad N, Sparrow OC. Endoscopic third ventriculostomy in the treatment of childhood hydrocephalus: Validation of a success score that predicts long-term outcome. J Neurosurg Pediatr. 2011. 8: 489-93
9. Fain JS, Tomlinson FH, Scheithauer BW, Parisi JE, Fletcher GP, Kelly PJ. Symptomatic glial cysts of the pineal gland. J Neurosurg. 1994. 80: 454-60
10. Kiely MJ. Neuroradiology case of the day. Pineal cyst with cerebral aqueduct obstruction. AJR Am J Roentgenol. 1993. 160: 1338-9
11. Klein P, Rubinstein LJ. Benign symptomatic glial cysts of the pineal gland: A report of seven cases and review of the literature. J Neurol Neurosurg Psychiatry. 1989. 52: 991-5
12. Koenigsberg RA, Faro S, Marino R, Turz A, Goldman W. Imaging of pineal apoplexy. Clin Imaging. 1996. 20: 91-4
13. Majeed K, Enam SA. Recurrent pineal apoplexy in a child. Neurology. 2007. 69: 112-4
14. Mamourian AC, Towfighi J. Pineal cysts: MR imaging. AJNR Am J Neuroradiol. 1986. 7: 1081-6
15. Mandera M, Marcol W, Bierzyńska-Macyszyn G, Kluczewska E. Pineal cysts in childhood. Childs Nerv Syst. 2003. 19: 750-5
16. McNeely PD, Howes WJ, Mehta V. Pineal apoplexy: Is it a facilitator for the development of pineal cysts?. Can J Neurol Sci. 2003. 30: 67-71
17. Metellus P, Fuentes S, Levrier O, Adetchessi T, Dufour H, Donnet A. Endoscopic treatment of a voluminous benign symptomatic cyst of the pineal region responsible for an obstructive hydrocephalus. Neurochirurgie. 2005. 51: 173-8
18. Michielsen G, Benoit Y, Baert E, Meire F, Caemaert J. Symptomatic pineal cysts: Clinical manifestations and management. Acta Neurochir (Wien). 2002. 144: 233-42
19. Mukherjee KK, Banerji D, Sharma R. Pineal cyst presenting with intracystic and subarachnoid haemorrhage: Report of a case and review of the literature. Br J Neurosurg. 1999. 13: 189-92
20. Musolino A, Cambria S, Rizzo G, Cambria M. Symptomatic cysts of the pineal gland: Stereotactic diagnosis and treatment of two cases and review of the literature. Neurosurgery. 1993. 32: 315-20
21. Nimmagadda A, Sandberg DI, Ragheb J. Spontaneous involution of a large pineal region hemorrhagic cyst in an infant: Case report. J Neurosurg. 2006. 104: 275-8
22. Sarikaya-Seiwert S, Turowski B, Hänggi D, Janssen G, Steiger HJ, Stummer W. Symptomatic intracystic hemorrhage in pineal cysts. Report of 3 cases. J Neurosurg Pediatr. 2009. 4: 130-6
23. Sawamura Y, Ikeda J, Ozawa M, Minoshima Y, Saito H, Abe H. Magnetic resonance images reveal a high incidence of asymptomatic pineal cysts in young women. Neurosurgery. 1995. 37: 11-5
24. Schroeder HW, Wagner W, Tschiltschke W, Gaab MR. Frameless neuronavigation in intracranial endoscopic neurosurgery. J Neurosurg. 2001. 94: 72-9
25. Swaroop GR, Whittle IR. Pineal apoplexy: An occurrence with no diagnostic clinicopathological features. Br J Neurosurg. 1998. 12: 274-6
26. Tamura Y, Yamada Y, Tucker A, Ukita T, Tsuji M, Miyake H. Endoscopic surgery for hemorrhagic pineal cyst following antiplatelet therapy: Case report. Neurol Med Chir (Tokyo). 2013. 53: 625-9
27. Wisoff JH, Epstein F. Surgical management of symptomatic pineal cysts. J Neurosurg. 1992. 77: 896-900