- Infectious Disease Division, Winthrop-University Hospital, Mineola, New York, USA
- State University of New York, School of Medicine, Stony Brook, New York, USA
Correspondence Address:
Burke A. Cunha
Infectious Disease Division, Winthrop-University Hospital, Mineola, New York, USA
State University of New York, School of Medicine, Stony Brook, New York, USA
DOI:10.4103/sni.sni_67_18
Copyright: © 2018 Surgical Neurology International This is an open access journal, and articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as appropriate credit is given and the new creations are licensed under the identical terms.How to cite this article: Burke A. Cunha, Ismail Jimada, Karishma Chawla. Intracranial complications of acute bacterial endocarditis. 25-May-2018;9:107
How to cite this URL: Burke A. Cunha, Ismail Jimada, Karishma Chawla. Intracranial complications of acute bacterial endocarditis. 25-May-2018;9:107. Available from: http://surgicalneurologyint.com/surgicalint-articles/intracranial-complications-of-acute-bacterial-endocarditis/
Abstract
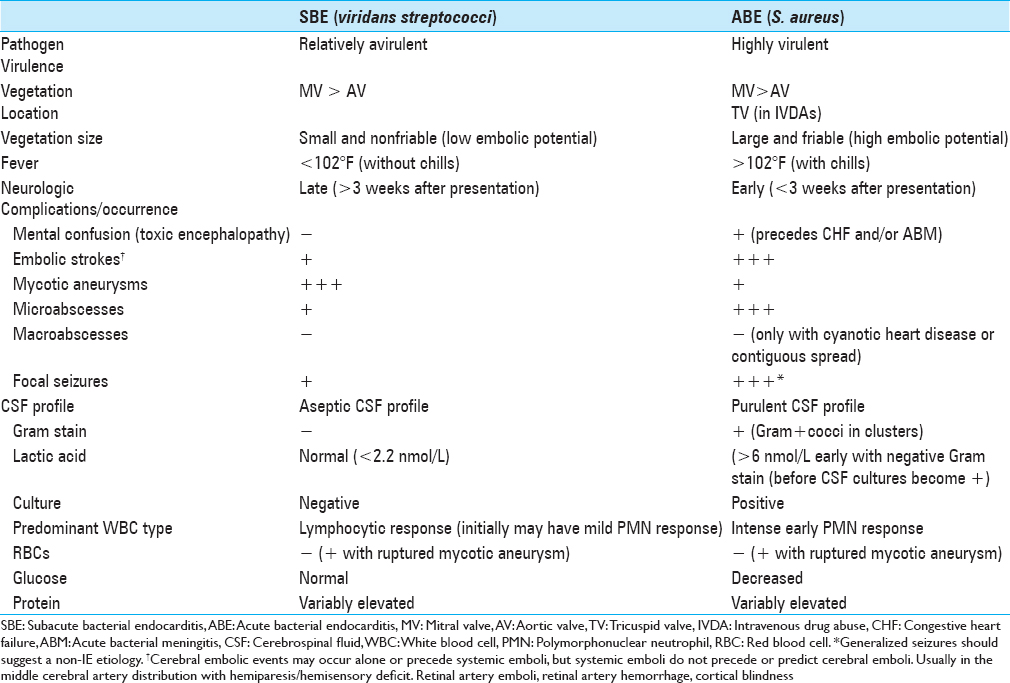
Background:Infectious endocarditis (IE) clinically manifests as either subacute bacterial endocarditis (SBE) or acute bacterial endocarditis (ABE). Neurologic manifestations are markedly different for these two entities. ABE is caused by invasive, highly virulent pathogens (e.g., Staphylococcus aureus), whereas SBE is attributed to relatively avirulent, non-invasive organisms (e.g., viridans streptococci).
Methods:Here, we reviewed the clinical and radiographic presentations of a patient with cranial complications attributed to ABE. Such patients typically develop central nervous system (CNS) septic emboli resulting in stroke (with/without intracranial hemorrhage (ICH)) and/or mycotic aneurysms resulting in ICH bleeds.
Results:With ABE, cerebrospinal fluid (CSF) seeding may result in acute bacterial meningitis (ABM), documented by positive Gram stain and/or culture for S. aureus, decreased glucose, highly elevated lactose acid levels, or ICH. Alternatively, in SBE, the CSF profile reflects an aseptic (viral) meningitis (i.e., Gram stain and culture negative, a normal glucose, and lymphocytic pleocytosis), while septic microemboli to the vasa vasorum contribute to an inflammatory reaction in the adventitia/muscle layer that weakens the vessel wall and results in mycotic aneurysms that may leak but often do not rupture causing ICH.
Conclusion:Here, we reviewed the literature for intracranial pathology accompanying ABE versus SBE. ABE typically results in acute ischemia, septic emboli, stroke/hemorrhagic infarcts, or ICH. SBE more classically produces septic microemboli and mycotic aneurysms that may leak, but rarely producing ICH. We also presented a patient with ABE attributed to S. aureus whose septic emboli/stroke was accompanied by a mycotic aneurysm; the ruptured resulting in a large right occipital ICH.
Keywords: Acute bacterial meningitis (ABM), mycotic aneurysms, SBE, acute bacterial endocarditis (ABE), intracranial hemorrhage (ICH)
INTRODUCTION
This review and case study focus on the presentation, diagnosis, treatment, and outcomes for patients with intracranial acute bacterial endocarditis (ABE) versus SBE. ABE may result in acute intracranial septic emboli and stroke and/or hemorrhagic infarct with/without accompanying mycotic aneurysms that may produce intracranial hemorrhage (ICH). Alternatively, subacute bacterial endocarditis (SBE) is more classically characterized by septic microemboli and mycotic aneurysms that may leak, but rarely rupture (e.g. fewer ICHs). Here, we present a case of ABE where an infected embolus (e.g. Staphylococcus aureus) resulted in a mycotic aneurysm and acute ICH.
CASE REPORT
A 26-year-old male with a history of hypertrophic cardiomyopathy (HOCM) and intravenous drug abuse (IVDA) presented with generalized body ache, fever, and shaking chills. His fever was 103°F, heart rate was 112 beats/minute, he had no cardiac murmur, but he did exhibit tender scattered macular lesions of the palm and soles of the feet (Janeway lesions).
Laboratory studies
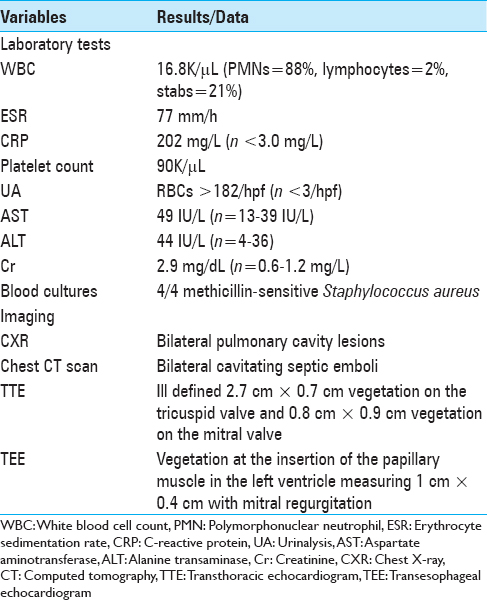
Laboratory studies revealed a white blood cell (WBC) count of 16.8K/μL (n = 3.9-11 K/μL) with 88% neutrophils, lymphocytes of 2% (n = 21%-51%), a platelet count of 90K/μL (n = 160-392K/μL), an erythrocyte sedimentation rate (ESR) of 77 mm/h, an elevated C-reactive protein (CRP) of 202 mg/L (n > 3 mg/L), and blood cultures positive for methicillin-sensitive S. aureus (MSSA; three or four bottles positive persisted for 4 days) [
Diagnostic studies
The Chest X-ray (CXR) showed bilateral pulmonary nodules, while the chest computed tomography (CT) confirmed peripheral bilateral, nodular, cavitating septic emboli [
Hospital course
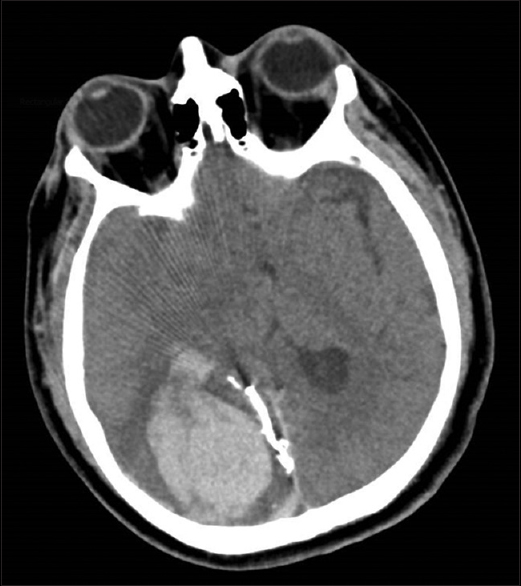
On the sixth hospital day, the patient became lethargic and developed blurred vision. The brain CT scan revealed an acute embolic infarction involving the right occipital lobe accompanied by marked mass effect [
Treatment: Endovascular embolization
The patient underwent successful endovascular embolization of the ruptured right occipital mycotic aneurysm; no craniotomy was required. After completing 6 weeks of nafcillin 2 g (IV) q4h (selected for its CNS penetration), he fully recovered.
DISCUSSION
There are significant differences between CNS findings for patients with SBE versus ABE [
SBE: Embolic strokes and mycotic aneurysms
With the more indolent SBE, patients rarely present with seizures attributed to peripheral septic emboli resulting in mycotic aneurysms that may leak, but rarely rupture [
ABE: Embolic strokes, ICH due to ruptured mycotic aneurysms, and microabscesses
Within 3 weeks of developing ABE, many patients initially present with intracranial embolic strokes. They may develop focal seizures, unexplained mental confusion, and/or retinal artery occlusion with accompanying visual loss.[
CONCLUSION
The cranial complications for patients with ABE versus SBE markedly differ. Those with ABE may develop unexplained seizures and cognitive dysfunction attributed to septic emboli/mycotic aneurysms, resulting in embolic strokes and massive ICH [
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Anderson DJ, Golstein LB, Wilkinson WE, Corey GR, Cabell CH, Sanders LL. Stroke location, characterization, severity and outcome in mitral vs aortic valve endocarditis. Neurology. 2003. 61: 1341-6
2. Brusch JL.editors. Infective Endocarditis. Cambridge, MA: Informa Healthcare; 2007. p. 143-181
3. Cantier MC, Mazighi M, Klein I, Desilles JP, Wolff M, Timsit JF. Neurologic complications of infective endocarditis: Recent findings. Curr Infect Dis Rep. 2017. 19: 41-
4. Cunha BA, Gill MV, Lazar JM. Acute infective endocarditis. Diagnostic and therapeutic approach. Infect Dis Clin North Am. 1996. 10: 811-34
5. Fernandez Guerrero ML, Gonzalez Lopez JJ, Goyenechea A, Fraile J, de Gorgolas M. Endocarditis caused by Staphyloccocus aureus. xA Reappraisal of the epidemiologic, clinical and pathologic manifestations with analysis of factors determining outcome. Medicine. 2009. 88: 1-22
6. Heiro M, Nikoskelainen J, Engblom E, Kotilainen E, Marttila R, Kotilainen P. Neurologic manifestations of infective endocarditis. A 17-year experience in a teaching hospital. Arch Intern Med. 2000. 160: 2781-7
7. Lerner PI. Neurologic complications of infective endocarditis. Med Clin North Am. 1985. 69: 385-98
8. Pruitt AA, Rubin RH, Karchmer AW, Duncan GW. Neurologic Complications of bacterial endocarditis. Medicine (Baltimore). 1978. 57: 329-43