- Department of Neurosurgery, Gandaki Medical College, Pokhara, Gandaki, Nepal.
- School of Medicine, Gandaki Medical College, Pokhara, Gandaki, Nepal.
Correspondence Address:
Mitesh Karn, School of Medicine, Gandaki Medical College, Pokhara, Gandaki, Nepal.
DOI:10.25259/SNI_310_2021
Copyright: © 2021 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Shabal Sapkota1, Mitesh Karn2. Extradural abscess following synthetic fabric duraplasty. 25-May-2021;12:234
How to cite this URL: Shabal Sapkota1, Mitesh Karn2. Extradural abscess following synthetic fabric duraplasty. 25-May-2021;12:234. Available from: https://surgicalneurologyint.com/surgicalint-articles/10826/
Abstract
Background: Duraplasty refers to the neurosurgical process of reconstructing dural defect. Variety of materials is used for such reconstruction, including natural, semisynthetic, and synthetic materials. Although synthetic materials are readily available and easy to apply, these are associated with foreign body reaction which may lead to serious consequences in some cases. We describe one such rare instance of extradural abscess after polypropylene synthetic fabric duraplasty.
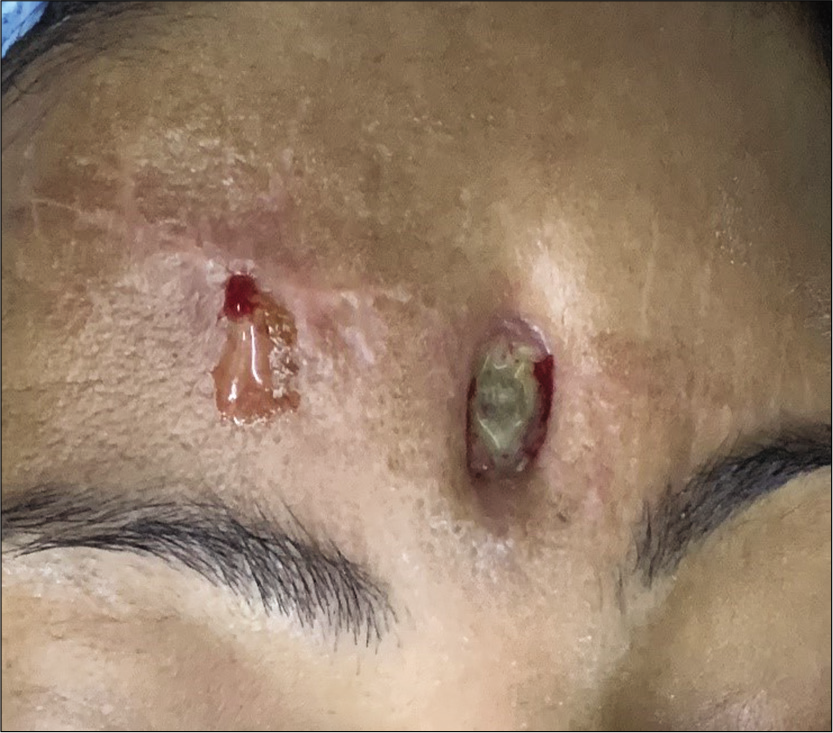
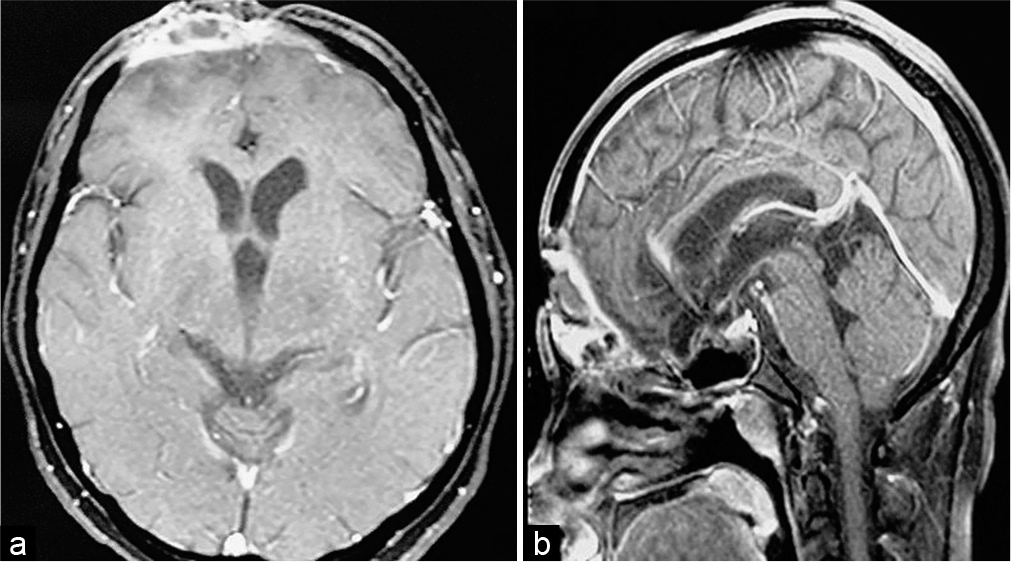
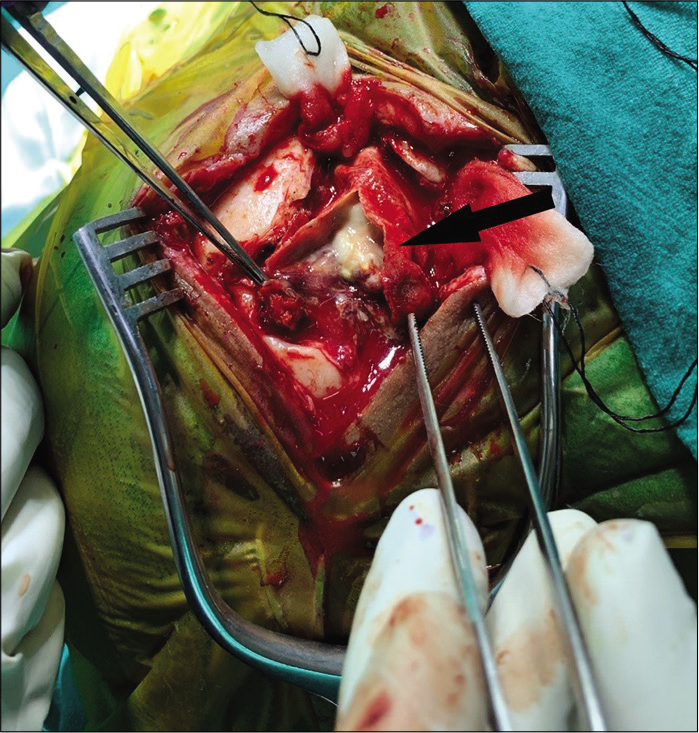
Case Description: Our patient is a 33-year-old lady who suffered road traffic accident leading to massive brain laceration, contusion of bilateral frontal lobes, and anterior skull base fractures. Emergency craniotomy was carried out and dural defect repaired with polypropylene (G-Patch; G. Surgiwear® Ltd.) synthetic fabric as the duraplasty material. Three months later, the patient presented with discharging wound at the incision site. Neuroimaging showed ring enhancing lesion in frontobasal extradural space with cutaneous extension. The lesion failed to heal despite intravenous antibiotics and surgery was planned. Intraoperatively, abscess was found between G-Patch and dura. Histopathology showed granulomatous foreign body reaction. The lesion healed after synthetic dura removal and abscess drainage.
Conclusion: Although various materials are used for duraplasty, there is no clear consensus on what material should be used for dural repair. Synthetic materials are bio-inert, offer good handling and malleability. Polypropylene has been used safely for both single- and double-layered duraplasty. However, foreign body reaction may occur and very rarely present as extradural abscess. Randomized trials should be done to establish the safety and efficacy profile of commonly used duraplasty materials.
Keywords: Epidural abscess, Foreign body reaction, Polypropylenes
INTRODUCTION
Duraplasty is a neurosurgical intervention that involves application of biological or synthetic graft over an area where the dural integrity has been compromised.[
We, herein, describe a rare case of an extradural abscess presenting as a discharging forehead sinus after polypropylene (G-Patch®) synthetic fabric duraplasty.
CASE DESCRIPTION
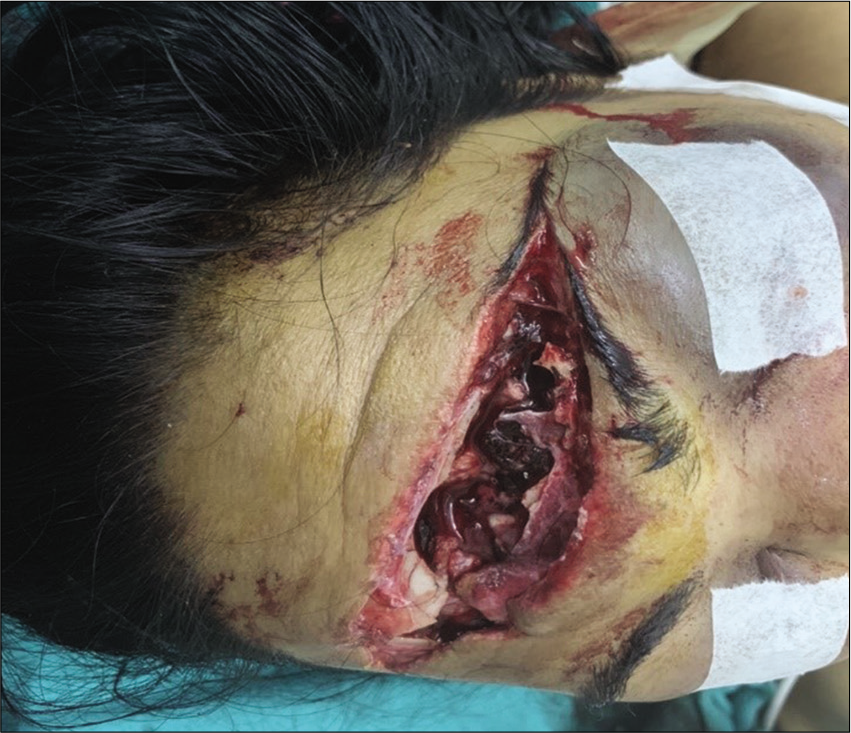
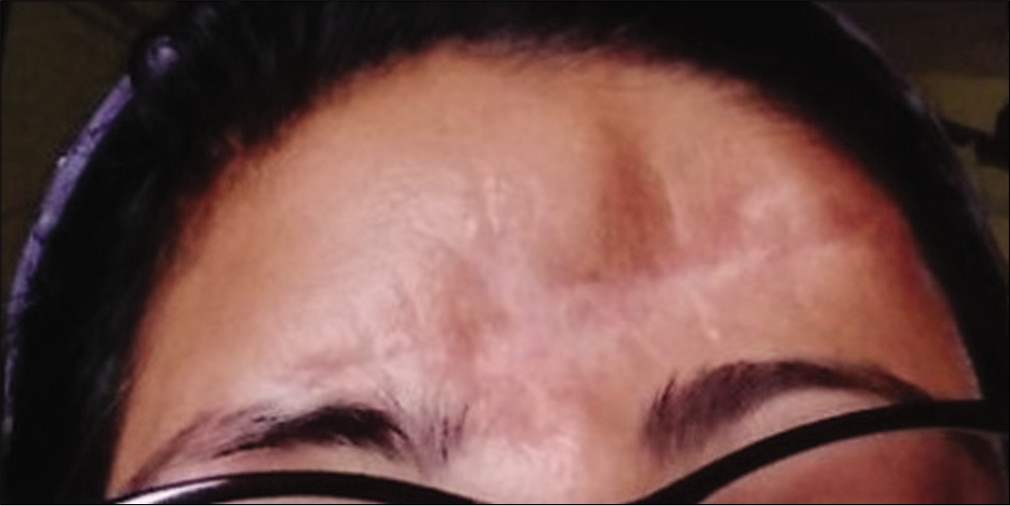
A 33-year-old female and her spouse were transferred to the emergency department of our teaching hospital after sustaining road traffic accident, when their motorbike was allegedly hit by a truck. The husband, who was in the driving seat, was pronounced dead immediately on arrival. The woman, who was a pillion rider, sustained severe head injuries with massive brain laceration and gross extrusion of brain tissues from the forehead [
DISCUSSION
In 1895, Robert Abbe described the use of sterilized rubber for closure of dural defect created after removing adhesions following pachymeningitis. Ever since, a variety of natural and synthetic materials has been used for duraplasty. Regardless of the material used, major aims of using dural substitute are to achieve watertight closure, provide surface for neodura formation,, and preventing infection.[
G-Patch (G. Surgiwear® Limited, India) is a synthetic polypropylene fabric patch used popularly as a dural substitute in Nepal and India. Polypropylene has been shown to induce very less inflammatory reaction and is relatively resistant to infections and biological degradation when used for inguinal and hiatal hernia repair.[
The use of both autologous and nonautologous materials for duraplasty in cranial surgery is associated with similar complication rates.[
CONCLUSION
Complication rates with various types of material used for duraplasty are similar. The choice of material used depends on the local protocol and individual surgeon preference. Although polypropylene has been used safely as both single-and double-layered duraplasty, it may trigger foreign body reaction and present rarely as an extradural abscess. This highlights the importance of continued postprocedural patient follow-up as late complications can occur. Since very less prospective studies have been carried out comparing different dural substitutes; large, randomized, multicentric studies should be carried out to establish the superiority among available materials. This evidence may help immensely in deciding which material should be used for duraplasty.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Publication of this article was made possible by the James I. and Carolyn R. Ausman Educational Foundation.
Conflicts of interest
There are no conflicts of interest.
References
1. Abla AA, Link T, Fusco D, Wilson DA, Sonntag VK. Comparison of dural grafts in Chiari decompression surgery: Review of the literature. J Craniovertebr Junction Spine. 2010. 1: 29-37
2. Akhaddar A, Turgut AT, Turgut M. Foreign body granuloma after cranial surgery: A systematic review of reported cases. World Neurosurg. 2018. 120: 457-75
3. Azzam D, Romiyo P, Nguyen T, Sheppard JP, Alkhalid Y, Lagman C. Dural repair in cranial surgery is associated with moderate rates of complications with both autologous and nonautologous dural substitutes. World Neurosurg. 2018. 113: 244-8
4. Esposito F, Cappabianca P, Fusco M, Cavallo LM, Bani GG, Biroli F. Collagen-only biomatrix as a novel dural substitute. Examination of the efficacy, safety and outcome: Clinical experience on a series of 208 patients. Clin Neurol Neurosurg. 2008. 110: 343-51
5. Kamalabai RP, Nagar M, Chandran R, Suharanbeevi SM, Prabhakar RB, Peethambaran A. Rationale behind the use of double-layer polypropylene patch (G-patch) dural substitute during decompressive craniectomy as an adhesion preventive material for subsequent cranioplasty with special reference to flap elevation time. World Neurosurg. 2018. 111: e105-12
6. Malliti M, Page P, Gury C, Chomette E, Nataf F, Roux FX. Comparison of deep wound infection rates using a synthetic dural substitute (neuropatch) or pericranium graft for dural closure: A clinical review of 1 year. Neurosurgery. 2004. 54: 599-604
7. Warren WL, Medary MB, Dureza CD, Bellotte JB, Flannagan PP, Oh MY. Dural repair using acellular human dermis: Experience with 200 cases: Technique assessment. Neurosurgery. 2000. 46: 1391-6
8. Wethington A, Gujjula N, Chamczuk A, Vivekanandan R. Bovine pericardium duraplasty: Epidural abscess as a rare complication. Interdiscip Neurosurg Adv Tech Case Manag. 2016. 6: 18-9
9. Wood AJ, Cozad MJ, Grant DA, Ostdiek AM, Bachman SL, Grant SA. Materials characterization and histological analysis of explanted polypropylene, PTFE, and PET hernia meshes from an individual patient. J Mater Sci Mater Med. 2013. 24: 1113-22