- Gamma Knife Center Cairo, Cairo, Egypt
- Department of Neurosurgery, Faculty of Medicine, Ain Shams University, Cairo, Egypt
- Department of Clinical Oncology, Faculty of Medicine, Ain Shams University, Cairo, Egypt
- Department of Radiation Oncology, National Cancer Institute, Cairo University, Cairo, Egypt
- Department of Neurosurgery, Faculty of Medicine, Benha University, Qalubya, Egypt
Correspondence Address:
Amr M. N. El-Shehaby
Gamma Knife Center Cairo, Cairo, Egypt
Department of Neurosurgery, Faculty of Medicine, Benha University, Qalubya, Egypt
DOI:10.4103/sni.sni_206_17
Copyright: © 2017 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Amr M. N. El-Shehaby, Wael A. Reda, Khaled M. Abdel Karim, Reem M. Emad Eldin, Ahmed M. Nabeel. Gamma knife radiosurgery for cerebellopontine angle epidermoid tumors. 24-Oct-2017;8:258
How to cite this URL: Amr M. N. El-Shehaby, Wael A. Reda, Khaled M. Abdel Karim, Reem M. Emad Eldin, Ahmed M. Nabeel. Gamma knife radiosurgery for cerebellopontine angle epidermoid tumors. 24-Oct-2017;8:258. Available from: http://surgicalneurologyint.com/surgicalint-articles/gamma-knife-radiosurgery-for-cerebellopontine-angle-epidermoid-tumors/
Abstract
Background:Intracranial epidermoid tumors are commonly found in the cerebellopontine angle where they usually present with either trigeminal neuralgia or hemifacial spasm. Radiosurgery for these tumors has rarely been reported. The purpose of this study is to assess the safety and clinical outcome of the treatment of cerebellopontine epidermoid tumors with gamma knife radiosurgery.
Methods:This is a retrospective study involving 12 patients harboring cerebellopontine angle epidermoid tumors who underwent 15 sessions of gamma knife radiosurgery. Trigeminal pain was present in 8 patients and hemifacial spasm in 3 patients. All cases with trigeminal pain were receiving medication and still uncontrolled. One patient with hemifacial spasm was medically controlled before gamma knife and the other two were not. Two patients had undergone surgical resection prior to gamma knife treatment. The median prescription dose was 11 Gy (10–11 Gy). The tumor volumes ranged from 3.7 to 23.9 cc (median 10.5 cc).
Results:The median radiological follow up was 2 years (1–5 years). All tumors were controlled and one tumor shrank. The median clinical follow-up was 5 years. The trigeminal pain improved or disappeared in 5 patients, and of these, 4 cases stopped their medication and one decreased it. The hemifacial spasm resolved in 2 patients who were able to stop their medication. Facial palsy developed in 1 patient and improved with conservative treatment. Transient diplopia was also reported in 2 cases.
Conclusion:Gamma knife radiosurgery provides good clinical control for cerebellopontine angle epidermoid tumors.
Keywords: Cerebellopontine, epidermoid, gamma knife, radiosurgery
INTRODUCTION
Intracranial epidermoid tumors are commonly found in the cerebellopontine angle,[
Recently, radiosurgery has emerged as a treatment option for cerebellopontine epidermoid tumors. Very few have reported on the radiosurgical treatment of these tumors. The purpose of this study was to assess the safety and clinical outcome of the treatment of cerebellopontine epidermoid tumors with gamma knife radiosurgery.
PATIENTS AND METHODS
This is a retrospective study involving 12 patients harboring cerebellopontine angle epidermoid tumors that were treated between May 2008 and November 2013. A single session was conducted in 9 patients. Volume-staged radiosurgery was done in 3 patients due to large tumor size; it was a two-staged treatment in these 3 patients. The interval between the treatment stages was 3 months.
The Leksell stereotactic head frame was attached to the patient's head using local anesthesia (model G, Elekta AB). Imaging was based on contrast-enhanced T1-weighted sequences plus T2-weighted magnetic resonance (MR) sequences with 1.6-mm slice thickness using high-resolution 1.5-T magnetic resonance imaging (MRI) (Genesis Sigma, General Electric). Stereotactic images were imported into the GammaPlan workstation (Elekta AB). The treatment was carried out using the Gamma Knife Model C and Gamma Knife PERFEXION (Elekta Instruments, Inc.). The target volume was drawn in all the MRI slices. We selected a prescription dose of 11 Gy or less to keep the dose to the adjacent brainstem below the 12 Gy toxicity threshold.
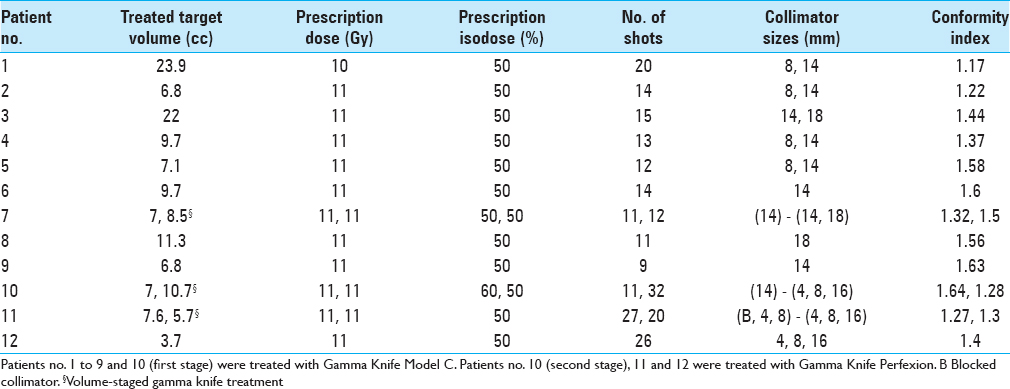
The median prescription dose was 11 Gy (10–11 Gy). The tumor volumes ranged from 3.7 to 23.9 cc (median 10.5 cc). The median treated target volume (i.e., the target volume/treatment) was 7.6 cc, taking in consideration the target volumes in the staged treatment cases [
Imaging follow-up examinations using contrast-enhanced MRI were carried out at 6-monthly intervals for the first 2 years and then annually thereafter. Additional imaging was obtained when a patient developed new symptoms or experienced worsening of any preexisting symptoms.
Every patient's history and clinical examination findings were recorded and compared with those documented prior to treatment. Radiological follow-up was undertaken by performing contrast-enhanced MRI. In addition to imaging, an audiometry with speech discrimination score was performed at every follow-up to assess the hearing status due to the presence of the hearing apparatus (cochlear nerve and cochlea) in the tumor vicinity.
The trigeminal pain after gamma knife treatment at the last clinical follow-up was assessed and classified according to the Barrow Neurosurgical Institute (BNI) score.[
RESULTS
Patient demographics
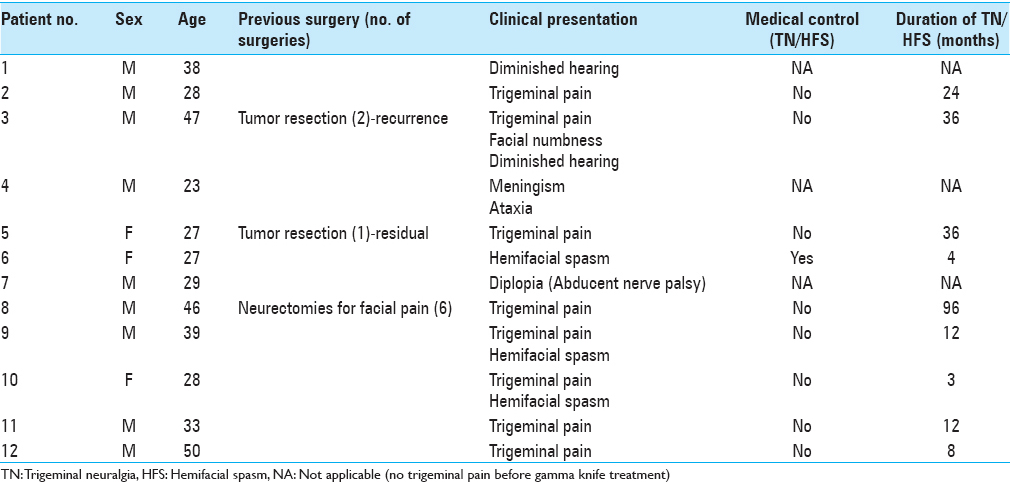
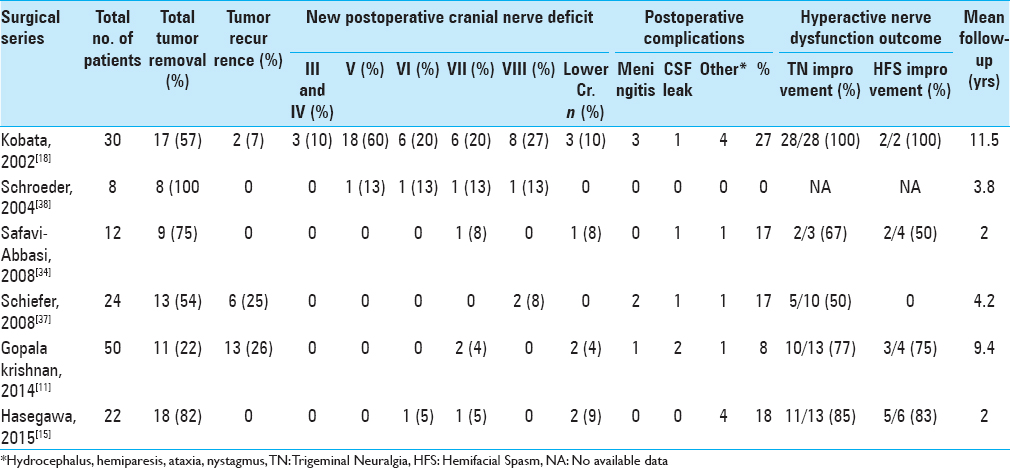
The patients were 23–50 years (median 35 years) of age. There were 9 males and 3 females. Two patients underwent previous surgery. One had been operated twice and recurred for the third time (the first time after 2 years and the second after 3 years). The other had a residual tumor after surgery. The remaining 10 patients were diagnosed based on MRI radiological features. Hyperactive cranial nerve dysfunction (trigeminal neuralgia or hemifacial spasm) was present in 9 patients (9/12, 75%). This was in the form of trigeminal pain in 8 patients (8/12, 67%) and hemifacial spasm in 3 patients (3/12, 25%). Among these 9 patients, 2 had combined trigeminal pain and hemifacial spam (2/12, 17%) and 1 patient had hemifacial spasm only (1/12, 8%). All cases with trigeminal pain were receiving medication and still uncontrolled. One patient with hemifacial spasm was medically controlled before gamma knife and the other two were not [
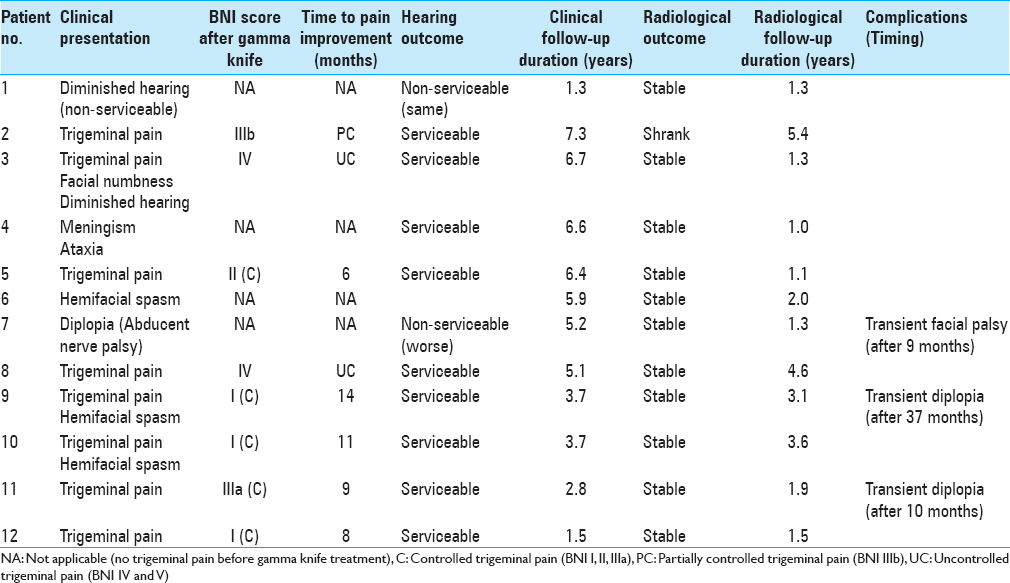
Treatment outcome [ Table 3 ]
Hyperactive cranial nerve dysfunction improved or resolved in 5 out of 9 patients (56%). The trigeminal pain was controlled in 5 out of 8 (63%) patients, and of these, 4 cases stopped their medication and 1 decreased it. The hemifacial spasm resolved in 2 of 3 (67%) patients who were able to stop their medication. The two patients who had combined trigeminal pain and hemifacial spasm showed resolution of their symptoms and were able to stop all medication. The median time to pain improvement was 9 months (6–14 months).
Trigeminal pain was partially controlled in 1 patient and was uncontrolled in 2 patients. The duration of symptoms before treatment was 2–8 years. The first patient had initial pain improvement at 6 months which recurred at 1 year (BNI IIIb) [
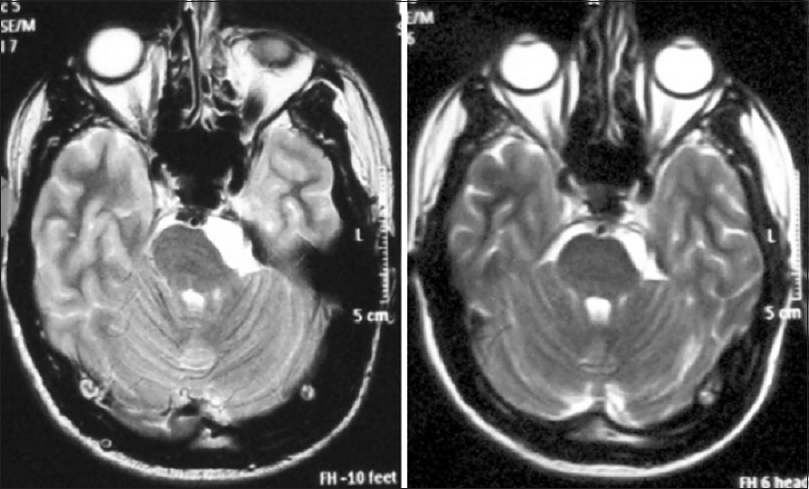
Figure 1
A 28-year-old male presented with left V2, V3 trigeminal neuralgia (BNI IV) for 2 years which had become unresponsive to medication. MRI T2-weighted image (left) showed he had a left cerebellopontine angle epidermoid 6.8 cc in volume. The tumor was treated with 11 Gy to the 50% isodose. At 6-month follow-up, the tumor shrank and the patient's trigeminal pain improved (BNI IIIa) and he was able to decrease his medication. One year later, however, his trigeminal pain had recurred (BNI IIIb). At 5 years, the tumor was still under control (right)
Hemifacial spasm was uncontrolled in 1 patient. He presented 4 months after onset of his symptoms. There was initial improvement of spasm at 6 months and was even able to stop his medication. The hemifacial spasm recurred at 18 months.
Ten patients had serviceable hearing before treatment. All 10 patients retained serviceable hearing after treatment. One patient who had nonserviceable hearing before treatment developed further worsening of his hearing and eventually became deaf at 2-year follow-up. This was a patient who had a large tumor that was treated by volume-staging. All tumors were radiologically controlled. One tumor shrank and the others remained stable.
Complications
One patient developed transient facial nerve palsy (House-Brackmann Grade 3) at 8 months after treatment. It improved with conservative treatment to Grade 2 approximately 6 months later. Transient diplopia developed in 2 patients 10 and 37 months after treatment.
DISCUSSION
Epidermoid tumors are typically benign lesions that commonly occur in cerebellopontine angle. They usually present with hyperactive cranial nerve dysfunction, mainly trigeminal neuralgia and hemifacial spasm. The incidence of trigeminal neuralgia in patients with cerebellopontine epidermoid tumors is reported to vary from 0 to 77%,[
Surgery
Surgery remains the primary treatment option of cerebellopontine angle epidermoid tumors. The controversy has been whether to proceed with radical tumor resection or subtotal/near total resection. Radical resection should involve removal of the tumor capsule that has tumor cells, which desquamate giving rise to the cyst contents.[
Previous studies have reported recurrence of epidermoid tumors to occur from 1 year to several years after surgery in up to 26% of the cases, even with complete tumor removal.[
Gamma Knife results
Gamma knife radiosurgery for cerebellopontine angle epidermoid tumors has rarely been reported. Kida et al.[
Previous gamma knife reports have indicated complete trigeminal pain or hemifacial spasm relief or improvement in all treated cases.[
Radiological tumor control was reported in all cases in the current series as with other series in all cases.[
Complications
Kida et al. reported 1 patient who developed permanent diplopia and another who had hearing loss after gamma knife treatment. In the current study, we had two cases of transient diplopia and one case of hearing loss. We reported one case of facial nerve palsy after treatment, which was not observed in any of the previous series. In this case, it was a transient occurrence and facial nerve function eventually improved. This was unusual because of the lower dose used compared to the other studies. There is the possibility that the patient had undergone volume-staged treatment so the facial nerve supposedly received radiation twice. Yet, no facial nerve dysfunction was observed in the other two volume-staged cases. In our opinion, it may be related to individual patient radiosensitivity, the length of facial nerve irradiated in the cerebellopontine angle, or the position of the hotspot inside the tumor.
Surgical series have reported the incidence of postoperative facial nerve palsy to be up to 20%,[
Study limitations
The main limitation of this study is the relatively short radiological follow-up compared to similar studies. The short follow-up does not allow differentiation from natural history. However, we could clinically follow-up all the patients by phone for a relatively long-term period to establish clinical efficacy.
Although surgery remains the gold standard for the treatment of cerebellopontine angle epidermoid tumors, we suggest gamma knife may have a role in small symptomatic tumors, residual tumors, or as an alternative when surgery cannot be performed.
CONCLUSION
Gamma knife radiosurgery provides long-term symptomatic relief for the hyperactive nerve dysfunction associated with these tumors. The minimal and acceptable complications make gamma knife radiosurgery a safe treatment option for cerebellopontine angle epidermoid tumors. Future prospective long-term studies should be conducted, preferably with controls, to further substantiate the efficacy.
Disclosure
The authors report no conflict of interest concerning the materials or methods used in this study or the findings specified in this paper.
Financial support and sponsorship
Nil.
Conflicts of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
References
1. Abramson RC, Morawetz RB, Schlitt M. Multiple Complications from an Intracranial Epidermoid Cyst: Case Report and Literature Review. Neurosurgery. 1989. 24: 574-8
2. Ahmed I, Auguste KI, Vachhrajani S, Dirks PB, Drake JM, Rutka JT. Neurosurgical Management of Intracranial Epidermoid Tumors in Children. Clinical Article. J Neurosurg Pediatr. 2009. 4: 91-6
3. Altschuler EM, Jungreis CA, Sekhar LN, Jannetta PJ, Sheptak PE. Operative Treatment of Intracranial Epidermoid Cysts and Cholesterol Granulomas: Report of 21 Cases. Neurosurgery. 1990. 26: 606-13
4. Alvord EC. Growth Rates of Epidermoid Tumors. Ann Neurol. 1977. 2: 367-70
5. Auger RG, Piepgras DG. Hemifacial Spasm Associated with Epidermoid Tumors of the Cerebellopontine Angle. Neurology. 1989. 39: 577-80
6. Barker FG, Jannetta PJ, Babu RP, Pomonis S, Bissonette DJ, Jho HD. Long-Term Outcome after Operation for Trigeminal Neuralgia in Patients with Posterior Fossa Tumors. J Neurosurg. 1996. 84: 818-25
7. Berger MS, Wilson CB. Epidermoid Cysts of the Posterior Fossa. J Neurosurg. 1985. 62: 214-9
8. deSouza CE, deSouza R, da Costa S, Sperling N, Yoon TH, Abdelhamid MM, Sharma RR, Goel A. Cerebellopontine Angle Epidermoid Cysts: A Report on 30 Cases. J Neurol Neurosurg Psychiatry. 1989. 52: 986-90
9. Fox H, South EA. Squamous Cell Carcinoma Developing in an Intracranial Epidermoid Cyst (Cholesteatoma). J Neurol Neurosurg Psychiatry. 1965. 28: 276-81
10. Gardner WJ. Trigeminal Neuralgia. Clin Neurosurg. 1968. 15: 1-56
11. Gopalakrishnan CV, Ansari KA, Nair S, Menon G. Long Term Outcome in Surgically Treated Posterior Fossa Epidermoids. Clin Neurol Neurosurg. 2014. 117: 93-9
12. Guidetti B, Gagliardi FM. Epidermoid and Dermoid Cysts. Clinical Evaluation and Late Surgical Results. J Neurosurg. 1977. 47: 12-8
13. Haig PV. Primary Epidermoids of the Skull Including a Case with Malignant Change. Am J Roentgenol Radium Ther Nucl Med. 1956. 76: 1076-80
14. Hamel E, Frowein RA, Karimi-Nejad A. Intracranial Intradural Epidermoids and Dermoids. Surgical Results of 38 Cases. Neurosurg Rev. 1980. 3: 215-9
15. Hasegawa M, Nouri M, Nagahisa S, Yoshida K, Adachi K, Inamasu J. Cerebellopontine Angle Epidermoid Cysts: Clinical Presentations and Surgical Outcome. Neurosurg Rev. 2016. 39: 259-
16. Iwasaki K, Kondo A, Otsuka S, Hasegawa K, Ohbayashi T. Painful Tic Convulsif Caused by a Brain Tumor: Case Report and Review of the Literature. Neurosurgery. 1992. 30: 916-9
17. Kida Y, Yoshimoto M, Hasegawa T, Fujitani S. [Radiosurgery of Epidermoid Tumors with Gamma Knife: Possiblility of Radiosurgical Nerve Decompression]. No Shinkei Geka. 2006. 34: 375-81
18. Kobata H, Kondo A, Iwasaki K. Cerebellopontine Angle Epidermoids Presenting with Cranial Nerve Hyperactive Dysfunction: Pathogenesis and Long-Term Surgical Results in 30 Patients. Neurosurgery. 2002. 50: 276-
19. Lewis AJ, Cooper PW, Kassel EE, Schwartz ML. Squamous Cell Carcinoma Arising in a Suprasellar Epidermoid Cyst. Case Report. J Neurosurg. 1983. 59: 538-41
20. Lunardi P, Missori P, Gagliardi FM, Fortuna A. Long-Term Results of the Surgical Treatment of Spinal Dermoid and Epidermoid Tumors. Neurosurgery. 1989. 25: 860-4
21. Maccarty CS, Leavens ME, Love JG, Kernohan JW. Dermoid and Epidermoid Tumors in the Central Nervous System of Adults. Surg Gynecol Obstet. 1959. 108: 191-8
22. Mohanty A, Venkatrama SK, Rao BR, Chandramouli BA, Jayakumar PN, Das BS. Experience with Cerebellopontine Angle Epidermoids. Neurosurgery. 1997. 40: 24-
23. Nagata S, Matsushima T, Fujii K, Fukui M, Kuromatsu C. Hemifacial Spasm Due to Tumor, Aneurysm, or Arteriovenous Malformation. Surg Neurol. 1992. 38: 204-9
24. Netsky MG. Epidermoid Tumors. Review of the Literature. Surg Neurol. 1988. 29: 477-83
25. Obrador S, Lopez-Zafra JJ. Clinical Features of the Epidermoids of the Basal Cisterns of the Brain. J Neurol Neurosurg Psychiatry. 1969. 32: 450-4
26. Otsuka S, Nakatsu S, Matsumoto S, Sato S, Motozaki T, Ban S. Epidermoid Tumor Presenting with Trigeminal Neuralgia and Ipsilateral Hemifacial Spasm: A Case Report. Nihon Geka Hokan. 1989. 58: 245-9
27. Parikh S, Milosevic M, Wong CS, Laperriere N. Recurrent Intracranial Epidermoid Cyst Treated with Radiotherapy. J Neurooncol. 1995. 24: 293-7
28. Pikis S, Margolin E. Malignant Transformation of a Residual Cerebellopontine Angle Epidermoid Cyst. J Clin Neurosci. 2016. p.
29. Regis J. Radiosurgery as Neuromodulation Therapy!. Acta Neurochir Suppl. 2013. 116: 121-6
30. Rogers CL, Shetter AG, Fiedler JA, Smith KA, Han PP, Speiser BL. Gamma Knife Radiosurgery for Trigeminal Neuralgia: The Initial Experience of the Barrow Neurological Institute. Int J Radiat Oncol Biol Phys. 2000. 47: 1013-9
31. Rothberg S, Crounse RG, Lee JL. Glycine-C-14-Incorporation into the Proteins of Normal Stratum Corneum and the Abnormal Straum Corneum of Psoriasis. J Invest Dermatol. 1961. 37: 497-505
32. Rubin G, Scienza R, Pasqualin A, Rosta L, Da Pian R. Craniocerebral Epidermoids and Dermoids. A Review of 44 Cases. Acta Neurochir (Wien). 1989. 97: 1-16
33. Sabin HI, Bordi LT, Symon L. Epidermoid Cysts and Cholesterol Granulomas Centered on the Posterior Fossa: Twenty Years of Diagnosis and Management. Neurosurgery. 1987. 21: 798-805
34. Safavi-Abbasi S, Di Rocco F, Bambakidis N, Talley MC, Gharabaghi A, Luedemann W. Has Management of Epidermoid Tumors of the Cerebellopontine Angle Improved? A Surgical Synopsis of the Past and Present. Skull Base. 2008. 18: 85-98
35. Salazar J, Vaquero J, Saucedo G, Bravo G. Posterior Fossa Epidermoid Cysts. Acta Neurochir (Wien). 1987. 85: 34-9
36. Samii M, Tatagiba M, Piquer J, Carvalho GA. Surgical Treatment of Epidermoid Cysts of the Cerebellopontine Angle. J Neurosurg. 1996. 84: 14-9
37. Schiefer TK, Link MJ. Epidermoids of the Cerebellopontine Angle: A 20-Year Experience. Surg Neurol. 2008. 70: 584-90
38. Schroeder HW, Oertel J, Gaab MR. Endoscope-Assisted Microsurgical Resection of Epidermoid Tumors of the Cerebellopontine Angle. J Neurosurg. 2004. 101: 227-32
39. Talacchi A, Sala F, Alessandrini F, Turazzi S, Bricolo A. Assessment and Surgical Management of Posterior Fossa Epidermoid Tumors: Report of 28 Cases. Neurosurgery. 1998. 42: 242-51
40. Tartaro S, Stroffolini F, Lepore M. The Importance of Etiology for the Successful Treatment of Secondary Trigeminal Neuralgia. Reviews of Some Clinical Cases. J Med. 1979. 10: 121-8
41. Vasquez JA, Fonnegra JR, Diez JC, Fonnegra A. Treatment of Epidermoid Tumors with Gamma Knife Radiosurgery: Case Series. Surg Neurol Int. 2016. 7: S116-20
42. Vinchon M, Pertuzon B, Lejeune JP, Assaker R, Pruvo JP, Christiaens JL. Intradural Epidermoid Cysts of the Cerebellopontine Angle: Diagnosis and Surgery. Neurosurgery. 1995. 36: 52-
43. Yamakawa K, Shitara N, Genka S, Manaka S, Takakura K. Clinical Course and Surgical Prognosis of 33 Cases of Intracranial Epidermoid Tumors. Neurosurgery. 1989. 24: 568-73
44. Yasargil MG, Abernathey CD, Sarioglu AC. Microneurosurgical Treatment of Intracranial Dermoid and Epidermoid Tumors. Neurosurgery. 1989. 24: 561-7
45. Yawn RJ, Patel NS, Driscoll CL, Link MJ, Haynes DS, Wanna GB. Primary Epidermoid Tumors of the Cerebellopontine Angle: A Review of 47 Cases. Otol Neurotol. 2016. 37: 951-5