- Professor of Clinical Neurosurgery, School of Medicine, State University of N.Y. at Stony Brook, New York, USA
- Chief of Neurosurgical Spine and Education, NYU Winthrop Hospital, NYU Winthrop NeuroScience, Mineola, New York, USA
Correspondence Address:
Nancy E. Epstein
Professor of Clinical Neurosurgery, School of Medicine, State University of N.Y. at Stony Brook, New York, USA
Chief of Neurosurgical Spine and Education, NYU Winthrop Hospital, NYU Winthrop NeuroScience, Mineola, New York, USA
DOI:10.4103/sni.sni_85_18
Copyright: © 2018 Surgical Neurology International This is an open access journal, and articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as appropriate credit is given and the new creations are licensed under the identical terms.How to cite this article: Nancy E. Epstein. Major risks and complications of cervical epidural steroid injections: An updated review. 23-Apr-2018;9:86
How to cite this URL: Nancy E. Epstein. Major risks and complications of cervical epidural steroid injections: An updated review. 23-Apr-2018;9:86. Available from: http://surgicalneurologyint.com/surgicalint-articles/major-risks-and-complications-of-cervical-epidural-steroid-injections-an-updated-review/
Abstract
Background:Too many patients, with or without significant cervical disease, unnecessarily undergo cervical epidural steroid injections (CESIs). These include interlaminar (ICESI) and transforaminal ESI (TF-CESI) injections that are not Food and Drug Administration (FDA) approved, have no documented long-term efficacy, and carry severe risks and complications.
Methods:Here we reviewed recent reports of morbidity and mortality attributed to the various types of CESIS. Major complications included; epidural hematomas, infection (abscess/meningitis), increased neurological deficits due to intramedullary (quadriparesis/quadriplegia), and intravascular injections (e.g., vertebral artery injections leading to cord, brain stem, and cerebellar strokes). The latter injections leading to strokes were typically attributed to the particulate steroid matter (e.g., within the methylprednisolone injection solution) that embolized into the distal arterial branches.
Results:Complications of cervical CESI/TF-CESI injections producing epidural hematoma, new neurological deficits (intramedullary injections), or intravascular injections resulting in strokes to the cord, brain stem, and cerebellum are often underreported. Interestingly, several other cases involving adverse events of CESI/TF-CESI may now be found in the medicolegal literature.
Conclusions:Cervical epidural injecions (e.g., CESI, ICESI, and TF-CESI) which are not FDA approved, provide no long-term benefit, and are being performed for minimal to no indications. They contribute to significant morbidity and mortality, including; epidural hematomas, infection, inadvertent intramedullary cord injections or cord, brain stem, and cerebellar strokes. Furthermore, these injections are increasingly required by insurance carriers prior to granting permission for definitive surgery, thus significantly delaying in some cases necessary operative intervention, while also subjecting patients at the hands of the insurance companies, to the additional hazards of these procedures.
Keywords: Cervical, coma, death, epidural steroid injections, infection, intramedullary, intravascular, paralysis, quadriplegia
INTRODUCTION
Many patients with even minor cervical complaints without significant magnetic resonance/computed tomography (MR/CT) documentation of significant cervical pathology are increasingly being subjected to high-risk cervical epidural steroid injections (CESIs) [e.g., interlaminar (ICESI) and transforaminal (TF-CESI)]. Major complications of these injections include; epidural hematoma, infection (abscess, meningitis), new neurological deficits (e.g., monoparesis to quadriplegia) due to intramedullary injections, and strokes to the spinal cord, brain stem, and/or cerebellum attributed to intravascular injections among others. Further, these cervical injections are still not approved by the Food and Drug Administration (FDA), and have not been proven to be either safe or effective in the cervical or lumbar spine.
LACK OF FDA APPROVAL OF CERVICAL EPIDURAL INJECTIONS WITHOUT DOCUMENTATION OF SAFETY/EFFICAY
Here we reviewed the literature between 2004–2013 and 2017–2018 regarding the severe risks/complications/mortality of CESIs, interlaminar (ICESI), and TF-CESI along with selective lumbar ESI (LESI) and TF-LESI. Further, cervical and lumbar epidural injections are still not approved by FDA as their safety/efficacy have never been established. In 2007, Abbasi et al. recommended performing prospective blinded randomized controlled trials (RCTs) to document such safety/efficacy; to date, none have yet been adequately completed [
SUMMARY OF RISKS/COMPICATIONS OF CERVICAL AND LUMBAR EPIDURAL INJECTIONS 2004-2013
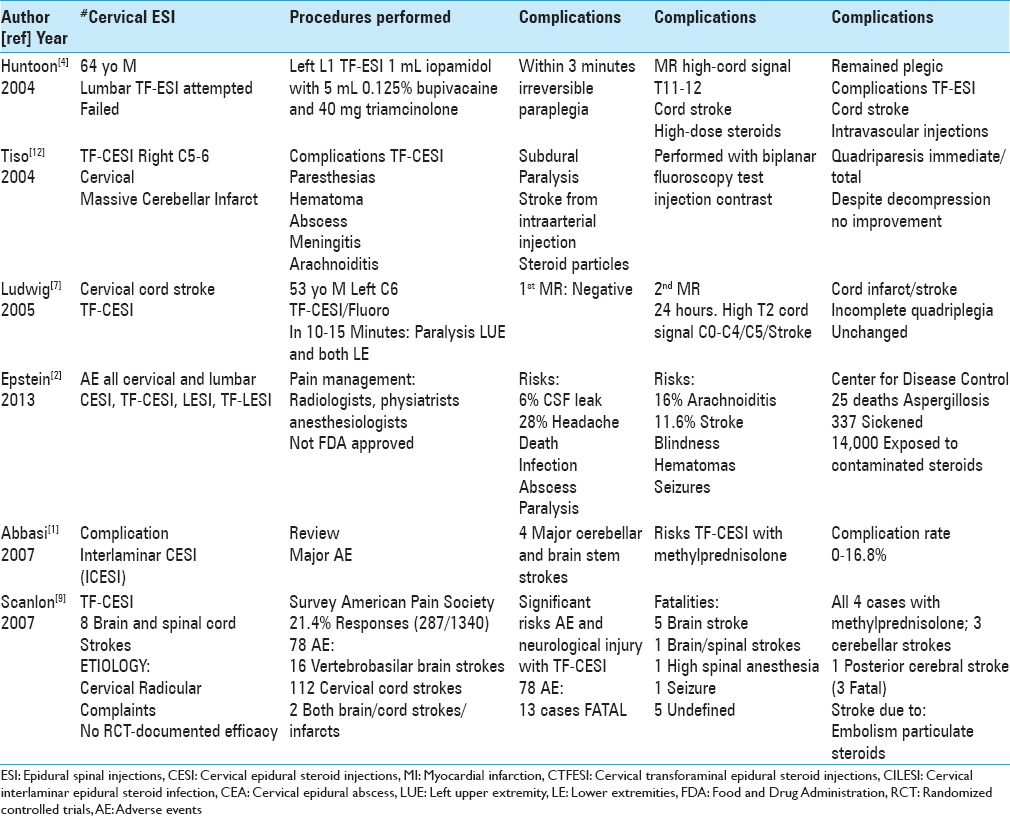
Multiple risk and complications of CESI (ICESI, TF-CESI) were reported between 2004 and 2013. In 2004, Tiso et al. quoted the major morbidity/mortality of TF-CESI that included; paresthesias, hematoma, epidural abscess, meningitis, arachnoiditis, subdural/subarachnoid injections, and intraarterial injections resulting in stroke [
SUMMARY OF RISKS/COMPLICATIONS OF CERVICAL AND LUMBAR EPIDURAL INJECTIONS 2017-2018
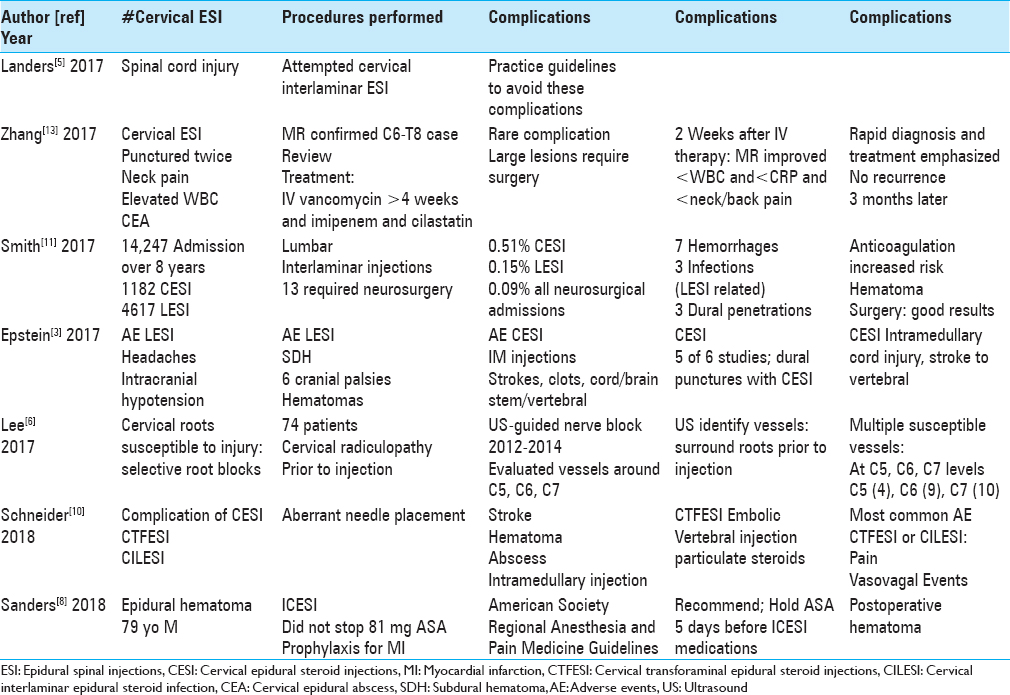
In 2017, Epstein (2017) again focused on the continued risks/complications of both cervical (CESI, ICESI, TF-CESI) and lumbar (LESI, ILESI, TF-LESI) ESI [
RISKS OF INTRAMEDULLARY EPIDURAL SPINAL INJECTIONS
Cervical ESIs and rarely high LESIs (e.g., CESI, ICESI, TF-CESI, TF-LESI) may result in inadvertent intramedullary cervical and thoracic/conus cord injections [Tables
CERVICAL INTRAVASCULAR EPIDURAL INJECTIONS
Several articles identified intravascular CESI resulting in major morbidity/mortality [
UTILIZING ULTRASOUND TO IDENTIFY VESSELS SURROUNDING C5-C7 CERVICAL NERVE ROOTS PRIOR TO SELECTIVE CERVICAL C5-C7 NERVE ROOT BLOCKS
Lee et al. (2017) utilized ultrasound (US) to document the size/location/number of vulnerable and susceptible perineural blood vessels prior to performing selective nerve root blocks from C5 to C7 (2012–2014) [
HIGH LUMBAR L1 TRANSFORAMINAL ESI INJECTION/CORD STROKE
In 2004, Huntoon and Matin (2004) identified a 64-year-old male with a history of multiple prior lumbar operations, who underwent a left L1 TF-LESI; it contained 1 mL of iopamidol (Isovue), 5 mL of 0.125% bupivacaine, and 40 mg of triamcinolone [
INCIDENCE OF HEMORRHAGES DUE TO VARIOUS CERVICAL/LUMBAR EPIDURAL STEROID INJECTIONS
Two studies cited the risks of peri-procedural hematomas in patients undergoing CESI and LESI [
CERVICAL EPIDURAL ABSCESS DUE TO CESI
Zhang et al. (2017) noted that CESI rarely result in cervical epidural abscess (CEA).[
CONCLUSION
Cervical ESI (CESI, ICESI, TF-CESI) and lumbar ESI (LESI, ILESI, TF-LESI) are not FDA-approved and have no RCT-documented long-term safety or efficacy. Further, CESI and occasionally high LESI (e.g., TF-LESI at the L1 level) risks include severe morbidity and mortality. Intramedullary or intravascular injections result in irreversible paralysis and/or strokes (e.g., cord, brain stem, and cerebellar strokes).
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Abbasi A, Malhotra G, Malanga G, Elovic EP, Kahn S. Complications of interlaminar cervical epidural steroid injections: A review of the literature. Spine (Phila Pa 1976). 2007. 32: 2144-51
2. Epstein NE. The risks of epidural and transforaminal steroid injections in the Spine: Commentary and a comprehensive review of the literature. Surg Neurol Int. 2013. 4: S74-93
3. Epstein NE. Neurological complications of lumbar and cervical dural punctures with a focus on epidural injections. Surg Neurol Int. 2017. 8: 60-
4. Huntoon MA, Martin DP. Paralysis after transforaminal epidural injection and previous spinal surgery. Reg Anesth Pain Med. 2004. 29: 494-5
5. Landers MH. Spinal cord injury during attempted cervical interlaminar epidural injection of steroids. Pain Med. 2017. p.
6. Lee HH, Park D, Oh Y, Ryu JS. Ultrasonography evaluation of vulnerable vessels around cervical nerve roots during selective cervical nerve root block. Ann Rehabil Med. 2017. 41: 66-71
7. Ludwig MA, Burns SP. Spinal cord infarction following cervical transforaminal epidural injection: A case report. Spine (Phila Pa 1976). 2005. 30: E266-8
8. Sanders RA, Bendel MA, Moeschler SM, Mauck WD. Epidural hematoma following interlaminar epidural injection in patient taking aspirin. Reg Anesth Pain Med. 2018. p.
9. Scanlon GC, Moeller-Bertram T, Romanowsky SM, Wallace MS. Cervical transforaminal epidural steroid injections: More dangerous than we think?. Spine (Phila Pa 1976). 2007. 32: 1249-56
10. Schneider BJ, Maybin S, Sturos E. Safety and Complications of Cervical Epidural Steroid Injections. Phys Med Rehabil Clin N Am. 2018. 29: 155-69
11. Smith GA, Pace J, Strohl M, Kaul A, Hayek S, Miller JP. Rare Neurosurgical complications of epidural injections: An 8-yr single-institution experience. Oper Neurosurg (Hagerstown). 2017. 13: 271-9
12. Tiso RL, Cutler T, Catania JA, Whalen K. Adverse central nervous system sequelae after selective transforaminal block: The role of corticosteroids. Spine J. 2004. 4: 468-74
13. Zhang JH, Wang ZL, Wan L. Cervical epidural analgesia complicated by epidural abscess: A case report and literature review. Medicine (Baltimore). 2017. 96: e7789-






Julian Carrillo
Posted May 2, 2018, 12:07 pm
Do you have any information about de cryo or radiofrequency complucations??
RR
Posted September 16, 2018, 5:24 pm
Do not under any circumstance take a Cervical Epidural !!!!!! You can be damaged for life. FYI
RR
Patrick peng
Posted November 1, 2018, 5:11 am
I did nearly 20 cervical epidural steroid every week for years and didn’t have mentioned complication even once.l was an anesthesiologist. I did epidural injection by standard method for epidural anesthesia. I always put an epidural catheter into epidural space before injecting testing lidocaine or discipline. It’s safe to avoid giving drugs into intramural space or vessels. If you give testing lidocaine 0.5-1
cc you can find a lot of textbook-unmentioned hypothesis or truth.
Paula Riscoe
Posted April 17, 2019, 11:32 am
Im supposed to get a cervical epidural injection next week are they dangerous Can I be paralyzed?
Tammy Rondalain Fields
Posted June 28, 2019, 12:58 pm
Hi, I’ve been getting cervical and lumbar injections here in Indiana for 19 years. My doctor is a pain management/ anesthesiologist. My question is, what should I do the day or after an injection? What are any warning signs if any days after? Thank you for any assistance you can provide.
jim
Posted June 28, 2019, 1:19 pm
Our apologies, but we are not able to offer medical advice beyond what can be read in our published articles. We wish you the best.
Melissa
Posted July 3, 2019, 11:24 am
I had a cervical injection for a ruptured disc in my neck several months ago. About a month or 2 after the injection my skin started burning & severely itching when i would get hot, just before a sweat. No hives, whelps, bumps, nothing, just the feeling. I told my pain management Dr. about it. They suggested I might be starting with SHINGLES!! That never happened. My question is, have you heard of this happening in any on your studies? If so, please, please, I am so very miserable, what can I do to stop it.
RodButler
Posted July 18, 2019, 2:11 pm
Is there any evidence of steroid epidural injections may cause eye damage? A neuroopthomologist diagnosed a friend with vision damage and suspects the steroid epidural procedure as a likely cause because of body sensitivity to steroids causing this side effect.
Laurie
Posted September 7, 2019, 7:02 am
I am a glaucoma suspect who has lost field vision due to these injections. I think the pain clinics are moneymakers for the hospitals and a way to decrease dependence on opioids. No follow up to see how I was feeling was ever done after the injection. I was never advised to have my eye pressure and vision checked. A sizable percentage of patients develop steroid inducted glaucoma and the pain clinic either do not know or care.
Jen
Posted October 18, 2019, 12:46 pm
I have a cervical epidural scheduled for Nov 6, and after reading these posts I’m seriously considering cancelling it. My C6 (anterior tubercle) was fractured in an auto accident back in April. I have had headaches that start at the base of my neck and travel up the back of my head to my forehead and temples. My PA at Resurgens says the CESI is the only thing that can help, since PT wasn’t much help. I’m scared after reading this article though and don’t know what to do.
Stephanie Wolfram
Posted November 2, 2019, 11:32 pm
Jen, sorry to hear, sounds familiar, but these injections are not your only option. I’m looking into homeopathy. For migraines & fibromyalgia &_nerve pain. They look at your whole body and mind for a holistic treatment. Best of luck
Cheri WALKER
Posted December 20, 2019, 9:35 pm
Dont do it. I suffer from c5,c6,c7 damage causing both arms to go numb and constant headaches. I just had an injection at c6 and when i COUGH it feels like my spine is dislocating. Pain across my back from shoulder to shoulder and neck bulge feels twice bigger to where i can barely turn my head of lay down comfortably
John
Posted January 15, 2020, 3:38 pm
Hi I had a eperderal steroid injectionfor c5-c7I was sent to the hospital for 10 days,I couldn’t walk the first 4 days,one doctor said it was a bad reaction to lidocaine,which I’ve had 4 injections before this so that couldn’t be it,then another doctor said my spine was irritated and nicked,could anyone explain and let me know what you think,the doctors are so worried about the doctor that gave the injection,more the what’s wrong with me.i can not get a straight answer.
Victoria L Bankston
Posted January 22, 2020, 7:14 am
I am a 58 yr old female. Have had C4-5 fusion @ 16. And cadaver bone w/plate & screws@ C5-6 in 2016. Now they want to do ECI at C6-7.