- Departments of Neurosurgery, Centro Médico ABC, Mexico City, México.
- Department of Neurosurgery, High Specialty Medical Unit, La Raza Hospital, Mexican Social Security Institute, Mexico City, México.
- Departments of Anesthesiology, Centro Médico ABC, Mexico City, México.
Correspondence Address:
Enrique de Font-Réaulx
Departments of Anesthesiology, Centro Médico ABC, Mexico City, México.
DOI:10.25259/SNI_435_2019
Copyright: © 2020 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Enrique de Font-Réaulx, Ramón López López, Luis Guillermo Díaz López. Infrared thermography mapping plus neuronavigation target location in an eloquent area cavernoma resection. 13-Mar-2020;11:44
How to cite this URL: Enrique de Font-Réaulx, Ramón López López, Luis Guillermo Díaz López. Infrared thermography mapping plus neuronavigation target location in an eloquent area cavernoma resection. 13-Mar-2020;11:44. Available from: https://surgicalneurologyint.com/surgicalint-articles/9899/
Abstract
Background: Safety and efficacy are irrebuttable goals in neurosurgery.
Methods: We performed a subcortical cavernoma resection in an eloquent area, where we recorded and compared the maximal and minimal brain temperature measured by an infrared thermographic camera and thermometer with the neuronavigation (NN) target location and real anatomical lesion location.
Results: The hottest cortical point correlated to the subcortical cavernoma location. The NN located the target at 10 mm away from the hottest point.
Conclusion: More studies are needed to better understand the thermic radiation of the brain in health and in disease, but we believe that evaluating brain temperature, it could be possible to improve accuracy in neurosurgery and generate more knowledge about brain metabolism in vivo.
Keywords: Brain shift, Brain temperature, Cavernoma, Eloquent area, Infrared thermography mapping, Neuronavigation
INTRODUCTION
Accuracy in neurosurgery is mandatory. Neuronavigation (NN) systems are useful surgical guiding systems, but they have well known limitations. In many surgical scenarios, NN can misguide to false locations mainly due to the brain shift phenomenon and deformation.[
Cavernomas make up approximately 8–15% of all intracranial vascular malformations, and the most common presenting symptom is seizures.[
Since the first documented inflammation description by Aulus Cornelius Celsus (25 AC–50 AD) in De Medicina, it is known that inflammation causes hyperthermia. The thermic radiation evaluation of the brain could be useful trans-operatory information, as thermographic cameras, thermometers, and qualitative thermosensitive materials can measure brain metabolism by its caloric radiation. They have been used for studying the brain temperature in different pathophysiological processes[
MATERIALS AND METHODS
Clinical Presentation
A right-handed 22-year-old male described progressive anomia, aphasia, and agraphia for the past 2 years, with 2–5 min episodes of acute worsening of symptoms presented in average every 2 weeks. He was treated with a short course of valproate acid without improvement under the supervision of another physician. He is the product of a twin normoevolutive pregnancy. During the physical examination, a non-fluent agrammatism, anomia, and a subtle right hemiparesis were documented. A TC showed a subcortical hyperdense lesion of 2 cm × 1.6 cm × 1.5 cm in the left supramarginal gyrus region. A recent electroencephalogram was normal.
RESULTS
Description of the procedure and results
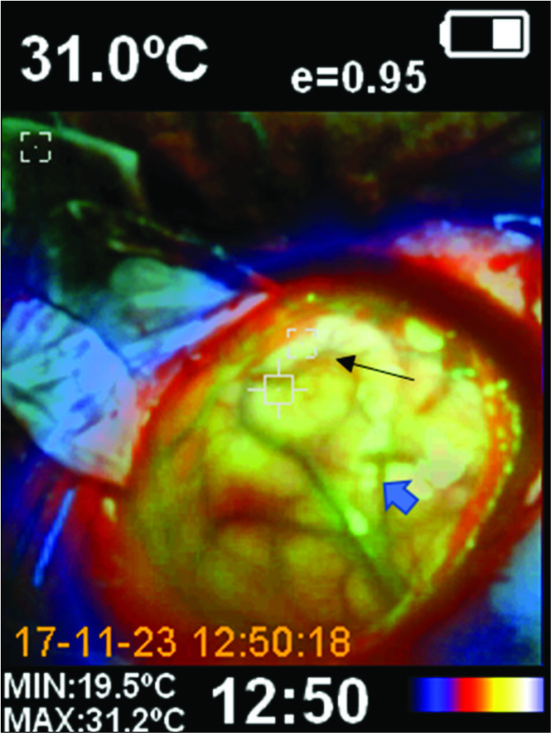
Under entropy-monitored general intravenous anesthesia, we performed a conventional frontoparietotemporal left craniotomy guided by NN (Medtronic® optical neuronavigation system). Anesthetic profundity measured by entropy was 46, inspired fraction of oxygen was 60%, oxygen saturation was 100%, hemoglobin (14.3 g), hematocrit (42%), and carbon dioxide (32.3) by arterial blood gas. Corporal temperature was 35.2°C and the operating room temperature was 20°C, with a relative humidity of 40% during the ITM. Immediately after the dura incision, we performed a panoramic ITM of the exposed brain surface using a professional handheld thermal imaging camera that combines the functions of surface temperature and real-time thermal imaging that can turn a thermal image into a visible image [
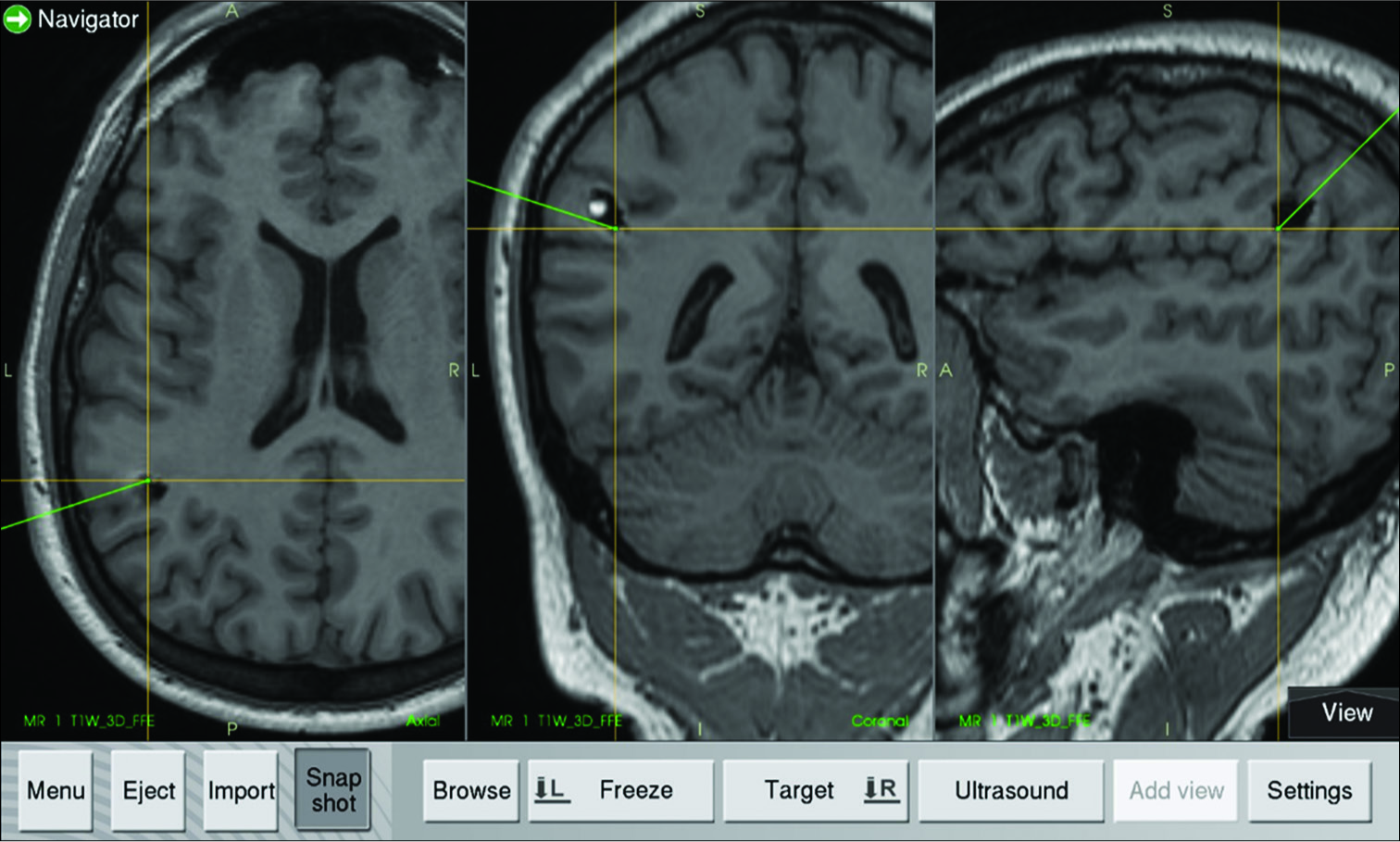
Using the NN, we located the shortest cortex entry zone to the lesion [
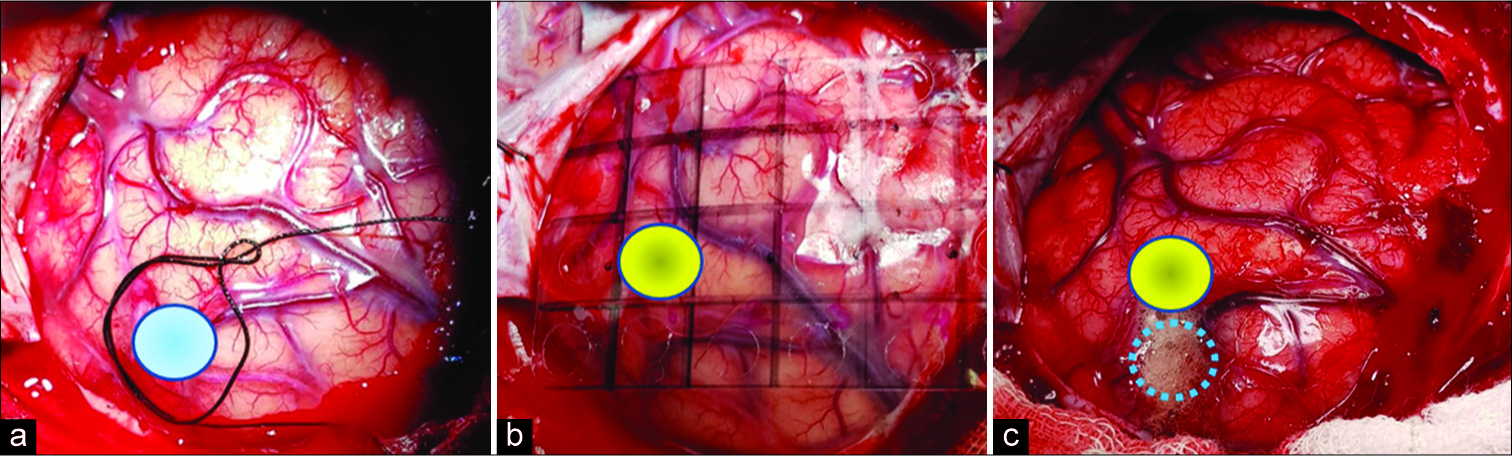
Figure 3:
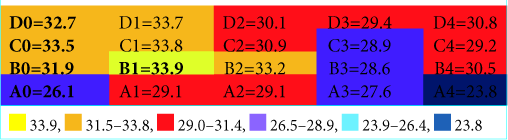
(a) Neuronavigation system localization of the target (blue circle); (b) Grid placement for the pointer thermographic recording where the yellow circle represents the hottest thermographic recording [coordinate B1 of
DISCUSSION
Watson et al. observed that changes in blood temperature are related to changes in brain blood flow, whereas cortical brain temperature is determined by cerebral blood flow-metabolism coupling and the infrared image could be sufficiently sensitive to detect ischemia.[
Based on the previously described evidence that establishes the efficacy of ITM for detecting even subtle blood flow changes, we describe a case where a subcortical cavernoma below the supramarginal gyrus was the surgical target. The NN system located an entry point that was approximately 10 mm misplaced due to the brain shift, as was demonstrated later during the surgery. Before the trans-sulcal approach was performed, using a thermographic camera and thermographic thermometer, we detected a hotter cortical point (difference in temperature between the hottest and coldest cortical recording was 10.1°C) in the supramarginal gyrus surface at approximately 4 mm dorsal to the shortest cortical distance to the reallocation of the cavernoma. Two different technologies acted together to improve the target location accuracy in this case. The patient stayed within stable physiological hemodynamic parameters during the surgical procedure to maintain cerebral self-regulation.
CONCLUSION
More studies are needed to understand and predict the thermic behavior of the brain in physiological and in different pathological conditions, to incorporate temperature measurement devices, such as thermographic thermometers, cameras, or qualitative thermographic devices in daily neurosurgical procedures, but we believe that this is a promising avenue to explore. It could be useful to incorporate more than one device to improve target location accuracy in neurosurgery.
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. de Font-Réaulx Rojas E, Martínez Ochoa EE, López López R, López Díaz LG. Infrared thermography brain mapping surveillance in vascular neurosurgery for anterior communicating artery aneurysm clipping. Surg Neurol Int. 2018. 9: 188-
2. Fan X, Roberts DW, Schaewe TJ, Ji S, Holton LH, Simon DA. Intraoperative image updating for brain shift following dural opening. J Neurosurg. 2017. 126: 1924-33
3. Gorbach AM, Heiss J, Kufta C, Sato S, Fedio P, Kammerer WA. Intraoperative infrared functional imaging of human brain. Ann Neurol. 2003. 54: 297-309
4. Hoffmann N, Weidner F, Urban P, Meyer T, Schnabel C, Radev Y. Framework for 2D-3D image fusion of infrared thermography with preoperative MRI. Biomed Tech (Berl). 2017. 62: 599-607
5. Hollmach J, Hoffmann N, Schnabel C, Küchler S, Sobottka S, Kirsch M. Highly sensitive time-resolved thermography and multivariate image analysis of the cerebral cortex for intrasurgical diagnostics. Ther Diagn. 2013. 8565: 1605-7422
6. Keles GE, Lamborn KR, Berger MS. Coregistration accuracy and detection of brain shift using intraoperative sononavigation during resection of hemispheric tumors. Neurosurgery. 2003. 53: 556-62
7. Mery F. Tecnología y neurocirugía. Rev Chil Neuro Psiquiat. 2010. 48: 173-4
8. Nabavi A, Black PM, Gering DT, Westin CF, Mehta V, Pergolizzi RS. Serial intraoperative magnetic resonance imaging of brain shift. Neurosurgery. 2001. 48: 787-97
9. Nakagawa A, Fujimura M, Arafune T, Sakuma I, Tominaga T. Intraoperative infrared brain surface blood flow monitoring during superficial temporal artery-middle cerebral artery anastomosis in patients with childhood moyamoya disease. Childs Nerv Syst. 2008. 24: 1299-305
10. Naydenov E, Minkin K, Penkov M, Nachev S, Stummer W. Infrared thermography in surgery of newly diagnosed glioblastoma multiforme: A technical case report. Case Rep Oncol. 2017. 10: 350-5
11. Neves EB, Vilaça-Alves J, Rosa C, Reis VM. Thermography in neurologic practice. Open Neurol J. 2015. 9: 24-7
12. Nimsky C, Ganslandt O, Cerny S, Hastreiter P, Greiner G, Fahlbusch R. Quantification of, visualization of, and compensation for brain shift using intraoperative magnetic resonance imaging. Neurosurgery. 2000. 47: 1070-9
13. Pamias-Portalatin E, Duran IS, Ebot J, Bojaxhi E, Tatum W, Quiñones-Hinojosa A. Awake-craniotomy for cavernoma resection. Neurosurg Focus. 2018. 45: V3-
14. Raabe A, Nakaji P, Beck J, Kim LJ, Hsu FP, Kamerman JD. Prospective evaluation of surgical microscope-integrated intraoperative near-infrared indocyanine green videoangiography during aneurysm surgery. J Neurosurg. 2005. 103: 982-9
15. Roberts DW, Hartov A, Kennedy FE, Miga MI, Paulsen KD. Intraoperative brain shift and deformation: A quantitative analysis of cortical displacement in 28 cases. Neurosurgery. 1998. 43: 749-58
16. Watson JC, Gorbach AM, Pluta RM, Rak R, Heiss JD, Oldfield EH. Real-time detection of vascular occlusion and reperfusion of the brain during surgery by using infrared imaging. J Neurosurg. 2002. 96: 918-23