- Department of Neurosurgery, Hospital de Especialidades, Centro Médico Nacional La Raza, Mexico City, Mexico.
Correspondence Address:
Jorge Arturo Santos-Franco, Department of Neurosurgery, Hospital de Especialidades, Centro Médico Nacional La Raza, Mexico City, Mexico.
DOI:10.25259/SNI_873_2022
Copyright: © 2022 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Jorge Arturo Santos-Franco, Carlos Antonio Cruz-Argüelles, Fernando Agustin-Aguilar, Adrián Alejandro Abrego-Salinas, Martín Roberto Casas-Martínez, Jorge Luis Olivares-Peña. Intracranial aneurysms in pediatric population treated with flow diverters: A single-center experience. 11-Nov-2022;13:522
How to cite this URL: Jorge Arturo Santos-Franco, Carlos Antonio Cruz-Argüelles, Fernando Agustin-Aguilar, Adrián Alejandro Abrego-Salinas, Martín Roberto Casas-Martínez, Jorge Luis Olivares-Peña. Intracranial aneurysms in pediatric population treated with flow diverters: A single-center experience. 11-Nov-2022;13:522. Available from: https://surgicalneurologyint.com/?post_type=surgicalint_articles&p=11994
Abstract
Background: Pediatric intracranial aneurysms (PIAs) are uncommon. Flow diverters (FDs) have shown to be effective on treatment of selected aneurysms.
Methods: We describe 10 cases of PIAs treated with FDs at one medical center in Mexico, from April 2015 to April 2020.
Results: Out of 230 patients treated with FDs, 10 (4.3%) were pediatric. Average age was 9.4 years old (R: 6–15). Two patients (20%) had subarachnoid hemorrhage, 3 had epilepsy (30%), 3 (30%) had clinical signs of cranial nerve compression, and 4 (40%) had only headache. Two patients were in 1a grade of Hunt and Kosnik scale. Out of the nonruptured aneurysms, 7 (70%) were in 15 points of Glasgow Coma Scale and 1 patient (10%) was in 13 points. Treatment was performed without complications; nevertheless, appropriate distal deployment was not achieved in one case. At discharge, nine patients had 5 points of Glasgow Outcome Scale. All patients underwent computed tomography angiography or digital subtraction angiography at 1, 3, 6, and 12 months, 2 patients (20%) had a 2-year follow-up, and 3 patients (30%) had a 3-year follow-up. According to Kamran grading scale, 9 patients (90%) were classified as Grade 4 and 1 patient (10%) as Grade 3.
Conclusion: Even though it is a small series, as this is an uncommon disease, we may suggest that FDs are useful to treat properly selected PIAs. Our study has consecutive imaging assessment at least a year of follow-up in which aneurysm stable occlusion was observed in 90% of patients.
Keywords: Flow diverter, Intracranial aneurysm, Intracranial pediatric aneurysm
INTRODUCTION
Intracranial aneurysms (IAs) in pediatric population (pediatric IAs [PIAs]) are rare, representing <5% of events (0.17–4.6%).[
MATERIALS AND METHODS
Population
We reviewed the clinical and imaging records of all patients with IAs which were treated by endovascular means at La Raza National Medical Center between April 2015 and April 2020.
It should be noticed that our department of neurosurgery has both vascular microsurgery and endovascular intervention, and all cases are analyzed in an expert session and with Institutional Review Board approval. We registered all IAs cases which were treated with FDs and then we specifically selected patients under 18 years of age. After analyzing each case, the following characteristics were important indications for the use of the FD: (1) aneurysm fusiform appearance, (2) very wide neck, and (3) our lack of experience in by-pass in children.
Initial clinical and imaging data and follow-up imaging findings were reviewed. Before treatment, the clinical status was assessed using the Glasgow Coma Scale (GCS)[
Incidents and complications of the procedures were registered. Patients were assessed at hospital discharge and during follow-up using the Glasgow Outcome Scale (GOS).[
All patients were followed up with digital subtraction angiography (DSA) or computed tomography (CT) angiography at 3, 6, 9, and 12 months after treatment. Aneurysm thrombosis degree was assessed according to a scale proposed by Kamran et al.[
Treatment description
Patients were submitted to double antiplatelet regime daily during 5 days before procedure, with clopidogrel 37.5 mg and aspirin 100 mg for children weighing <45 kg, and clopidogrel 75 mg and acetylsalicylic acid 100 mg for children weighing >45 kg. All patients were treated under general anesthesia at angiography suite. Femoral approach was performed using the Seldinger technique to place a 6F femoral sheath introducer. A 100 UI/Kg heparin bolus was administered to maintain a coagulation time activity approximately twice the basal value. In all cases, a 6F Chaperon guiding catheter (MicroVention, Aliso Viejo, California) was used to cannulate the cervical internal carotid artery (ICA) or the vertebral artery (VA) V2 segment, as required. In almost all cases, a 5F distal Sophia catheter (MicroVention, Tustin, California, USA) was used to reach the ICA at cavernous or clinoid segment or the VA at V3 segment. The FD selection was performed according to parent artery diameter. The deployment of the FDs was carried out using a well-known international technique, as previously described.[
After the procedures, the patients were kept on a double antiplatelet therapy for 6 months, at the doses shown above. Subsequently, treatment with clopidogrel was continued permanently [
Figure 1:
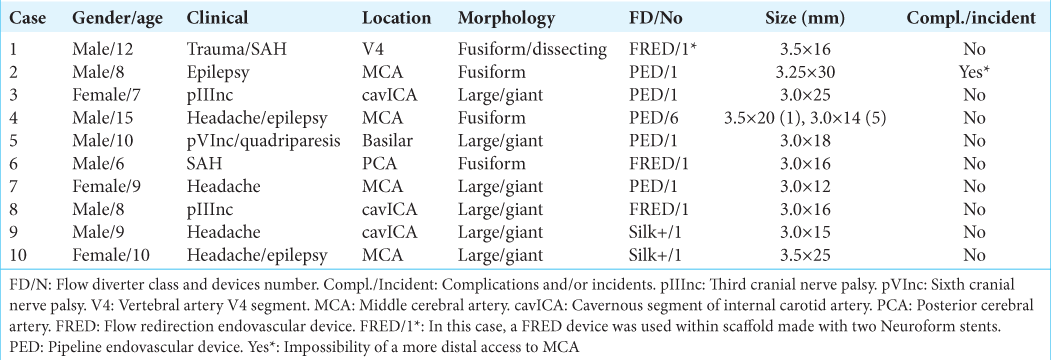
(Case 2) (a) Fusiform aneurysm affecting the entire left M1 segment and part of the M2 segment, together with parts of a saccular appearance, the M1 segment being the largest, with blebs (thin arrow). An stenosis at the origin of the middle cerebral artery (thick arrow) is evident. (b and c) Flow diverters (FD) deployed (dotted arrows) along the stenotic segment over the middle cerebral artery (arrow) to the M1 segment. The stenosis caused great difficulty and inability to navigate and deploy it more distally. Some calcified segments of the aneurysm (star) can be seen. In c, the arrow shows the stenotic site that only slightly widened despite being balloon treated twice. (d and e) Contrast injection revealed stent patency, which was initially slow with only flow toward the FD center (dotted arrows on d). At the end of the procedure and through a digital subtraction angiography (DSA) on AP projection, appropriate flow is observed through the FD and left middle cerebral artery, regardless of the stenotic segment (thick arrow). (f) One month follow-up transvenous contrast injection CTA with axial section where a stenotic segment is observed (thick arrow) and even residual flow on aneurysm saccular part (dotted arrow). Some calcified segments of the aneurysm can be seen (arrows). g: One-year DSA control on AP projection where an excellent flow is observed through FD toward MCA distal segments, regardless to stenotic part (thick arrow). A very reduced residual filling is seen at old saccular part at M1 segment (dotted arrow). h: A 24-month follow-up CTA on axial section where persistence of the stenotic part is seen (thick arrow) but with a filling of less that 1% regarding old saccular part at M1 segment (dotted line). Distal flow toward FD can be seen (arrow).
Figure 2:
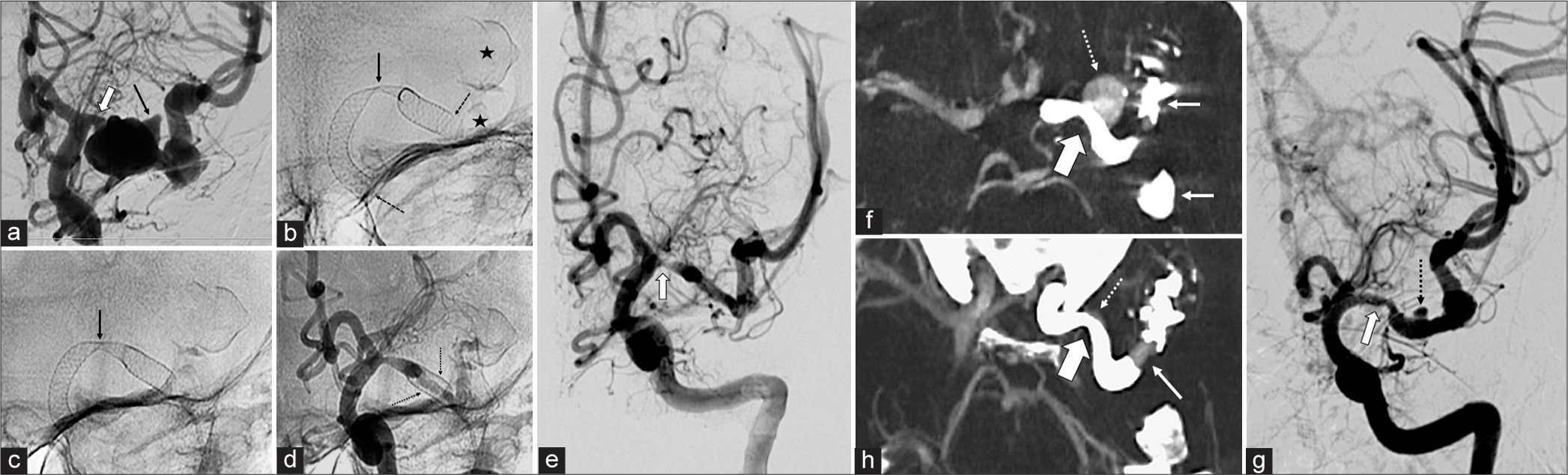
(Case 3) (a) Digital subtraction angiography (DSA) on lateral projection showing a large aneurysm on cavernous segment with a bleb that seems to be under an intradural location (thick arrow). (b) Lateral X-ray taken immediately after placing flow diverter, highlighted with dotted arrows. (c and d) Immediate follow-up DSA on lateral projections showing reduction of aneurysm flow velocity. (e and f) Follow-up DSA in lateral and AP projections at 6 months showing the absence of intra-aneurysmal flow. Intrastent stenosis is observed (arrow), presumably due to endothelium hyperplasia. (g and h) Lateral and AP projections of follow-up at 24 months with persistent absence of aneurysmal flow. The stenosis described in E is no longer observed.
Figure 3:
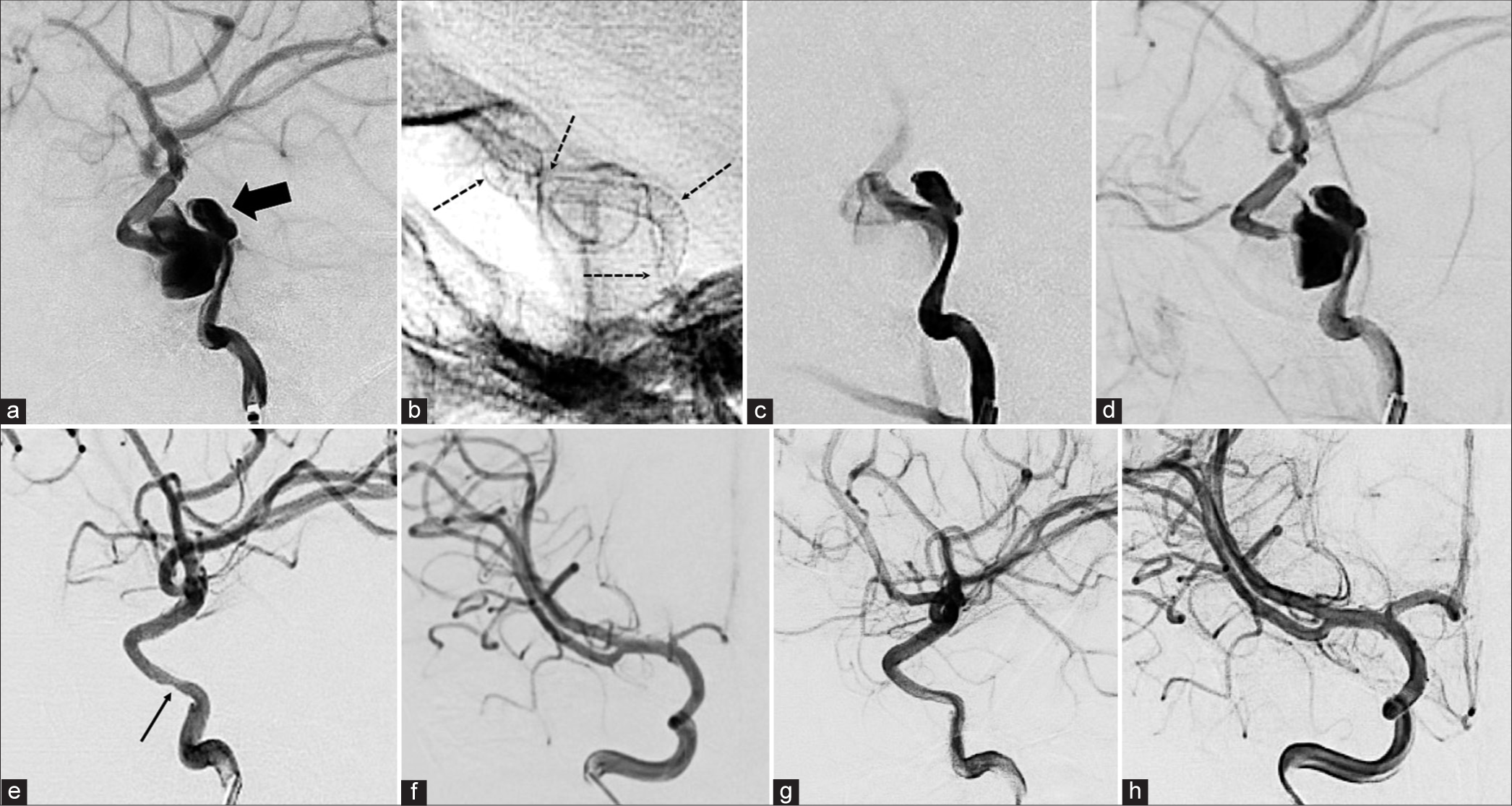
(Case 4) (a and b) T2-weighed MRI showing a partially thrombosed fusiform aneurysm on the left middle cerebral artery at sphenoidal segment. The lesion mass effect is very evident over cerebral peduncle as well as ipsilateral central core structures (arrows). (c) Digital subtraction angiography (DSA) in AP projection showing a fusiform aneurysm originating from the bifurcation of the internal carotid artery, in the entire M1 segment of the left MCA and beyond the Sylvian point. Inadequate iodinated contrast filling is evident in the distal branches. (d) The artery was reconstructed with six tandem flow diverters (arrows show start and end of devices). (e and f) AP and lateral projections DSA showing appropriate blood flow through flow diverters (FDs) and MCA distal branches. There are no obvious changes in the fusiform aneurysm. (g and h) One-month follow-up CTA shows no evidence of aneurysmal lesion around flow diverters as well as excellent distal flow (thick arrows). Dotted arrows show aneurysm calcifications. (i and j) Follow-up DSA with AP and lateral projections after 3 months of treatment showing appropriate reconstruction of middle cerebral artery and excellent distal flow, which, in turn, is better than the flow observed before the treatment (refer to c).
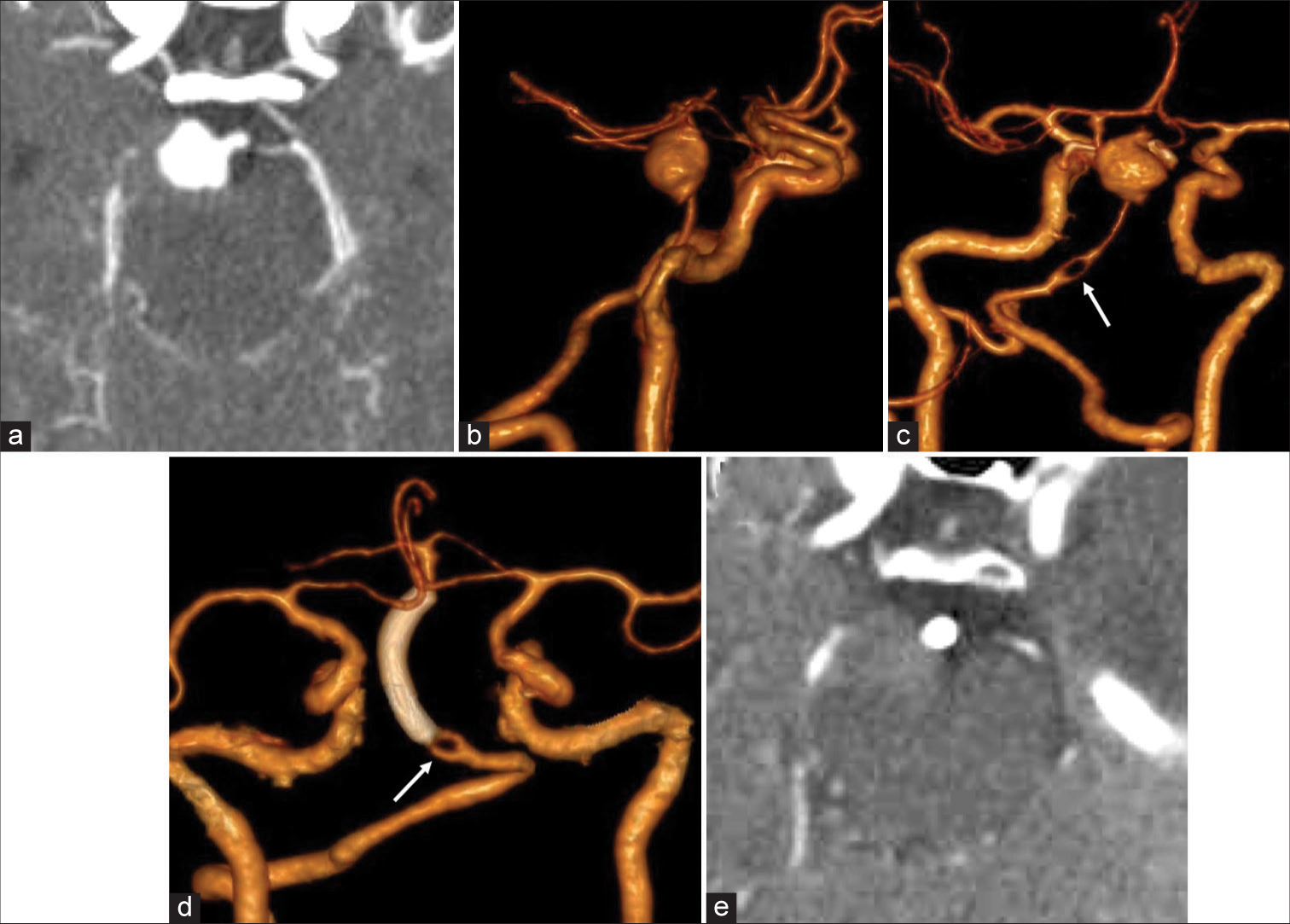
Figure 4:
(Case 5) (a-c) A saccular aneurysm is seen occupying on basilar artery mid and distal portions. The MPR image clearly shows the aneurysm compressing the right midbrain peduncle (a). An fenestration related to basilar artery proximal third portion can be noticed (arrow on c). (d and e) CTA performed 2 months after the treatment showing FD with distal patency in addition to absence of aneurysm. The MPR image already shows the absence of filling of the aneurysmal sac. FD position is evident just distal to aforementioned fenestration (arrows on d).
Figure 5:
(Case 6) (a and b) A large fusiform aneurysm located on the right posterior cerebral artery (PCA) P2 segment is shown. (c and d) Superselective microcatheterization, initially of aneurysm with contrast injection and subsequent microcatheterization on PCA more distal segments (microcatheter is indicated by the arrows). (e) Digital subtraction angiography (DSA) in lateral projection performed during flow diverters (FDs) deployment where the distal end can be observed (thick arrow) in addition to a part of microcatheter where the device is attached (arrow) before a complete detachment. (f) One-month DSA showing absence of aneurysm with adequate artery reconstruction along and excellent distal filling. It is possible to see the proximal end of the FD (thin arrow).
RESULTS
From April 2015 to April 2020, at La Raza National Medical Center, 505 endovascular therapeutic procedures were performed in 463 patients; 400 were IAs in 383 patients, from which 230 patients with 247 aneurysms were treated with FDs, and out of these, 10 patients (4.3%) were under 18 years old. It is worth noting that four other children with ruptured aneurysms were excluded from this study because they were treated by clipping.
Seven patients (70%) were male. The average age was 9.5 years (R: 7–15). No child had a remarkable medical history during gestation and they had no congenital disease or any important recent infectious disease. Only a 12-year-old male patient (10%) had a V4 segment dissecting aneurysm, as well as an indirect cervical trauma history due to a car accident.
The initial presentation of 2 patients (20%) was a subarachnoid hemorrhage (SAH), in 2 (20%), it was epilepsy, 2 patients (20%) had clinical signs of cranial nerve compression, and 4 patients (4%) had a history of headache.
Patients whose initial presentation was SAH were admitted with Hunt and Kosnik scale Grade 1a and were treated 20 days after the bleeding event. Of eight patients with an initial presentation different than SAH, 7 (70%) had a GCS score of 15, and 1 (10%), who had a large basilar aneurysm, had a GCS of 13 in addition to quadriparesis and bilateral paresis of the sixth cranial nerve [
Procedures were performed without complications in all patients. For Patient 2 [
Patients recovered from general anesthesia without any additional deficit and their progress was unremarkable, with the exception of slight headache in Patients 3, 5, and 7. Based on GOS, patients were rated as follows: 9 (90%) in Grade 5 and 1 (10%) in Grade 4. The latter was the case for a large aneurysm on the basilar artery that was admitted with a GCS of 13.
In our institution, as a standard practice, we follow-up with a DSA and/or CT angiography (CTA) at 1, 3, 6, and 12 months after the procedure and subsequently every year. Ionizing radiation in children has been shown to affect the growth plates, gonads, and thyroid gland. For that reason, lead shields were used over some parts of the body, including the pelvis and neck. We avoided carrying out the follow-up at 1 month and 3 months in children under 10 years of age, due to the risk of leukemia or malignant brain tumor. Nevertheless; we decided that a follow-up at 6 months and at 1 year was needed, either by CTA or DSA, since the possible benefits obtained with follow-up images outweighed by far the actual and potential risks.
All patients underwent follow-up assessments at 1 year, 4 patients (40%) at 2 years, and 4 patients (40%) at 3 years. Imaging follow-up was excellent, the aneurysms showed intra-aneurysmal thrombosis progression with parent vessel blood flow patency. The final imaging studies showed nine patients with Kamran’s Grade 4. It is important to mention that in Case number 2 in which we had a diverter deployment incident, DSA after 1 year showed thrombosis beyond 90%, with a Karman’s Grade 3. The plan was to perform a new DSA follow-up every 2 years. However, the patient’s mother refused, so a 2-year CTA was performed and the aneurysm was found to be already occluded [
DISCUSSION
IAs in pediatric population (PIAs) are rare, representing <5%.[
Dissecting aneurysms are generally secondary to trauma, infection, or congenital diseases.[
Hetts et al.[
Then, in the anterior circulation, 27% are located on ICA and 26% on MCA, while basilar artery was the most common location on posterior circulation. It has been reported that approximately 22% of cases involved multiple aneurysms.[
In most cases, PIAs are symptomatic[
There is no clear consensus regarding treatment of IAs on children. Treatments are based on the application of algorithms and treatment criteria created for the adult population, with specific modifications and “customized” according to the center experience and ultimately in the surgeon’s experience.[
Already used techniques include: (a) simple clipping/coil embolization, which does not apply to dissecting, fusiform and/or giant aneurysms and (b) trapping/sacrifice, which is related to by-pass in case of poor collaterals. Although children with aneurysms usually develop adequate collaterals, it is absolutely necessary to carry out a balloon occlusion test to assess collaterals.
Endovascular management has shown to be safe, effective, and long lasting for this type of aneurysms.[
Even today, there are not many cases of PIAs in the world treated with FDs, nevertheless, the experience continues to grow.[
At early 2017, Barburoglu and Arat reported a successful use of FDs on five PIAs and in their review of reports around the world, they gathered information about 15 successful PIAs treatment.[
The growth of brain arteries has been suggested as a future potential problem, for this reason, it is necessary to consider that approximately at 48 months, a vascular diameter between 81 and 99% is achieved, which, in turn, is similar to those seen in adults.[
We should also mention that ionizing radiation can triple the risk of leukemia or brain tumors, especially in children under 10 years old. However, it has been stated that the risk ratio in children under 10 years old is one new case for every 10,000 CT scans.[
CONCLUSION
IAs in the pediatric population are infrequent; therefore, the cases treated with FDs are few, but enough to demonstrate their efficacy in well-selected cases. It is also important to notice that in our study, all patients had consecutive imaging assessments with 100% compliance at 1 year and 74% at 2 or more years. As more cases emerge globally and patients get older, we will have more data to definitely assess effectiveness and durability of treatment with FDs.
Declaration of patient consent
Institutional Review Board (IRB) permission obtained for the study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
References
1. Agid R, Souza MP, Reintamm G, Armstrong D, Dirks P, TerBrugge KG. The role of endovascular treatment for pediatric aneurysms. Childs Nerv Syst. 2005. 21: 1030-6
2. Aeron G, Abruzzo TA, Jones BV. Clinical and imaging features of intracranial arterial aneurysms in the pediatric population. Radiographics. 2012. 32: 667-81
3. Arat YO, Arat A, Aydin K. Angiographic morphometry of internal carotid artery circulation in Turkish children. Turk Neurosurg. 2015. 25: 608-16
4. Barburoglu M, Arat A. Flow diverters in the treatment of pediatric cerebrovascular diseases. AJNR Am J Neuroradiol. 2017. 38: 113-8
5. Beez T, Steiger HJ, Hänggi D. Evolution of management of intracranial aneurysms in children: A systematic review of the modern literature. J Child Neurol. 2016. 31: 773-83
6. Binh NT, Luu VD, Thong PM, Cuong NN, Anh NQ, Tuan TA. Flow diverter stent for treatment of cerebral aneurysms: A report of 130 patients with 134 aneurysms. Heliyon. 2020. 6: e03356
7. Brinjikji W, Murad MH, Lanzino G, Cloft HJ, Kallmes DF. Endovascular treatment of intracranial aneurysms with flow diverters: A meta-analysis. Stroke. 2013. 44: 442-7
8. Buis DR, van Ouwerkerk WJ, Takahata H, Vandertop WP. Intracranial aneurysms in children under 1 year of age: A systematic review of the literature. Childs Nerv Syst. 2006. 22: 1395-409
9. Burrows AM, Zipfel G, Lanzino G. Treatment of a pediatric recurrent fusiform middle cerebral artery (MCA) aneurysm with a flow diverter. J Neurointerv Surg. 2013. 5: e47
10. Consoli A, Vignoli C, Renieri L, Rosi A, Chiarotti I, Nappini S. Assisted coiling of saccular wide-necked unruptured intracranial aneurysms: Stent versus balloon. J Neurointerv Surg. 2016. 8: 52-7
11. D’Urso PI, Lanzino G, Cloft HJ, Kallmes DF. Flow diversion for intracranial aneurysms: A review. Stroke. 2011. 42: 2363-8
12. Ferrante L, Fortuna A, Celli P, Santoro A, Fraioli B. Intracranial arterial aneurysms in early childhood. Surg Neurol. 1988. 29: 39-56
13. Ghali MG, Srinivasan VM, Cherian J, Wagner KM, Chen SR, Johnson J. Multimodal treatment of intracranial aneurysms in children: Clinical case series and review of the literature. World Neurosurg. 2018. 111: e294-307
14. Herman JM, Rekate HL, Spetzler RF. Pediatric intracranial aneurysms: Simple and complex cases. Pediatr Neurosurg. 1991. 17: 66-72
15. Hetts SW, Narvid J, Sanai N, Lawton MT, Gupta N, Fullerton HJ. Intracranial aneurysms in childhood: 27-year single-institution experience. AJNR Am J Neuroradiol. 2009. 30: 1315-24
16. Hetts SW, English JD, Dowd CF, Higashida RT, Scanlon JT, Halbach VV. Pediatric intracranial aneurysms: New and enlarging aneurysms after index aneurysm treatment or observation. AJNR Am J Neuroradiol. 2011. 32: 2017-22
17. Huang J, McGirt MJ, Gailloud P, Tamargo RJ. Intracranial aneurysms in the pediatric population: Case series and literature review. Surg Neurol. 2005. 63: 424-32
18. Hunt WE, Kosnik EJ. Timing and perioperative care in intracranial aneurysm surgery. Clin Neurosurg. 1974. 21: 79-89
19. Ikeda DS, Marlin ES, Shaw A, Powers CJ. Successful endovascular reconstruction of a recurrent giant middle cerebral artery aneurysm with multiple telescoping flow diverters in a pediatric patient. Pediatr Neurosurg. 2015. 50: 88-93
20. Kamran M, Yarnold J, Grunwald IQ, Byrne JV. Assessment of angiographic outcomes after flow diversion treatment of intracranial aneurysms: A new grading schema. Neuroradiology. 2011. 53: 501-8
21. Kan P, Mokin M, Puri AS, Wakhloo AK. Successful treatment of a giant pediatric fusiform basilar trunk aneurysm with surpass flow diverter. J Neurointerv Surg. 2016. 8: e23
22. Kaschner MG, Kraus B, Petridis A, Turowski B. Endovascular treatment of intracranial “blister” and dissecting aneurysms. Neuroradiol J. 2019. 32: 353-65
23. Lasjaunias P, Wuppalapati S, Alvarez H, Rodesch G, Ozanne A. Intracranial aneurysms in children aged under 15 years: Review of 59 consecutive children with 75 aneurysms. Childs Nerv Syst. 2005. 21: 437-50
24. Lena G, Choux M. Giant intracranial aneurysms in children 15 years old or under. Case reports and literature review. J Pediatr Neurosci. 1985. 1: 84-93
25. Li JS, Yow E, Berezny KY, Bokesch PM, Takahashi M, Graham TP. Dosing of clopidogrel for platelet inhibition in infants and young children: Primary results of the platelet inhibition in children On cLOpidogrel (PICOLO) trial. Circulation. 2008. 117: 553-9
26. Locksley HB. Natural history of subarachnoid hemorrhage, intracranial aneurysms and arteriovenous malformations .Based on 6368 cases in the cooperative study. J Neurosurg. 1966. 25: 219-39
27. Lylyk P, Miranda C, Ceratto R, Ferrario A, Scrivano E, Luna HR. Curative endovascular reconstruction of cerebral aneurysms with the pipeline embolization device: The Buenos Aires experience. Neurosurgery. 2009. 64: 632-42
28. McMillan T, Wilson L, Ponsford J, Levin H, Teasdale G, Bond M. The glasgow outcome scale-40 years of application and refinement. Nat Rev Neurol. 2016. 12: 477-85
29. Navarro R, Brown BL, Beier A, Ranalli N, Aldana P, Hanel RA. Flow diversion for complex intracranial aneurysms in young children. J Neurosurg Pediatr. 2015. 15: 276-81
30. Nishioka H. Report on the cooperative study of intracranial aneurysms and subarachnoid hemorrhage. Section VII. I. Evaluation of the conservative management of ruptured intracranial aneurysms. J Neurosurg. 1966. 25: 574-92
31. Orozco M, Trigueros F, Quintana F, Dierssen G. Intracranial aneurysms in early childhood. Surg Neurol. 1978. 9: 247-52
32. Ostergaard JR, Voldby B. Intracranial arterial aneurysms in children and adolescents. J Neurosurg. 1983. 58: 832-7
33. Patel AN, Richardson AE. Ruptured intracranial aneurysms in the first two decades of life. A study of 58 patients. J Neurosurg. 1971. 35: 571-6
34. Pearce MS, Salotti JA, Little MP, McHugh K, Lee C, Kim KP. Radiation exposure from CT scans in childhood and subsequent risk of leukaemia and brain tumours: A retrospective cohort study. Lancet. 2012. 380: 499-505
35. Pollo C, Meagher-Villmure K, Bernath MA, Vernet O, Regli L. Ruptured cerebral aneurysm in the early stage of life-a congenital origin?. Neuropediatrics. 2004. 35: 230-3
36. Proust F, Toussaint P, Garniéri J, Hannequin D, Legars D, Houtteville JP. Pediatric cerebral aneurysms. J Neurosurg. 2001. 94: 733-9
37. Regelsberger J, Heese O, Martens T, Ries T, Kunkel P, Westphal M. Intracranial aneurysms in childhood: Report of 8 cases and review of the literature. Cent Eur Neurosurg. 2009. 70: 79-85
38. Saini S, Speller-Brown B, Wyse E, Meier ER, Carpenter J, Fasano RM. Unruptured intracranial aneurysms in children with sickle cell disease: Analysis of 18 aneurysms in 5 patients. Neurosurgery. 2015. 76: 531-8
39. Saraf R, Shrivastava M, Siddhartha W, Limaye U. Intracranial pediatric aneurysms: Endovascular treatment and its outcome. J Neurosurg Pediatr. 2012. 10: 230-40
40. Sarica C, Tanrikulu B, Sahin Y, Dağçınar A, Baltacioglu F, Bayri Y. Acute obstructive hydrocephalus due to a giant posterior cerebral artery aneurysm in a pediatric patient. Pediatr Neurosurg. 2018. 53: 247-53
41. Shlobin NA, Raz E, Shapiro M, Moretti L, Cantrell DR, Lam SK. Pipeline embolization of cerebral aneurysms in pediatric patients: Combined systematic review of patient-level data and multicenter retrospective review. J Neurosurg Pediatr. 2021. 27: 668-76
42. Stehbens WE. Intracranial berry aneurysms in infancy. Surg Neurol. 1982. 18: 58-60
43. Teasdale G, Maas A, Lecky F, Manley G, Stocchetti N, Murray G. The Glasgow Coma scale at 40 years: Standing the test of time. Lancet Neurol. 2014. 13: 844-54
44. Trivelato FP, Rezende MT, Fonseca LV, Bonadio LE, Ulhôa AC, Abud DG. Pipeline embolization device for the treatment of a traumatic intracranial aneurysm in a child. Childs Nerv Syst. 2017. 33: 869-72
45. Vachhani JA, Nickele CM, Elijovich L, Klimo P, Arthur AS. Flow diversion for treatment of growing A2 aneurysm in a child: Case report and review of flow diversion for intracranial aneurysms in pediatric patients. World Neurosurg. 2016. 96: 607.e13-7
46. Vargas SA, Diaz C, Herrera DA, Dublin AB. Intracranial aneurysms in children: The role of stenting and flow-diversion. J Neuroimaging. 2016. 26: 41-5
47. Zarzecka A, Gory B, Turjman F. Implantation of two flow diverter devices in a child with a giant, fusiform vertebral artery aneurysm: Case report. Pediatr Neurol. 2014. 50: 185-7