- Department of Anesthesiology, Advocate Illinois Masonic Medical Center, Chicago, Illinois, USA
- Department of Anesthesiology, JHS Hospital of Cook County, Chicago, Illinois, USA
- Ghaly Neurosurgical Associates, Aurora, Chicago, Illinois, USA
- Department of Anesthesiology, University of Illinois, Chicago, Illinois, USA
Correspondence Address:
Ramsis F. Ghaly
Department of Anesthesiology, Advocate Illinois Masonic Medical Center, Chicago, Illinois, USA
Department of Anesthesiology, University of Illinois, Chicago, Illinois, USA
DOI:10.4103/2152-7806.198737
Copyright: © 2017 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Ramsis F. Ghaly, Tatiana Tverdohleb, Kenneth D. Candido, Nebojsa Nick Knezevic. Management of parturients in active labor with Arnold Chiari malformation, tonsillar herniation, and syringomyelia. 19-Jan-2017;8:10
How to cite this URL: Ramsis F. Ghaly, Tatiana Tverdohleb, Kenneth D. Candido, Nebojsa Nick Knezevic. Management of parturients in active labor with Arnold Chiari malformation, tonsillar herniation, and syringomyelia. 19-Jan-2017;8:10. Available from: http://surgicalneurologyint.com/surgicalint_articles/management-of-parturients-in-active-labor-with-arnold-chiari-malformation-tonsillar-herniation-and-syringomyelia/
Abstract
Background:Arnold-Chiari malformation Type 1 (ACM-1) in parturients is a topic of ongoing discussion between obstetricians and anesthesiologists. The primary unanswered question remains; How should the anesthesia provider proceed with labor analgesia and anesthesia for cesarean section when confronted with an advanced, asymptomatic, or minimally symptomatic case of ACM-1 during labor?
Case Description:A 24-year-old, ASA II, G1P0 full-term parturient presented to Labor and Delivery for vaginal delivery. A diagnosis of ACM-1 was made 12 years ago when a brain magnetic resonance imaging (MRI) was performed for right-sided numbness following a rear-end motor vehicle collision. The patient had been asymptomatic since then and had been seen by an outside neurologist frequently for the past 10 years. During the anesthesia evaluation, it was noted that she had an exaggerated patellar reflex, and a questionable left-sided Babinski; subsequently, an MRI study was requested. Review of a brain MRI demonstrated an advanced form of ACM with a 1.7 cm transtonsillar herniation and a large syrinx extending from C1 down to C5. Following a discussion with the patient, family, and primary OB team, a plan for elective cesarean section was made per neurosurgical recommendations. This was conducted uneventfully under general anesthesia. The patient had no complaints in the post-anesthesia care unit.
Conclusion:Unfamiliarity of health care providers with regards to ACM-1 parturients can be countered by increasing awareness of this condition throughout medical specialties involved in their care. The Ghaly Obstetric Guide to Arnold-Chiari malformation Type 1, along with proper training of anesthesia care providers regarding the specificities of ACM-1 parturients aids in better management and understanding of this complex condition.
Keywords: ACM-1, anesthesia, delivery, multidisciplinary, parturient
INTRODUCTION
Arnold-Chiari malformation Type 1 (ACM-1) in parturients is a topic of ongoing discussion between obstetricians and anesthesiologists. Several studies have been conducted on the optimal anesthetic management of these patients during labor and delivery, however, no consensus has been reached. “How should the anesthesia provider proceed when confronted with an advanced, asymptomatic or minimally symptomatic case of ACM-1 during labor?,” remains an unanswered clinical question.
ACM-1, the most common of all four types, is a neurosurgical condition that is characterized by downward herniation of the hindbrain into the foramen magnum.[
Arnold-Chiari malformation (ACM) was first identified in 1883 by Cleland, and was first decribed and classified into four types in 1891 by Chiari. The mechanism of ACM-1 remains uncertain; however, studies support the role of the posterior cranial fossa morphometry, describing underdevelopment of the occipital bone, which causes overcrowding of the normally developed hindbrain in the posterior cranial fossa.[
Increased frequency in magnetic resonance imaging (MRI) use has revealed a higher incidence of ACM-1 than initially thought, accounting for 0.6–0.7% of the cases.[
Symptomatology
ACM-1 presents with different degrees of neurologic manifestations that can be evident during the second or third decades of life. The most commonly reported symptoms are suboccipital headache (81%), occular manifestations (78%), otoneurologic disturbances (74%), pain in the extremities, cranial nerves disturbances (hoarseness, cough, dysphagia), weakness and muscle atrophy, and sensory disturbances (numbness, pain, and temperature deficits).[
Clinical diagnosis
MRI or computed tomography (CT) scan are usually sufficient to determine the position of cerebellar tonsils, abnormalities in the posterior fossa, and the presence of syringomyelia.[
Another important component of the ACM-1 is the disturbance in CSF dynamics which can be assessed through CSF fluid flow MRI.[
On imaging studies, syringomyelia is commonly detected at the C4-C6 spinal cord levels, however, it can involve the entire length of the spinal cord.[
Obstetric concerns in ACM-I
ACM-1 is of a significant clinical concern in pregnant patients, first because of physiological increase in CSF pressure associated with pregnancy, and second because delivery represents a vulnerable period that puts parturients at a great risk of complications due to an acute increase in intracranial pressure (ICP). The findings of the Meadows et al. study alerts us to the fact that an advanced form of ACM-1 with minimal symptoms or advanced asymptomatic ACM-1 can lead to catastrophic consequences during labor and delivery.[
Some authors have suggested that “pushing” during labor increases CSF pressure by approximately 20–51 mmHg; however, Marx et al. suggested that the increase in CSF pressure occcurs due to the pain experienced by the patient during uterine contractions.[
Throughout the course of the first stage of labor, when the cervix effaces and dilates (early labor), and uterine contractions are becoming longer, stronger, and closer together (active labor), it was found that the basal CSF pressure was only 13 mmHg ± 2.5 mmHg after eliminating the pain factor.[
During the second stage of the labor (full cervical dilatation followed by delivery of the baby) and third stage (delivery of placenta), where most of the “pushing” takes place, the CSF pressure increases, on average, by 8 mmHg or by 20–25% at the time of delivery.[
Although some authors state that, it is not known whether uterine contractions and “pushing” during labor worsen the ACM-1 condition or not, the obstetric and anesthesiologic management of such patients remains a dilemma.[
Despite the lack of evidence-based guidelines and no uniform recommendations regarding management of these patients throughout pregnancy and delivery, a multidisciplinary approach is implemented based on best clinical judgement and in-hospital protocols. We present a case of ACM-1 where the importance of a multidisciplinary team approach, including anesthesiologists, neurosurgeons, and obstetricians can actively intervene in the diagnostic process and mode of delivery to prevent possible severe morbidity and mortality.
CASE DESCRIPTION
A 24-year-old, ASA II, G1P0 full-term parturient presented to the Labor and Delivery for vaginal delivery. Her past medical history was significant for gestational diabetes and ACM-1.
A diagnosis of ACM-1 was made 12 years ago when a brain MRI was performed for right-sided numbness developing following a motor vehicle collision. The patient had been entirely asymptomatic since then, and had been seen by an outside neurologist frequently for the past 10 years. She was assured by her neurologist 2 months prior that vaginal delivery and labor epidural placement would be safe, however she had not been seen by an anesthesiologist in the anesthesia preop clinic before.
In the 39th week of pregnancy, she presented to our hospital for rupture of membranes and uterine contractions; anesthesia was consulted for an epidural placement. During the anesthesia evaluation, it was noted that she had an exaggerated patellar reflex and a questionable left-sided Babinski; subsequently, MRI images were requested from an outside hospital. A thorough review of the brain MRI demonstrated an advanced form of ACM-1 with 1.7 cm transtonsillar herniation and a large syrinx extending down from C1 to C5. Concerned with the progression of the transtonsillar herniation during labor, an urgent neurosurgery consult was requested. Following a discussion with the patient, family, primary OB team, and the neurosurgeon, a plan for cesarean section was made per neurosurgical recommendations, including avoidance of neuraxial anesthesia and use of careful laryngeal manipulation and endotracheal intubation.
A bolus dose of terbutaline was given to stop active contractions and the patient was brought to the operation room. The patient was placed in a left lateral uterine displacement position, and standard ASA monitors were applied. Concern regarding further progression of the herniation due to increased ICP during head and neck movement and laryngoscopy, led to an awake fiberoptic intubation was performed under generous topical anesthesia consisting of 6 ml of 1% lidocaine spray. A multimodal general anesthesia was used with the mixture of 1 MAC of desflurane, oxygen, and air and controlled ventilation. Mild hyperventilation was provided to ensure an end-tidal CO2 of 30 mmHg to prevent a possible increase in ICP. A healthy infant male was delivered with Apgar scores at 1 and 5 minutes of 9/9. After umbilical cord clamping, an infusion of mannitol 0.5 mg/kg and dexamethasone 10 mg was given.
The remainder of the case was without incident. Post-extubation neurological assessment was grossly normal. The patient had no complaints of headache or pain in the post-anesthesia care unit and she was advised to follow-up with the neurosurgeon later.
DISCUSSION
The case described above reflects an example of unfamiliarity of the consultant neurologist with regards to the management of parturients with ACM-1, leading to an underestimation of the patient's ACM-1 severity, and assuring the patient that vaginal delivery with an epidural anesthetic is safe. Therefore, the first author, based on his dual training, provided a thorough understanding of the physiology of ACM-1 in terms of the neurosurgical and anesthesiology concerns. Therefore, he was able to establish a correct diagnosis by personally interpreting the patient's MRI.
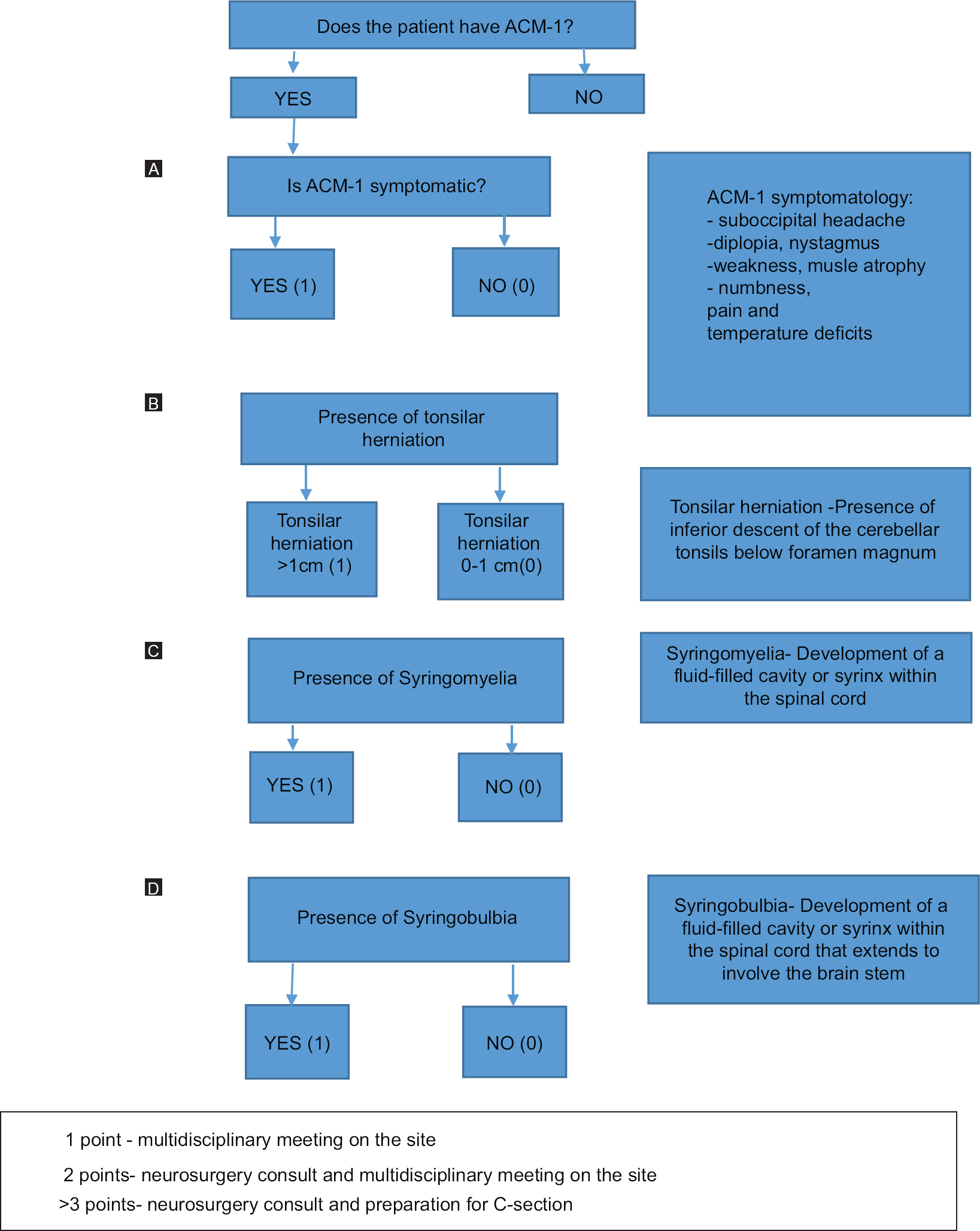
Based on this case report and other similar cases found in the literature where the obstetricians found themselves alone with an ACM-1 patient, the first author developed a questionnaire to utilize when faced with such cases [
In order to prevent the occurrence of a preventable catastrophe, the obstetric specialists need to raise awareness of the ACM-1 condition in pregnant patients to their primary care provider and specialists and vice-versa. In addition, a thorough examination of the most recent MRI with the patient, anesthesia provider, and a neurospecialist should be performed prior to deciding on the best mode of delivery. The first author has developed a set of guidelines [
This case demonstrates how the anesthesia team was instrumental in changing the course of labor and delivery by using a comprehensive physical examination and a thorough review of the brain MR imaging, thus helping prevent possible maternal-newborn morbidity and mortality.
It is paramount to review the MRI report, as well as to independently interpret the images, searching for the position of the tonsils and the presence of a syrinx while correlating with the patient's symptomatology. Neurosurgical consult has to be available and performed before proceeding with the delivery. It is critical that the anesthesiology specialist has proper training regarding the specificities of ACM-1 and can intervene in the diagnosis and peripartum management of this classification of patients.
Pathophysiology of syringomyelia and syringobulbia
Syringomyelia presents in two forms, namely, noncommunicating and communicating.[
The main goal of anesthesia management in parturients with ACM-1 and presence of syringomyelia is to avoid increase in craniospinal pressure. An increase in this gradient or ICP can lead to the herniation of the cerebellum/brainstem or further extension of the syrinx. Avoiding increases in intrathoracic pressure, such as with sneezing, coughing, vomiting, and Valsalva maneuver, is vital, and thus Cesarean section under general anesthesia is recommended for delivery.[
Syringobulbia can occur in a previously established syringomyelia due to upward pulsatile fluid movements.[
Management of parturients with ACM-1 during delivery
General anesthesia in ACM-1 parturients
General anesthesia avoids the risk of dural puncture in patients with increased ICP. It also can help manage the sudden increases in intracranial pressure intraoperatively by hyperventilating the patient, keeping the airway secure, and controlling blood pressure.[
Chantigian et al. conducted a retrospective study on parturients from their institution diagnosed with ACM-1 and the mode of delivery.[
Despite such promising results, laryngeal manipulation and endotrachial intubation can lead to abrupt increases in ICP with serious consequences; for this reason, some authors prefer awake intubation with local airway anesthesia to prevent serious complications from occuring,[
Parturients that present with ACM-1 and syringomyelia have an increased sensitivity to neuromuscular blocking agents; some anesthesiologists prefer to avoid them altogether, whereas others closely monitor neuromuscular function for prolonged effects and then provide complete reversal.[
Spinal anesthesia in ACM-1 parturients
Two cases reported in the literature demonstrated mild-to-more severe complications following spinal anesthesia performed in two parturients with ACM-1.[
Epidural anesthesia for parturients with ACM-1
Epidural anesthesia is preferred over general anesthesia in some instances in parturients with ACM-1 during delivery. It is worth mentioning that, in the nonpregnant population with an elevated baseline ICP, a transient increase of ICP occurs after epidural injection of 10 mL bolus of local anesthetic given over 20–30 s that lasts for 4.5 min (average increase of 21 mmHg) (from 18.8 to 39.5 mmHg). In comparison, in the nonpregnant population with pre-injection normal ICP, there was an average 6 mmHg (9.3–15.6 mmHg) increase in ICP that lasted for 2.3 min. Reduction in the volume of bolus to 5 mL in patients with an elevated ICP significantly reduced the increase in ICP to 5 mmHg for a period of 2.8 min.[
Semple et al. reported an uncomplicated cesarean delivery conducted under epidural anesthesia with incremental local anesthetic boluses followed by continuous infusion in a patient with ACM-1.[
One parturient developed severe complications following unintentional dural puncture during epidural anesthetic placement and demonstrated symptoms of episodic headaches and progression to gait instability 1 year later.[
Although successful deliveries were acomplished with this method of anesthesia, the potential risks of inadvertant dural puncture during needle or catheter insertion can lead to serious complications. In order to prevent them it is recommended to use small bolus doses of local anesthetic medications, which would have less effect on increasing ICP in susceptible patients.
Other ACM-1 particularities
In patients with known ACM-1, comprehensive neurologic evaluation should be conducted before deciding about the mode of delivery, and again after delivery to look for any signs of worsened neurological symptoms or evolution of new symptoms. Neurological assessment needs to include identification of autonomic neuropathy, especially cardiac autonomic neuropathy as it can present with tachiarrhythmias and wide fluctuations in arterial blood pressure intraoperatively. Furthermore, sudden cardiac death and respiratory arrest were reported in patients with syringomyelia and concomitant diabetic neuropathy following general and regional anesthesia.[
CONCLUSION
Unfamiliarity of health care providers with regard to ACM-I parturients can be countered with increasing awareness to this condition throughout the medical specialties involved in their care. The Ghaly Obstetric Guide to Arnold-Chiari malformation type 1 (GOGAC-1) can assist in making the right decision when faced with such cases. The GOGAC-1 guideline is an excellent tool in aiding with the process of taking the next best step in the management and diagnosis for providers, especially for those who come in contact with such patients for their first time.
Depending on the degree of ACM-1 severity, the Ghaly Obstetric Guide to ACM-1 dictates that a multidisciplinary approach determines the mode of delivery. Correct interpretation of the imaging tests with thorough neurological, cardiac, and respiratory examination should be conducted before making a decision on anesthetic management. The table developed by the senior author regarding the recommendations to be followed for ACM-1 parturients perioperatively helps prevent and manage potential complications. Furthermore, postoperative pain should be addressed in such patients; thus, a preemptive discussion on the best options available for pain management has to take place between specialists involved in the care of the patient.
These steps along with proper training of the anesthesia providers regarding the specificities of ACM-1 parturients aids in better management and understanding of this complex condition.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Aydin S, Hanimoglu H, Tanriverdi T, Yentur E, Kaynar MY. Chiari type I malformations in adults: A morphometric analysis of the posterior cranial fossa. Surg Neurol. 2005. 64: 237-41
2. Barkovich A, Wippold FJ, Sherman JL, Citrin CM. Significance of cerebellar tonsillar position on MR. AJNR Am J Neuroradiol. 1986. 7: 795-9
3. Barton JJ, Sharpe JA. Oscilopsia and horizontal nystagmus with accelerating slow phases following lumbar puncture in the Arnold-Chiari malformation. Ann Neurol. 1993. 33: 418-21
4. Cahan LD, Benston JR. Consideration in the diagnosis and treatment of syringomyelia and the Chiari malformation. J Neurosurg. 1982. 57: 24-31
5. Caldarelli M, Di Rocco C. Diagnosis of Chiari I malformation and related syringomyelia: Radiological and neurophysiological studies. Childs Nerv Syst. 2004. 20: 332-5
6. Chantigian RC, Koehm MA, Ramin KD, Warner MA. Chiari malformation in parturients. J Clin Anesth. 2002. 14: 201-5
7. Chiapparini L, Saletti V, Solero CL, Bruzzone MG, Valentini LG. Neuroradiological diagnosis of Chiari malformations. Neurol Sci. 2011. 32: S283-6
8. Dagtekin A, Avci E, Kara E, Uzmansel D, Dagtekin O, Koseoglu A. Posterior cranial fossa morphometry in symptomatic adult Chiari I malformation patients: Comparative clinical and anatomical study. Clin Neurol Neurosurg. 2011. 113: 399-403
9. Daskalakis GJ, Katsetos CN, Papageorgiou IJ, Antsaklis AJ, Vogas EK, Grivachevski VI. Syringomyelia and pregnancy-case report. Eur J Obstet Gynecol Reprod Biol. 2001. 97: 98-100
10. Deng X, Yang C, Gan J, Wu L, Yang T, Yang J. Long term outcomes after small-bone-window posterior fossa decompression and duraplasty in adults with Chiari Malformation Type-1. World Neurosurg. 2015. 84: 998-1004
11. Duffy GP. Lumbar punture in the presence of raised intracranial pressure. BMJ. 1969. 1: 407-9
12. Elster AD, Chen M. Chiari I malformations: Clinical and radiologic reappraisal. Radiology. 1992. 183: 347-53
13. Fakhri A, Shah M, Goyal M. Advanced imaging of Chiari I malformations. Neurosurg Clin N Am. 2015. 26: 519-26
14. Gardener WJ, Abdullah AF, McCormack LJ. Varying expressions of embrional atresia of the fourth ventricle in adults. Arnold-Chiari malformation, Dandy-Walker syndrome, “arachnoid” cyst of cerebellum, and syringonyelia. J Neurosurg. 1957. 14: 591-607
15. Ghaly RF, Candido KD, Sauer R, Knezevic NN. Anesthetic management during Cesarean section in a woman with residual Arnold-Chiari malformation Type I, cervical kyphosis, and syringomyelia. Surg Neurol Int. 2012. 3: 26-
16. Haughton V, Mardal KA. Spinal fluid biomechanics and imaging: An update for neuroradiologists. AJNR Am J Neuroradiol. 2014. 35: 1864-9
17. Haughton VM, Korosec FR, Medow JE, Dolar MT, Iskandar BJ. Peak systolic and diastolic CSF velocity in the foramen magnum in adult patients with Chiari I malformations and in normal control participants. Am J Neuroradiol. 2003. 24: 169-76
18. Hilt H, Gramm HJ, Link J. Changes in intracranial pressure associated with extradural anesthesia. Br J Anaesth. 1986. 58: 676-80
19. Hopkins EL, Hendricks CH, Cibils LA. Cerebrospinal fluid pressure in labor. Am J Obstet Gynecol. 1965. 93: 907-16
20. Hullander RM, Bogard TD, Leivers D, Moran D, Dewan DM. Chiari I malformation presenting as recurrent spinal headache. Anesth Analg. 1992. 75: 1025-6
21. Imperato A, Seneca V, Cioffi V, Colella G, Gangemi M. Treatment of Chiari malformation: Who, when and how. Neurol Sci. 2011. 32: S335-9
22. Isu T, Iwasaki Y, Akino M, Abe H. Hydrosyringomyelia associated with Chiari I malformation in children and adolescents. Neurosurgery. 1990. 26: 591-6
23. Kahn JK, Sisson JC, Vinik AI. QT interval prolongation and sudden cardiac death in diabetic autonomic neuropathy. J Clin Endocrinol Metab. 1987. 64: 751-4
24. Lin VW, Lin VW, Cardenas DD.editors. Syringomyelia. Spinal cord medicine: Principles and practice. New York: Demos Medical Publishing; 2003. p. 501-7
25. Marx GF, Oka Y, Orkin LR. Cerebrospinal fluid pressures during labor. Am J Obstet Gynecol. 1962. 84: 213-9
26. Mauer UM, Gottschalk A, Mueller C, Weselek L, Kunz U, Schulz C. Standard and cardiac-gated phase-contrast magnetic resonance imaging in the clinical course of patients with Chiari malformation Type I. Neurosurg Focus. 2011. 31: E5-
27. McCausland AM, Holmes F. Spinal fluid pressures during labor; preliminary report. West J Surg Obstet Gynecol. 1957. 65: 220-31
28. McGirt MJ, Atiba A, Attenello FJ, Wasserman BA, Datoo G, Gathinji M. Correlation of hindbrain CSF flow and outcome after surgical decompression for Chiari I malformation. Childs Nerv Syst. 2008. 24: 833-40
29. McGirt MJ, Nimjee SM, Fuchs HE, George TM. Relationship of cine phase-contrast MRI to outcome after decompression for Chiari I malformation. Neurosurgery. 2006. 59: 140-6
30. McIlroy WJ, Richardson JC. Syringomyelia: A clinical review of 75 cases. CMAJ. 1965. 93: 731-4
31. Meadows J, Kraut M, Guarnieri M, Haroun RI, Carson BS. Asymptomatic Chiari Type I malformations identified on magnetic resonance imaging. J Neurosurg. 2000. 92: 920-6
32. Milhorat TH, Chou MW, Trinidad EM, Kula RW, Mandell M, Wolpert C. Chiari malformation redefiened: Clinical and radiographic findings for 364 symptomatic patients. Neurosurgery. 1999. 44: 1005-17
33. Milhorat TH. Classification of syringomyelia. Neurosurg Focus. 2000. 8: E1-
34. Morgan D, Williams B. Syringobulbia: A surgical appraisal. J Neurol Neurosurg Psych. 1992. 55: 1132-41
35. Mustapha B, Chkoura K, Elhassan M, Ahtil R, Azendour H, Kamili ND. Difficult intubation in a parturient with syringomyelia and Arnold-Chiari malformation: Use of Airtraq™ laryngoscope. Saudi J Anaesth. 2011. 5: 419-22
36. Nielsen JL, Bejjani GK, Vallejo MC. Cesarean delivery in a parturient with syringomyelia and worsening neurological symptoms. J Clin Anesth. 2011. 23: 653-6
37. Nishikawa M, Sakamoto H, Hakuba A, Nakanishi N, Inoue Y. Pathogenesis of Chiari malformation: A morphometric study of the posterior cranial fossa. J Neurosurg. 1997. 86: 40-7
38. Nogues MA, Newman PK, Male VJ, Foster JB. Cardiovascular reflexes in syringomyelia. Brain. 1982. 105: 835-49
39. Penney DJ, Smallman JMB. Arnold-Chiari malformation and pregnancy. Int J Obstet Anesth. 2001. 10: 139-41
40. Radmanesh A, Greenberg JK, Chatterjee A, Smyth MD, Limbrick DD, Sharma A. Tonsillar pulsatility before and after surgical decompression for children with Chiari malformation type 1: An application for true fast imaging with steady state precession. Neuroradiology. 2015. 57: 387-93
41. Ruff ME, Oakes WJ, Fisher SR, Spock A. Sleep apnea and vocal cord paralysis secondary to type I Chairi malformation. Pediatrics. 1987. 80: 231-4
42. Semple DA, McClure JH. Arnold-Chiari malformation on pregnancy. Anesthesia. 1996. 51: 580-2
43. Shamji MF, Enrique CG, Ventureyra EC, Nzau M, Vassilyadi M. Classification of symptomatic Chiari I malformation to guide surgical strategy. Can J Neurol Sci. 2010. 37: 482-7
44. Sicuranza GB, Steinberg P, Figuoroa R. Arnold-Chiari Malformationb in a pregnant woman. Obstet Gynecol. 2003. 102: 1191-4
45. Speer MC, Enterline DS, Mehtretter L, Hammock P, Joseph J, Dickerson M. Chiari Type I malformation with or without syringomyelia: Prevalence and genetics. J Genet Couns. 2003. 12: 297-311
46. Tubbs RS, Bailey M, Barrow WC, Loukas M, Shoja MM, Oakes WJ. Morphometric analysis of the craniocervical juncture in children with Chiari I malformation and concomitant syringobulbia. Childs Nerv Syst. 2009. 25: 689-92
47. Walton Sir J.editorsBrain's diseases of the nervous system. Oxford: Oxford University Press; 1985. p. 412-6
48. Williams B. On the pathogenesis of syringomyelia: A review. J R Soc Med. 1980. 73: 798-806
49. Williams B, Brock M, Dietz H.editors. The valvular action of the Arnold-Chiari malformation. 1: Intracranial Pressure, experimental and clinical aspects. Heidelberg: Springer-Verlag; 1972. p. 86-90