- Department of Neurosurgery, “Riuniti” Hospital, Foggia, Puglia, Italy.
- Neurosurgical Unit, Miulli Hospital, Acquaviva delle Fonti, Puglia, Italy.
- Department of Neurosurgery, “Rummo” Hospital, Benevento, Campania, Italy.
- Department of Neurosurgery, University of Foggia, Foggia, Puglia, Italy.
Correspondence Address:
Francesco Carbone, Department of Neurosurgery, University of Foggia, Foggia, Puglia, Italy.
DOI:10.25259/SNI_964_2021
Copyright: © 2022 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Antonio Colamaria1, Matteo Sacco1, Savino Iodice1, Salvatore D’Oria2, Giovanni Parbonetti3, Francesco Carbone4, Matteo de Notaris3. Intradural extramedullary cavernous hemangioma of the cervicothoracic junction: A case report and review of the literature. 11-Feb-2022;13:53
How to cite this URL: Antonio Colamaria1, Matteo Sacco1, Savino Iodice1, Salvatore D’Oria2, Giovanni Parbonetti3, Francesco Carbone4, Matteo de Notaris3. Intradural extramedullary cavernous hemangioma of the cervicothoracic junction: A case report and review of the literature. 11-Feb-2022;13:53. Available from: https://surgicalneurologyint.com/surgicalint-articles/11378/
Abstract
Background: Intradural extramedullary cavernous hemangiomas of the spine are rare, benign lesions with only 40 published cases to date.
Case Description: The authors report a rare case of a histologically diagnosed intradural extramedullary cavernous hemangioma of the spine involving the cervicothoracic junction and causing sudden gait disturbances and urinary retention in a 24-year-old male. Gross total tumor removal allowed complete spinal decompression and sensible improvement of the clinical condition with no evidence of tumor relapse at 12-month follow-up examination.
Conclusion: More frequently found in the lower thoracic and lumbar spine, these tumors often cause subtle clinical manifestations including sensory and motor dysfunction secondary to nerve root compression; nonetheless, occasional cases of rapidly progressive worsening of the neurological condition with evidence of myelopathy and autonomic dysfunction have been described. In such cases, urgent surgical resection is crucial since the degree of neurological impairment and the time spanned from the onset of the symptoms are paramount for a good recovery.
Keywords: Case report, Cavernous hemangioma, Intradural spinal tumor, Vascular tumor
INTRODUCTION
Cavernous hemangiomas are uncommon, vascular tumors forming in virtually any area of the neuraxis[
In the present report, the authors describe a rare case of intradural extramedullary cavernous hemangioma located at the cervicothoracic junction causing rapidly progressive gait disturbances and acute urinary retention. Surgical posterior decompression and gross total tumor resection were followed by marked improvement of the clinical condition without radiological evidence of tumor recurrence at 12-month follow-up.
CASE REPORT
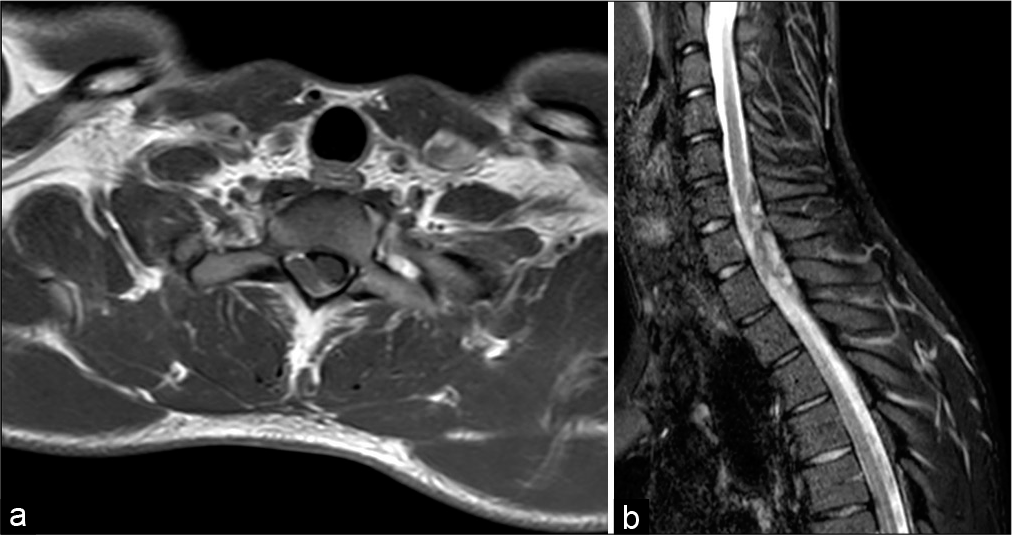
A 24-year-old male presented to the emergency department in March 2020 with acute urinary retention and fever without any other relevant medical history. The patient referred diffuse myalgia in the past 10 days and subsequent onset of painful dysesthesia in the left arm that was treated with corticosteroids. Physical examination revealed marked hyposthenia of the upper extremities, more pronounced in the left arm, and pain to deep manual palpation of the abdomen in the hypogastric region. Further examination disclosed a light spastic paraparesis and a decreased sensation of pain, position, and touch below T1 level, with negative Babinski’s signs bilaterally. Weak deep tendon reflexes (DTR) with slight difficulty in deambulation were also noticed. A urine sample was collected after the insertion of a Foley catheter, and microscopic examination revealed normal numbers of leukocytes and the absence of erythrocytes. Following the progressive worsening of the neurological state with sudden onset of gait instability, the patient was admitted to the neurological department where an emergency magnetic resonance imaging of the head and spine was performed. Spinal scans revealed an intradural, extramedullary tumor measuring 27 × 8 mm, located posterolateral to the spinal cord at C7-T1 level causing compression and dislocation without radiological evidence of myelopathy. The lesion was homogeneously hyperintense both in T1- and T2-weighted images, with dishomogeneous aspects in STIR sequences, suggesting a highly vascularized nature [
Figure 1:
Magnetic resonance of the spine. (a) Axial T1-weighted image showing an isointense intradural, extramedullary tumor, causing posterolateral compression of the spinal cord at C7-D1 level. (b) STIR sequence demonstrating dishomogeneous hyperintensity suggesting the highly vascularized nature of the lesion.
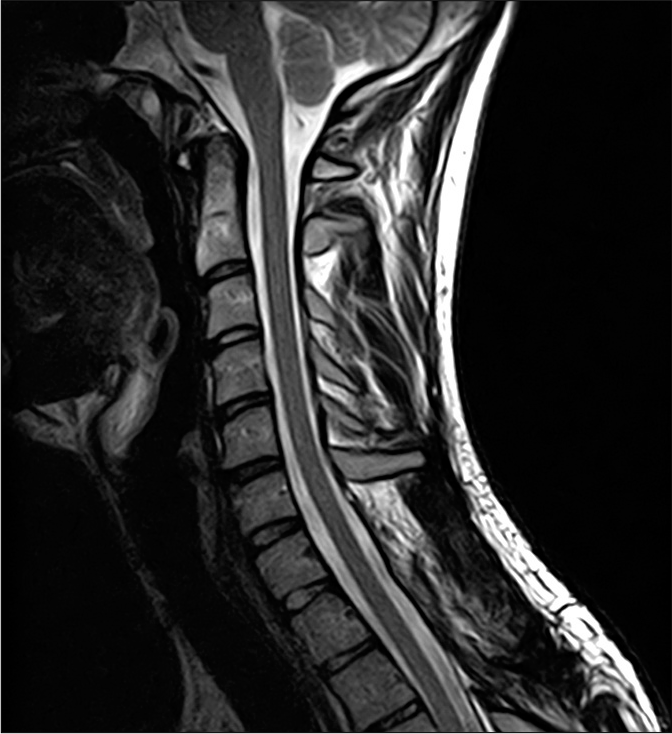
The postoperative course was uneventful and marked by the progressive improvement of the muscular strength as well as the absence of residual urinary retention. The patient was discharged 5 days after admission and referred to a rehabilitation center. At 12-month follow-up examination, the patient had good recovery of both motor and sensory functions, with no signs of residual hyposthenia of the upper extremities. Gait instability resolved completely and normal DTRs were observed. Contextual cervical spine magnetic resonance imaging (MRI) revealed no recurrence of the lesion and documented complete decompression of the spinal cord [
DISCUSSION
Intradural extramedullary cavernous hemangiomas of the spine are extremely rare tumors, with only 40 cases described to date.[
In the intradural extramedullary space, hemangiomas can arise from either root nerves, blood vessels, the inner layer of the dura mater, or the pial surface of the neuraxis.[
MRI is the investigation of choice for spinal lesions and thus also for intradural extramedullary tumors.[
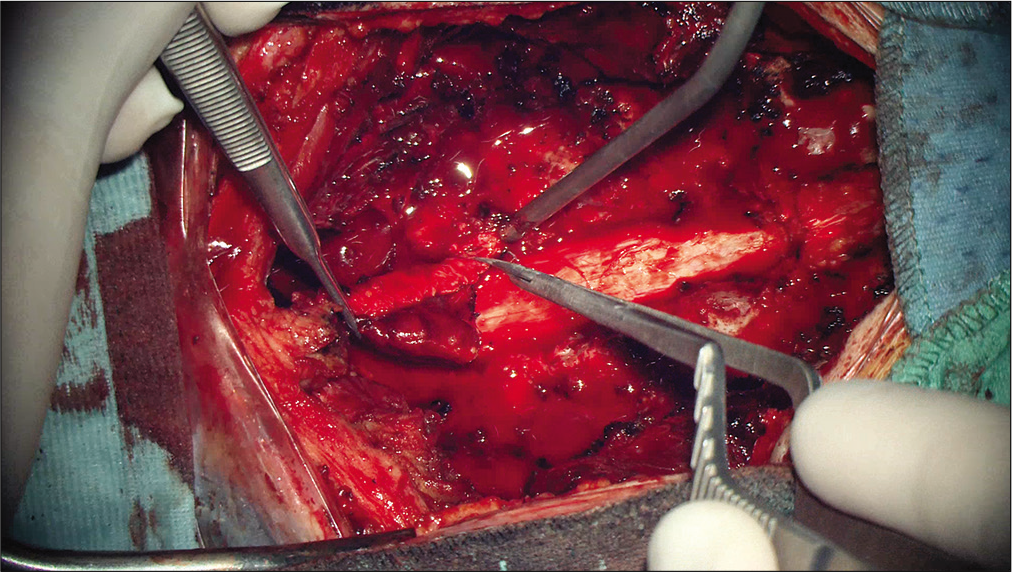
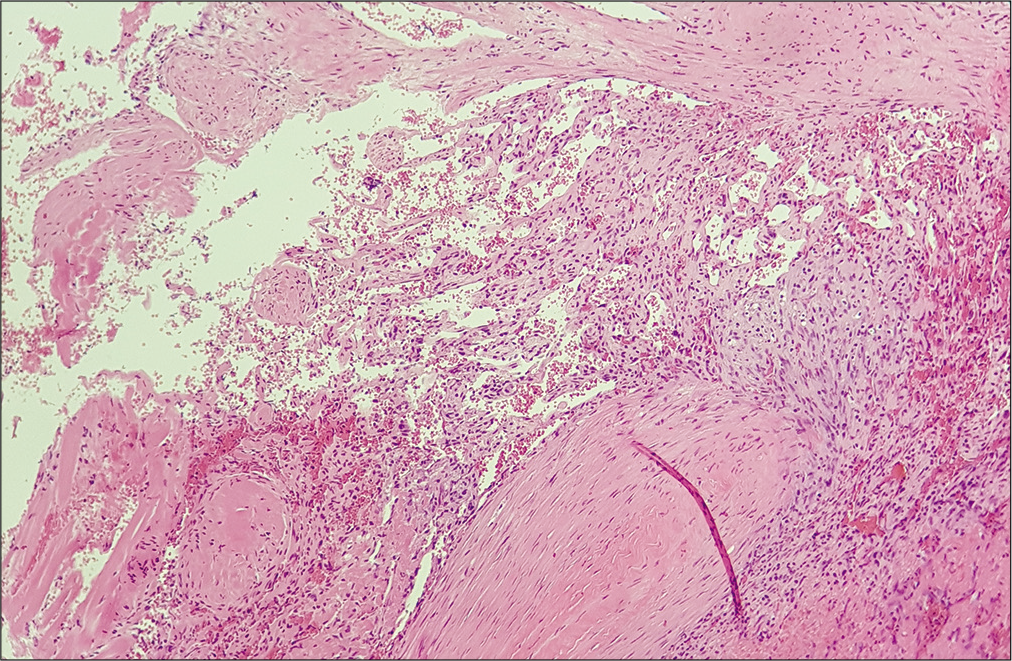
In the present report, the authors describe a rare case of intradural, extramedullary cavernous hemangioma located at the cervicothoracic junction in a 24-year-old patient. The patient presented with a short history of diffuse myalgia and painful dysesthesia of the left arm followed by sudden urinary retention and gait disturbances. MRI of the spine revealed a 27 × 8 mm tumor exhibiting dishomogeneous signal in T1- and T2-weighted images and peculiar features on STIR sequences. Posterior C7-T1 laminectomy followed by gross total resection of the lesion was performed and histological examination confirmed the diagnosis of cavernous hemangioma with thrombotic obliteration. Clinical and radiological follow-up showed complete decompression of the spinal cord without evidence of recurrency and full recovery of the motor and sensory function.
CONCLUSION
Despite the rareness, intradural extramedullary cavernous hemangiomas of the spine have to be taken into consideration as a potential cause in patients presenting with progressive spinal nerve compression syndromes or sudden signs of myelopathy. Although the interpretation of MRI may be complex, notably after an episode of microhemorrhage, it remains the investigation of choice for spinal lesions, especially for its ability to discern among several tumors, including meningiomas and schwannomas. When feasible, total surgical removal yields an excellent result and is the therapy of choice for symptomatic intradural extramedullary hemangiomas; nevertheless, a combination of subtotal or partial surgical excision and radiotherapy regimens has shown similar results.
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Abdullah DC, Raghuram K, Phillips CD, Jane JA, Miller B. Thoracic intradural extramedullary capillary hemangioma. AJNR Am J Neuroradiol. 2004. 25: 1294-6
2. Abe M, Tabuchi K, Tanaka S, Hodozuka A, Kunishio K, Kubo N, Nishimura Y. Capillary hemangioma of the central nervous system. J Neurosurg. 2004. 101: 73-81
3. Cervoni L, Celli P, Gagliardi FM. Cavernous angioma of the cauda equina: Report of two cases and review of the literature. Neurosurg Rev. 1995. 18: 281-3
4. Crispino M, Vecchioni S, Galli G, Olivetti L. Spinal intradural extramedullary haemangioma: MRI and neurosurgical findings. Acta Neurochir (Wien). 2005. 147: 1195-8
5. Grasso G, Alafaci C, Granata F, Cutugno M, Salpietro FM, Tomasello F. Thoracic spinal cord cavernous angioma: A case report and review of the literature. J Med Case Rep. 2014. 8: 271
6. Hanakita J, Suwa H, Nagayasu S, Suzuki H. Capillary hemangioma in the cauda equina: Neuroradiological findings. Neuroradiology. 1991. 33: 458-61
7. Harrison MJ, Eisenberg MB, Ullman JS, Oppenheim JS, Camins MB, Post KD. Symptomatic cavernous malformations affecting the spine and spinal cord. Neurosurgery. 1995. 37: 195-204
8. Mataliotakis G, Perera S, Nagaraju S, Marchionni M, Tzerakis N. Intradural extramedullary cavernoma of a lumbar nerve root mimicking neurofibroma. A report of a rare case and the differential diagnosis. Spine J. 2014. 14: e1-7
9. Merhemic Z, Stosic-Opincal T, Thurnher MM. Neuroimaging of spinal tumors. Magn Reson Imaging Clin North Am. 2016. 24: 563-79
10. Nowak DA, Widenka DC. Spinal intradural capillary haemangioma: A review. Eur Spine J. 2001. 10: 464-72
11. Padovani R, Acciarri N, Giulioni M, Pantieri R, Foschini MP. Cavernous angiomas of the spinal district: Surgical treatment of 11 patients. Eur Spine J. 1997. 6: 298-303
12. Roncaroli F, Scheithauer BW, Krauss WE. Capillary hemangioma of the spinal cord. Report of four cases. J Neurosurg. 2000. 93: 148-51
13. Tihan T, Chi JH, McCormick PC, Ames CP, Parsa AT. Pathologic and epidemiologic findings of intramedullary spinal cord tumors. Neurosurg Clin North Am. 2006. 17: 7-11
14. Tsao MN, Schwartz ML, Bernstein M, Halliday WC, Lightstone AW, Hamilton MG. Capillary hemangioma of the cavernous sinus. Report of two cases. J Neurosurg. 2003. 98: 169-74
15. Ueda S, Saito A, Inomori S, Kim I. Cavernous angioma of the cauda equina producing subarachnoid hemorrhage. Case report. J Neurosurg. 1987. 66: 134-6
16. Wurm G, Nussbaumer K, Kotzina L. Thoracic intra-and extramedullary cavernous hemangioma associated with innumerable cerebral vascular malformations: Case report and review of the literature. Cerebrovasc Dis. 2003. 16: 297-301
17. Ziechmann R, Zyck S, Krishnamurthy S, Galgano M. Intradural extramedullary thoracic cavernoma in a man with familial multiple cavernomas. Cureus. 2018. 10: e3115