- Department of Orthopedics, Division of Spine Services, Indian Spinal Injuries Centre, New Delhi, India.
DOI:10.25259/SNI_518_2020
Copyright: © 2021 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Rakesh Miryala, Nandan Marathe, Abhinandan Reddy Mallepally, Kalidutta Das, Bibhudendu Mohapatra. Iatrogenic postoperative spondylodiscitis attributed to Burkholderia cepacia infection in an immunocompetent patient. 08-Apr-2021;12:138
How to cite this URL: Rakesh Miryala, Nandan Marathe, Abhinandan Reddy Mallepally, Kalidutta Das, Bibhudendu Mohapatra. Iatrogenic postoperative spondylodiscitis attributed to Burkholderia cepacia infection in an immunocompetent patient. 08-Apr-2021;12:138. Available from: https://surgicalneurologyint.com/surgicalint-articles/10705/
Abstract
Background: Pyogenic spondylodiscitis (PS) is a rare infection involving the intervertebral disk space, adjacent vertebral endplates, and vertebral bodies. PS occurs in the elderly and immunocompromised patients, and is an uncommon cause of initial and/or postoperative PS. There are only seven cases involving this organism reported in literature.
Case Description: Here, we present a 35-year-old male who following a lumbar discectomy developed a postoperative iatrogenic PS uniquely attributed to Burkholderia cepacia. The patient was successfully managed with postoperative surgical debridement and antibiotic therapy.
Conclusion: Rarely, B. cepacia may be the offending organism resulting in a postoperative lumbar PS.
Keywords: Burkholderia cepacia, Iatrogenic, Immunocompetent, Spondylodiscitis
INTRODUCTION
Pyogenic spondylodiscitis (PS) is a rare infection involving the intervertebral disk space, its adjacent endplates and vertebral bodies. Although PS is common in elderly and immunocompromised patients, it can also occur after lumber discectomy.[
CASE DESCRIPTION
A 35-year-old male presented with low back pain of 6 months’ duration which radiated into both lower limbs. His clinical examination revealed motor deficit (MRC 3/5) in the L4 and L5 distributions, with accompanying decreased sensations (50% loss).
Diagnostic studies
X-rays revealed degenerative changes in the lower lumbar spine with evidence of instability at L4-5 on dynamic imaging [
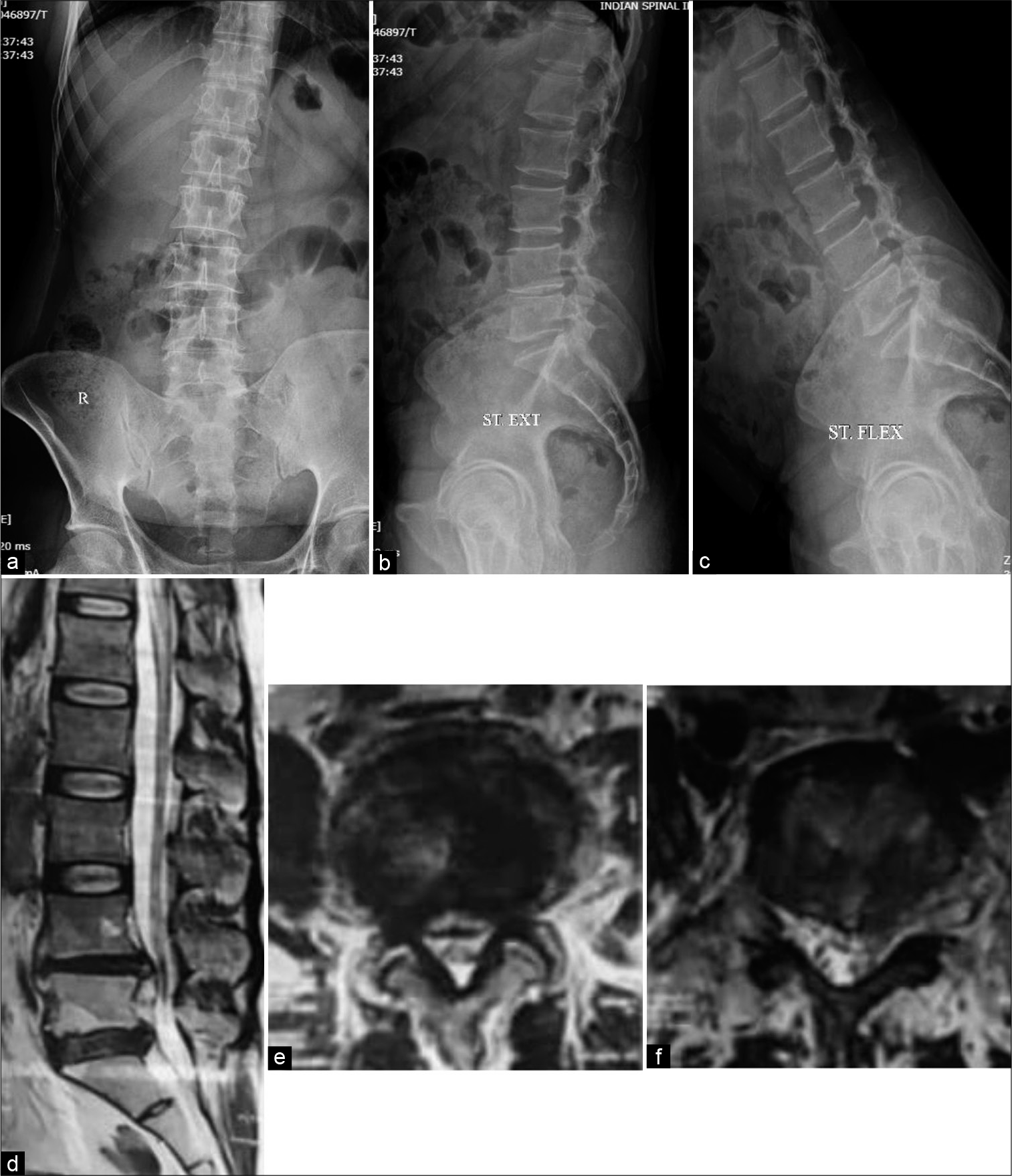
Figure 1:
Plain radiographs anteroposterior (a), lateral in flexion (b) and extension (c) showing decreased joint space and focal kyphosis at L4-L5 segment. Sagittal (d) and axial T2-weighted MR images showing L4-5 segmental kyphosis with decreased disk space and lumbar canal stenosis (e) and prolapsed intervertebral disk at L5-S1 (f).
Postoperative course including readmission
The initial postsurgical course was uneventful; the patient was mobilized the next day, and discharged the following day.
However, on the 10th postoperative day, he developed recurrent back pain (VAS 8), a high grade fever, difficulty in walking, and was readmitted. The following laboratory studies were abnormally elevated; CRP-115.18 mg/dl, ESR-99/1 h, and TLC-10200 with Absolute Neutrophil Count-8410. The fever profile, including tests for dengue fever, and malaria were negative. He was started on broad-spectrum antibiotics and his symptoms were partially relieved with Meropenem 1 g thrice a day. Blood cultures showed B. cepacia sensitive to meropenem, and clindamycin. The diagnosis of B. cepacia from blood culture specimens was established utilizing Bactec Model 9240 (Becton-Dickinson) or BacT/ALERT 3D. Vitek II automated antibiotic susceptibility system was utilized to detect the relevant sensitive antibiotics. VAS score for back pain reduced to two and fever subsided. Patient was discharged and advised 6 weeks of IV antibiotics.
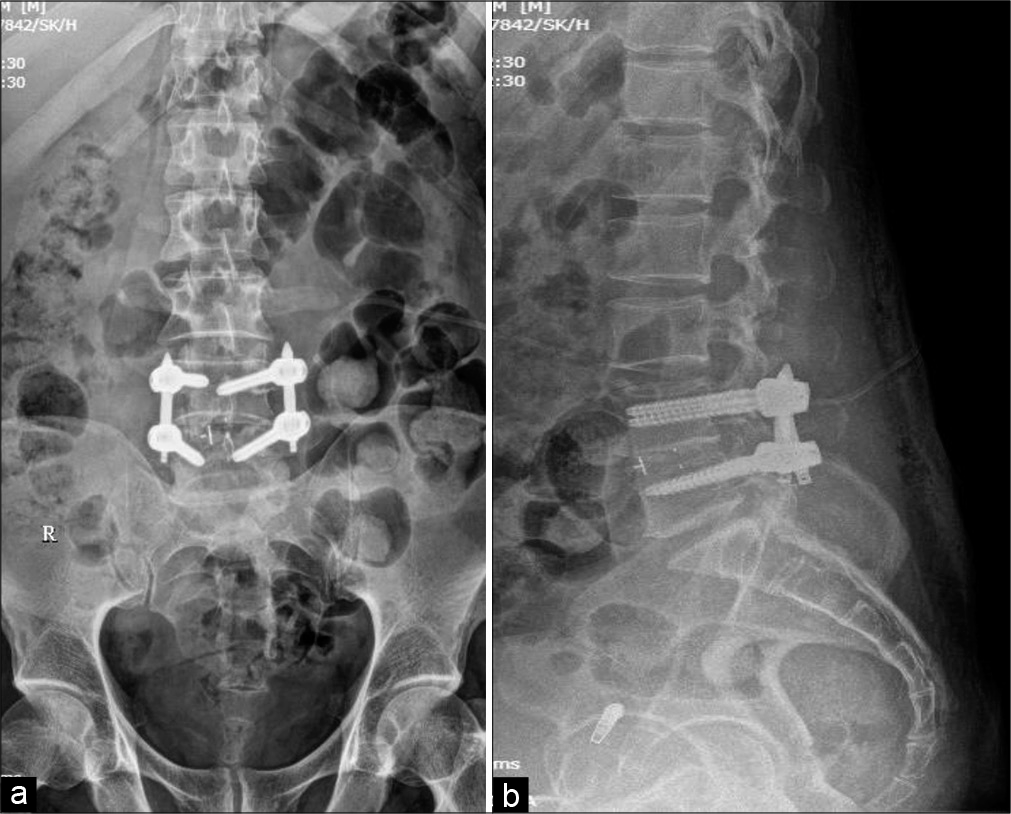
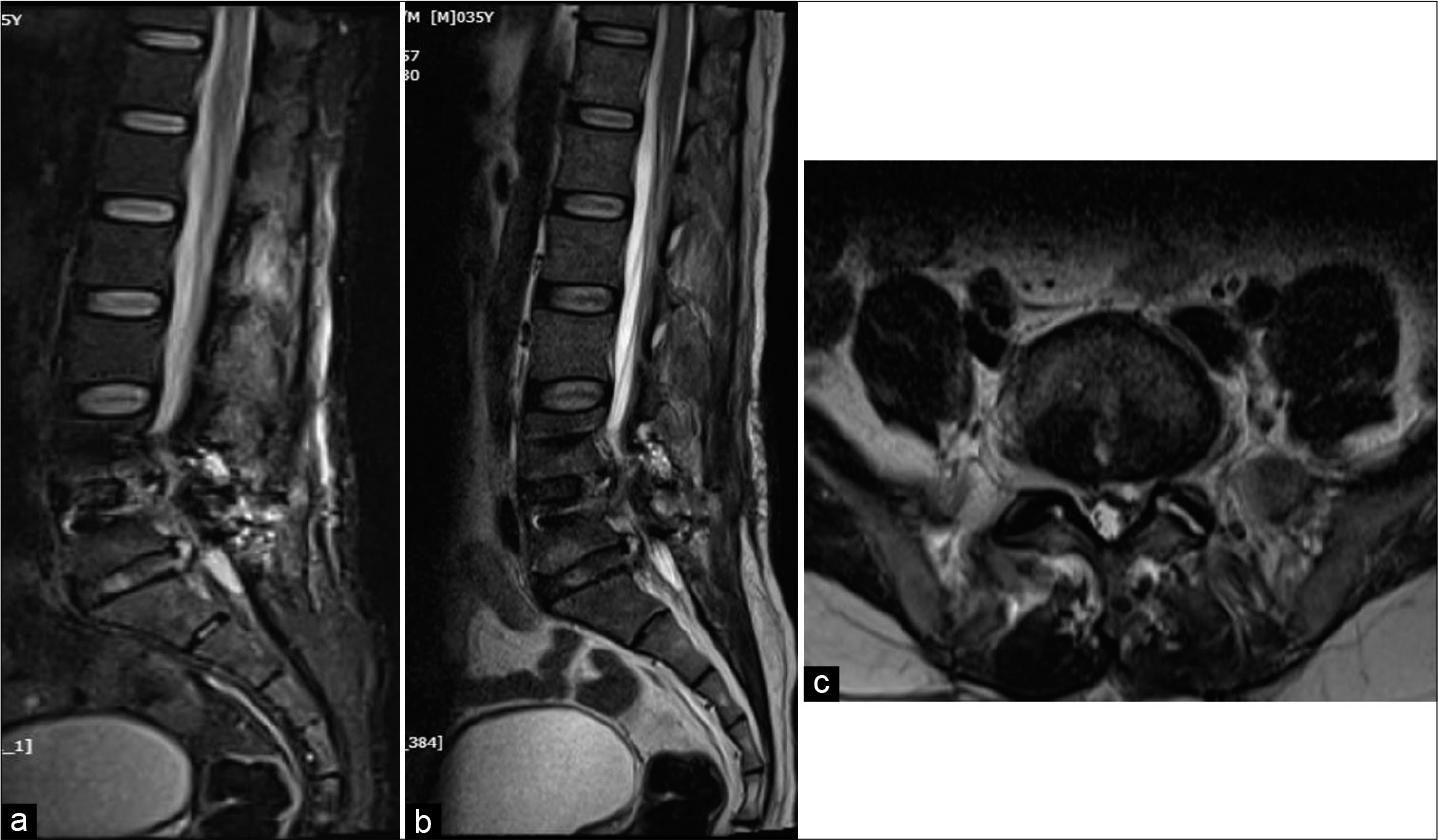
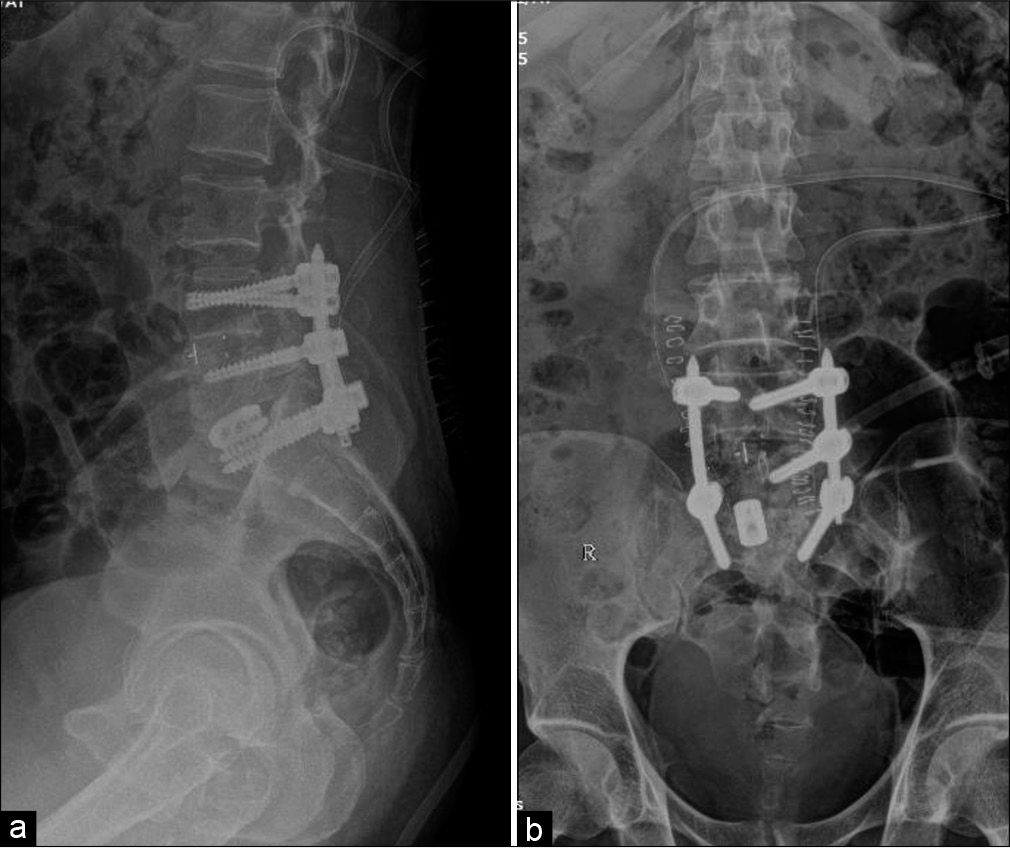
Subsequently, 3 weeks’ post surgery, patient represented with back pain (VAS 8) and left lower limb radiculopathy. He had remained on appropriate antibiotic therapy and laboratory investigations showed improvement in the ESR (99 down to 64), and CRP (115.18 mg/dl down to 34) levels. The MR was suggestive of spondylodiscitis at the L5-S1, and he underwent a TLIF L5-S1 with insertion of Titanium Cage [
DISCUSSION
Postoperative spinal infections are largely attributed to four surgery related risk factors: (1) longer duration; (2) intra-operative blood transfusions; (3) incidental durotomies; and (4) more than 10 people in the operation theater. PS is predominantly seen in the elderly immunocompromised individuals or those with significant co-morbidities (e.g., age over 50 with a male preponderance).
B. cepacia is a motile, aerobic Gram-negative bacillus that is ubiquitous in the environment. It can be isolated from soil, water, and plants.[
Establishing the rare diagnosis of B. cepacia
B. cepacia is an uncommon cause of spondylodiscitis with just seven cases described in previously published literature. Blood cultures best correctly identify the pathogen and appropriate antibiotic treatment can be initiated. B. cepacia has the ability to induce chromosomal beta-lactamase and alter penicillin-binding protein which often makes it resistant to beta-lactate antibiotics. It is, however, sensitive to meropenem, minocycline, fluoroquinolones, and third-generation cephalosporins.[
Management must be considered inadequate/unsuccessful if patients’ exhibit new or recurrent symptoms (e.g., increased pain/radiculopathy) while on antibiotics with persistently elevated or increasing ESR and/or CRP lab values and new pathological MRI findings consistent with progression of discitis.[
Management of PS
Management of B. cepacia as the cause of PS requires a combination of conservative treatment with appropriate antibiotics, with or without additional operative intervention (debridement, decompression with/without fusion if significantly unstable).[
CONCLUSION
B. cepacia is an uncommon hospital-acquired infection, but must be considered as a potential postoperative nosocomial infection in an immunosuppressed patient and/or in those with significant comorbidities. When it is the underlying cause of spondylodiscitis accompanied by bony destruction, it must be appropriately treated with antibiotic therapy and surgical debridement with/without fusion as indicated.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Acknowledgments
Nil.
References
1. Gouliouris T, Aliyu SH, Brown NM. Spondylodiscitis: Update on diagnosis and management. J Antimicrob Chemother. 2010. 65: iii11-24
2. Hsieh CT, Hsu SK, Chang CJ. Thoracic vertebral osteomyelitis caused by Burkholderia cepacia in an immunocompetent adult. Surg Infect. 2013. 14: 476-9
3. Kang BU, Lee SH, Ahn Y, Choi WC, Choi YG. Surgical site infection in spinal surgery: Detection and management based on serial C-reactive protein measurements. J Neurosurg. 2010. 13: 158-64
4. LiPuma JJ. Update on the Burkholderia cepacia complex. Curr Opin Pulmon Med. 2005. 11: 528-33
5. Przybylski GJ, Sharan AD. Single-stage autogenous bone grafting and internal fixation in the surgical management of pyogenic discitis and vertebral osteomyelitis. J Neurosurg. 2001. 94: 1-7
6. Weinstein L, Knowlton CA, Smith MA. Cervical osteomyelitis caused by Burkholderia cepacia after rhinoplasty. J Infect Dev Ctries. 2008. 2: 76-7
7. Yang BH, Lee MS, Lee JH, Lee HJ. Pyogenic spondylitis in a healthy adult caused by Burkholderia cepacia. Infect Chemother. 2008. 40: 233-6