- Department of Neurosurgery, Baylor College of Medicine, Houston, Texas, USA
Correspondence Address:
Edward A. M. Duckworth
Department of Neurosurgery, Baylor College of Medicine, Houston, Texas, USA
DOI:10.4103/2152-7806.157659
Copyright: © 2015 Patel AJ. This is an open-access article distributed under the terms of the Creative Commons Attribution-Noncommercial-Share Alike 3.0 Unported, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.How to cite this article: Patel AJ, M. Duckworth EA. Management of infections complicating the orbitocranial approaches: Report of two cases and review of literature. Surg Neurol Int 26-May-2015;6:89
How to cite this URL: Patel AJ, M. Duckworth EA. Management of infections complicating the orbitocranial approaches: Report of two cases and review of literature. Surg Neurol Int 26-May-2015;6:89. Available from: http://surgicalneurologyint.com/surgicalint_articles/management-infections-complicating-orbitocranial/
Abstract
Background:The orbitocranial approaches are now indispensible for treating lesions of the skull base, providing access to lesions in the anterior and middle cranial fossae, as well as the upper clivus and anterior brainstem. The management of infectious complications of the orbitocranial approaches, however, has evaded the literature.
Case Description:We present two cases of patients who underwent orbitocranial approach whose clinical course was complicated by wound infection and osteomyelitis. One patient was treated with antibiotics and then had a custom implant placed for cranioplasty. The other case was managed with removal of bone and wire-mesh cranioplasty.
Conclusion:Management of orbitocraniotomy infections can be difficult due to the complex geometry of the flap and to cosmetic considerations. Once the infection involves the bone, the bone can be replaced after cleaning or discarded and a cranioplasty performed. Cranioplasty can be performed with wire-mesh or a custom implant made by computer-assisted modeling.
Keywords: Cranioplasty, frontotemporal-orbitozygomatic, infection, orbitozygomatic
INTRODUCTION
The frontotemporal-orbitozygomatic (FTOZ) approach, initially described by Pellerin et al.[
Surgical technique is paramount in minimizing morbidity and optimizing the cosmetic outcome after such an extensive approach, although only a few reports focus on this.[
CASE REPORTS
Case 1
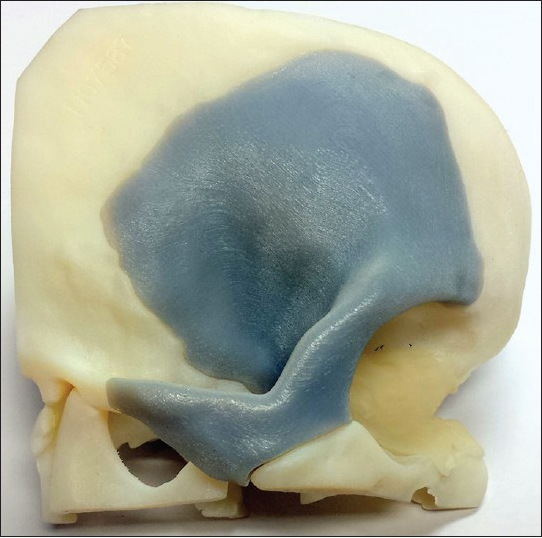
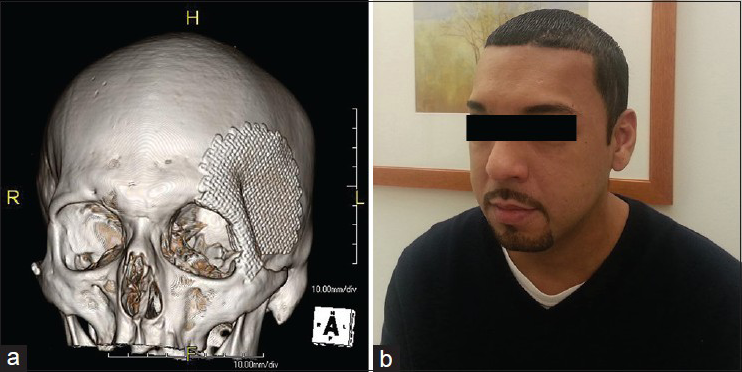
A 58-year-old male was found to have a sellar/suprasellar mass that he refused to have treated for many years. He then presented to the emergency room (ER) with near blindness bilaterally, decreased level of consciousness, dilated right pupil, and left facial paresis. Imaging studies demonstrated a very large sellar/suprasellar mass with brainstem compression. The patient underwent right cranio-orbitozygomatic approach for subtotal resection of the mass. Pathology was consistent with craniopharyngioma. Several months later, the patient had recurrence of a tumor cyst and some re-growth of solid tumor, for which he underwent a second resection via the same approach. Six weeks later, the patient returned to clinic with right facial and forehead swelling with associated periorbital edema and drainage from the wound. The patient was taken to the operating room for washout and debridement of the wound. The infected bone flap was removed and the patient treated with a 6-week course of culture-specific intravenous antibiotics. The removed, infected bone flap was then imaged ex vivo using thin cut computed tomography (CT) to make a custom one piece FTOZ implant, which would be guaranteed to fit. Three months later, the patient underwent cranioplasty with the custom implant and tolerated the procedure well [
Case 2
A 32-year-old male suffered a seizure and was found to have a large fronto-parietal AVM with a flow-related anterior communicating artery aneurysm. The patient underwent left orbitopterional craniotomy for clipping of the aneurysm. He had a very large frontal sinus that was noted preoperatively. In surgery, this was cranialized. He did well in the initial postoperative period, however, 2 weeks later, he returned with swelling at the surgical site and fluctuance. Imaging studies demonstrated epidural fluid collection and possible bony involvement. The patient was taken to the operating room for washout. The bone flap was noted to be infected and was then used as a template to mold a piece of titanium mesh that was affixed to the skull to recreate the contours of the orbitopterional bone flap. The patient tolerated the procedure well and his postoperative course was uncomplicated. He underwent 6 weeks of culture-specific IV antibiotics. In follow-up, the patient has had an excellent cosmetic outcome [
DISCUSSION
The orbitocranial approaches are a versatile and commonly used skull base approach that provides access to the anterior fossa, middle fossa, and posterior fossa anterior to the brainstem. The benefit of increased exposure and decreased brain retraction comes with the drawback of added morbidity and complication in a cosmetically sensitive area.[
The incidence of postoperative wound infection after craniotomy has been reported to be 3–5%.[
Involvement of the bone can be identified based on imaging, but is usually determined by intraoperative findings. CT is the best, commonly used imaging modality, with osteopenia, erosion of the cortex, or lytic destruction being diagnostic. Radionuclide scanning as well as positron emission tomography (PET) scan has also been shown to be sensitive in detecting osteomyelitis.[
If the bone is thought to be involved, the wound is washed out and debrided, the devitalized bone flap removed traditionally,[
Replacing devitalized bone after cleaning
While there have been many improvements in synthetic cranioplasty materials over the years, replacing the native bone flap is ideal when possible. Traditionally, the standard of care was to discard infected bone flaps, followed by a delayed cranioplasty.[
Wire-mesh cranioplasty
Wire-mesh cranioplasty has been used successfully for a long time. Methylmethacrylate or cement can be used to augment the repair and further contour as necessary to achieve a good cosmetic result. Wire-mesh can be used to perform the cranioplasty at the time of washout,[
Reproducing original bone flap using custom implant
Custom implants are generally made using computer-assisted modeling, and can be made of polymethylmethacrylate (PMMA),[
CONCLUSION
The orbitocranial approaches are an important approach in treating lesions of the skull base by allowing less brain retraction. However, the management of infectious complications of orbitocranial approach has evaded the literature. We report two patients who underwent an orbitocranial approach complicated by wound infection and osteomyelitis. Once the infection involves the bone, the bone can be replaced after cleaning or discarded and a cranioplasty performed. Cranioplasty can be performed with wire-mesh or a custom implant made by computer-assisted modeling.
References
1. Al-Mefty O. Supraorbital-pterional approach to skull base lesions. Neurosurgery. 1987. 21: 474-7
2. Alaywan M, Sindou M. Fronto-temporal approach with orbito-zygomatic removal. Surgical anatomy. Acta Neurochir. 1990. 104: 79-83
3. Andaluz N, Van Loveren HR, Keller JT, Zuccarello M. Anatomic and clinical study of the orbitopterional approach to anterior communicating artery aneurysms. Neurosurgery. 2003. 52: 1140-8
4. Auguste KI, McDermott MW. Salvage of infected craniotomy bone flaps with the wash-in, wash-out indwelling antibiotic irrigation system. Technical note and case series of 12 patients. J Neurosurg. 2006. 105: 640-4
5. Balasingam V, Noguchi A, McMenomey SO, Delashaw JB Jr. Modified osteoplastic orbitozygomatic craniotomy.Technical note. J Neurosurg. 2005. 102: 940-44
6. Balch RE. Wound infections complicating neurosurgical procedures. J Neurosurg. 1967. 26: 41-5
7. Blomstedt GC. Craniotomy infections. Neurosurg Clin N Am. 1992. 3: 375-85
8. Blumenkopf B, Hartshorne MF, Bauman JM, Cawthon MA, Patton JA, Friedman AH. Craniotomy flap osteomyelitis: A diagnostic approach. J Neurosurg. 1987. 66: 96-101
9. Bruce JN, Bruce SS. Preservation of bone flaps in patients with postcraniotomy infections. J Neurosurg. 2003. 98: 1203-7
10. Cabraja M, Klein M, Lehmann TN. Long-term results following titanium cranioplasty of large skull defects. Neurosurg Focus. 2009. 26: E10-
11. Chiang HY, Steelman VM, Pottinger JM, Schlueter AJ, Diekema DJ, Greenlee JD. Clinical significance of positive cranial bone flap cultures and associated risk of surgical site infection after craniotomies or craniectomies. J Neurosurg. 2011. 114: 1746-54
12. Delashaw JB, Tedeschi H, Rhoton AL. Modified supraorbital craniotomy: Technical note. Neurosurgery. 1992. 30: 954-6
13. Delgado-Lopez PD, Martin-Velasco V, Castilla-Diez JM, Galacho-Harriero AM, Rodriguez-Salazar A. Preservation of bone flap after craniotomy infection. Neurocirugia. 2009. 20: 124-31
14. DeMonte F, Tabrizi P, Culpepper SA, Suki D, Soparkar CN, Patrinely JR. Ophthalmological outcome after orbital entry during anterior and anterolateral skull base surgery. J Neurosurg. 2002. 97: 851-6
15. Geraghty J, Feely M. Antibiotic prophylaxis in neurosurgery. A randomized controlled trial. J Neurosurg. 1984. 60: 724-6
16. Gerber N, Stieglitz L, Peterhans M, Nolte LP, Raabe A, Weber S. Using rapid prototyping molds to create patient specific polymethylmethacrylate implants in cranioplasty. Conf Proc IEEE Eng Med Biol Soc 2010. 2010. p. 3357-60
17. Gonzalez LF, Crawford NR, Horgan MA, Deshmukh P, Zabramski JM, Spetzler RF. Working area and angle of attack in three cranial base approaches: Pterional, orbitozygomatic, and maxillary extension of the orbitozygomatic approach. Neurosurgery. 2002. 50: 550-5
18. Haines SJ, Goodman ML. Antibiotic prophylaxis of postoperative neurosurgical wound infection. J Neurosurg. 1982. 56: 103-5
19. Hakuba A, Liu S, Nishimura S. The orbitozygomatic infratemporal approach: A new surgical technique. Surg Neurol. 1986. 26: 271-6
20. Hakuba A, Tanaka K, Suzuki T, Nishimura S. A combined orbitozygomatic infratemporal epidural and subdural approach for lesions involving the entire cavernous sinus. J Neurosurg. 1989. 71: 699-704
21. Hill CS, Luoma AM, Wilson SR, Kitchen N. Titanium cranioplasty and the prediction of complications. Br J Neurosurg. 2012. 26: 832-7
22. Hlavin ML, Kaminski HJ, Fenstermaker RA, White RJ. Intracranial suppuration: A modern decade of postoperative subdural empyema and epidural abscess. Neurosurgery. 1994. 34: 974-80
23. Jane JA, Park TS, Pobereskin LH, Winn HR, Butler AB. The supraorbital approach: Technical note. Neurosurgery. 1982. 11: 537-42
24. Korinek AM. Risk factors for neurosurgical site infections after craniotomy: A prospective multicenter study of 2944 patients. The French Study Group of Neurosurgical Infections, the SEHP, and the C-CLIN Paris-Nord. Service Epidemiologie Hygiene et Prevention. Neurosurgery. 1997. 41: 1073-9
25. Koslow M, Ransohoff J. Primary wire mesh cranioplasty in flap infections. Neurosurgery. 1979. 4: 290-1
26. Lethaus B, Safi Y, ter Laak-Poort M, Kloss-Brandstatter A, Banki F, Robbenmenke C. Cranioplasty with customized titanium and PEEK implants in a mechanical stress model. J Neurotrauma. 2012. 29: 1077-83
27. McDermott MW, Durity FA, Rootman J, Woodhurst WB. Combined frontotemporal-orbitozygomatic approach for tumors of the sphenoid wing and orbit. Neurosurgery. 1990. 26: 107-16
28. Pellerin P, Lesoin F, Dhellemmes P, Donazzan M, Jomin M. Usefulness of the orbitofrontomalar approach associated with bone reconstruction for frontotemporosphenoid meningiomas. Neurosurgery. 1984. 15: 715-8
29. Saringer W, Nobauer-Huhmann I, Knosp E. Cranioplasty with individual carbon fibre reinforced polymere (CFRP) medical grade implants based on CAD/CAM technique. Acta Neurochir. 2002. 144: 1193-203
30. Schwartz MS, Anderson GJ, Horgan MA, Kellogg JX, McMenomey SO, Delashaw JB. Quantification of increased exposure resulting from orbital rim and orbitozygomatic osteotomy via the frontotemporal transsylvian approach. J Neurosurg. 1999. 91: 1020-6
31. Staffa G, Barbanera A, Faiola A, Fricia M, Limoni P, Mottaran R. Custom made bioceramic implants in complex and large cranial reconstruction: A two-year follow-up. J Craniomaxillofac Surg. 2012. 40: e65-70
32. Tenney JH, Vlahov D, Salcman M, Ducker TB. Wide variation in risk of wound infection following clean neurosurgery. Implications for perioperative antibiotic prophylaxis. J Neurosurg. 1985. 62: 243-7
33. van der Bruggen W, Bleeker-Rovers CP, Boerman OC, Gotthardt M, Oyen WJ. PET and SPECT in osteomyelitis and prosthetic bone and joint infections: A systematic review. Semin Nucl Med. 2010. 40: 3-15
34. Youssef AS, Willard L, Downes A, Olivera R, Hall K, Agazzi S. The frontotemporal-orbitozygomatic approach: Reconstructive technique and outcome. Acta Neurochir. 2012. 154: 1275-83
35. Zabramski JM, Kiris T, Sankhla SK, Cabiol J, Spetzler RF. Orbitozygomatic craniotomy. Technical note. J Neurosurg. 1998. 89: 336-41