- Department of Clinical Neurological Sciences (Neurosurgery), Western University, London, Ontario,
- Department of Surgery, Kuwait University,
- Department of Neurosurgery, Ibn Sina Hospital, Kuwait City, Kuwait,
- Department of Pathology, Al-Sabah Hospital, Kuwait, Kuwait City, Al-Asmah,
- Department of Biochemistry and Medical Genetics, University of Manitoba, Winnipeg, Manitoba.
Correspondence Address:
Mohammad Mohammad
Department of Biochemistry and Medical Genetics, University of Manitoba, Winnipeg, Manitoba.
DOI:10.25259/SNI_589_2019
Copyright: © 2020 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Mohammad Mohammad, Hisham Al-Khayat, Kenneth Katchy, Shervin Pejhan. Neurenteric cyst secondary to lumboperitoneal shunt. 24-Jan-2020;11:14
How to cite this URL: Mohammad Mohammad, Hisham Al-Khayat, Kenneth Katchy, Shervin Pejhan. Neurenteric cyst secondary to lumboperitoneal shunt. 24-Jan-2020;11:14. Available from: https://surgicalneurologyint.com/surgicalint-articles/9855/
Abstract
Background: Neurenteric cysts are rare lesions that typically present in the upper thoracic and cervical spine and are occasionally found intracranially. The optimal treatment is gross total excision as subtotal/partial excisions are associated with high recurrence rates.
Case Description: For the past 10 years, a patient with pseudotumor cerebri required repeated lumboperitoneal (LP) shunt revisions. This resulted in multiple neuroenterogenous cysts occurring around the proximal LP subarachnoid shunt catheter, a finding likely attributable to retrograde flow from the peritoneal cavity.
Conclusion: Unlike ventriculoperitoneal (VP) shunts and LP shunts do not contain valves, making the retrograde passage of enterogenous cells possible when abdominal pressure exceeds lumbar subarachnoid pressure, especially in the morbidly obese patient.
Keywords: Enterogenous cell, Neurenteric cyst, Pseudotumor cerebri, Morbid obesity, Idiopathic intracranial hypertension
INTRODUCTION
Neurenteric cysts, also known as enterogenous cysts, account for 0.3–1.3% of all spinal axis tumors.[
Here, we present a 42-year-old female with multiple enterogenous cell deposits in the lower thoracic spine found 22 years after the original insertion of a lumboperitoneal shunt.
CASE REPORT
Twenty-two years ago, a 42-year-old female presented with worsening vision/bilateral papilledema attributed to pseudotumor cerebri (idiopathic intracranial hypertension); a lumboperitoneal (LP) shunt was placed and required revision 10 years later. Now, 22 years after the original surgery, this morbidly obese patient (body mass index of 52) newly presented with the 2-month onset of severe thoracic pain, bilateral leg weakness (grade 1/5), and a T1 sensory level.
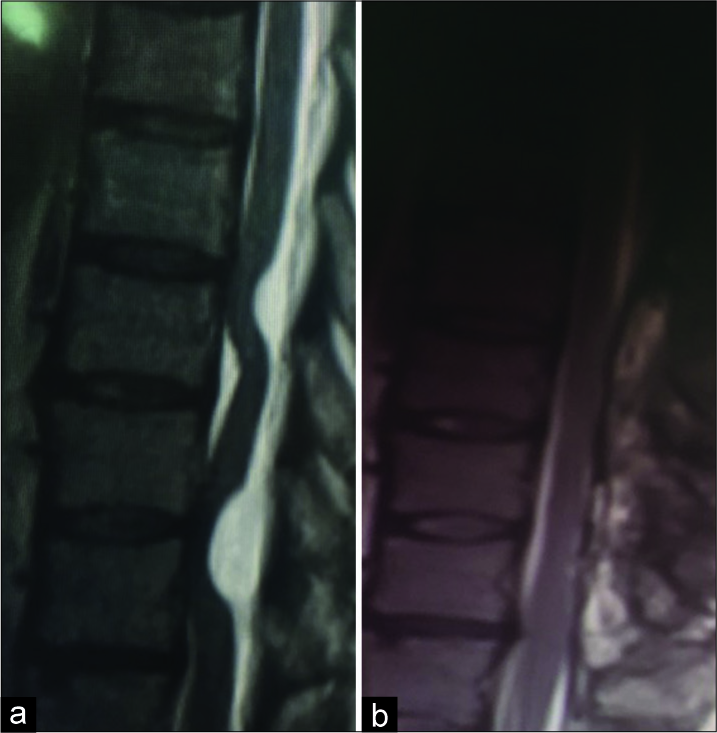
MR studies
The thoracic MR demonstrated a hypointense lesion on the T1-weighted images that did not enhance with gadolinium. Notably, two well-defined, dorsal hyperintense lesions opposite the T9 and T11 levels flattened the spinal cord as best seen on the T2-weighted images [
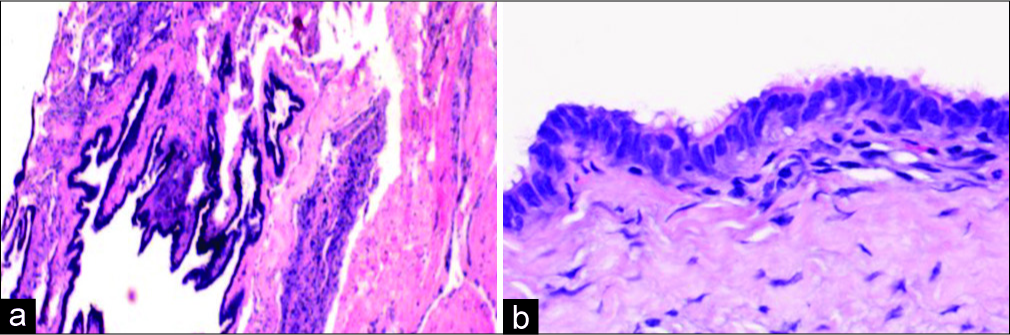
Pathology
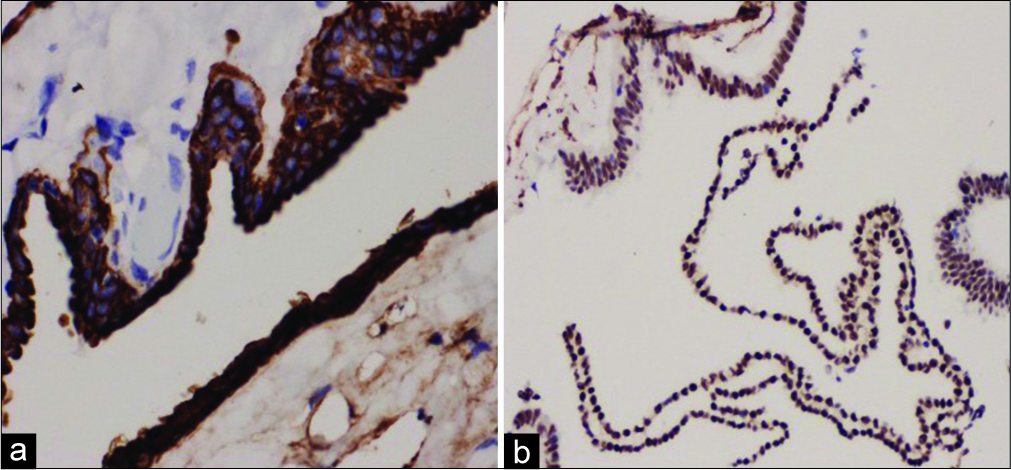
The gross pathological examination demonstrated multiple pieces of grey-white soft-tissue fragments measuring 2 × 1 × 0.2 cm. Microscopically, the specimen revealed a cyst lined by ciliated columnar epithelium [
Subsequent gastric bypass surgery
The patient later underwent a bariatric gastric bypass. She was asymptomatic for the next year. Three years later, she has maintained her neurological baseline, for example, a high level of independent function.
DISCUSSION
Unlike VP shunts and LP shunts do not contain valves, making retroperitoneal passage of enteric cells possible when intra-abdominal pressure exceeds lumbar subarachnoid pressure. These enlarged enteric cysts, depending on their spinal location (e.g., cervical/thoracic/ lumbar), are capable of causing significant compression leading to paraparesis. Lambert et al. previously established a relationship between morbid obesity and increased intra-abdominal pressure contributing to retrograde flow of enteric cells from the peritoneal cavity resulting in enterogenous mucin secreting cells located around a proximal LP shunt catheter.[
In Gallmann et al., multiple intraventricular fat deposits documented on MRI/computed tomography in a 25-year- old female were also attributed to retrograde flow from the abdominal fat occurring during multiple shunt revisions.[
In the patient presented with morbid obesity and pseudotumor cerebri, the patient developed an enterogenous cyst at the proximal end of an LP shunt catheter at the T9 and T11 levels 22 years after having originally undergone LP shunt placement. Due to the marked cord compression, these lesions were appropriately excised, and the patient’s symptoms resolved.
Here, the enterogenous cysts were attributed to retrograde flow occurring when intra-abdominal pressure exceeded subarachnoid pressure in this morbidly obese patient. Future reduction in the intra-abdominal pressure with stringent weight loss, including, in this case, bariatric surgery should reduce the future risk of retrograde flow of enteric cells from the abdominal cavity to the subarachnoid space.
CONCLUSION
Retrograde flow of enteric cells from the abdominal cavity to subarachnoid space can occur and can be prevented with weight reduction which include in this case bariatric surgery.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Acknowledgments
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for- profit sectors.
References
1. Chen CT, Lee CY, Lee ST, Chang CN, Wei KC, Wu CT. Neurenteric cysts: Risk factors and management of recurrence. Acta Neurochir (Wien). 2016. 158: 1325-31
2. Desai S, Friedman JA, Hlavin J, Briner RP, Toussaint LG, White JB. Disseminated enterogenous cells at the cervicomedullary junction causing communicating hydrocephalus. J Neurosurg. 2010. 113: 1284-6
3. Gallmann W, Gonzalez-Toledo E, Riel-Romero R. Intraventricular fat from retrograde flow through a lumboperitoneal shunt. J Neuroimaging. 2011. 21: 287-9
4. Lambert DM, Marceau S, Forse RA. Intra-abdominal pressure in the morbidly obese. Obes Surg. 2005. 15: 1225-32