- Department of Neurosurgery, University of Utah, Salt Lake City, Utah, United States.
- School of Medicine, University of Utah, Salt Lake City, Utah, United States.
- Department of Radiation Oncology, University of Utah, Salt Lake City, Utah, United States.
Correspondence Address:
John D. Rolston, Department of Neurosugery, Clinical Neurosciences Center, University of Utah, Salt Lake City, Utah, United States.
DOI:10.25259/SNI_91_2022
Copyright: © 2022 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Bornali Kundu1, Andrea A. Brock1, Jason G. Garry2, Randy L. Jensen1,3, Lindsay M. Burt3, Donald M. Cannon3, Dennis C. Shrieve3, John D. Rolston1. Outcomes using linear accelerator stereotactic radiosurgery for the treatment of trigeminal neuralgia: A single-center, retrospective study. 10-Jun-2022;13:246
How to cite this URL: Bornali Kundu1, Andrea A. Brock1, Jason G. Garry2, Randy L. Jensen1,3, Lindsay M. Burt3, Donald M. Cannon3, Dennis C. Shrieve3, John D. Rolston1. Outcomes using linear accelerator stereotactic radiosurgery for the treatment of trigeminal neuralgia: A single-center, retrospective study. 10-Jun-2022;13:246. Available from: https://surgicalneurologyint.com/?post_type=surgicalint_articles&p=11645
Abstract
Background: Linear accelerator (LINAC)-based stereotactic radiosurgery (SRS) treatment of trigeminal neuralgia (TN) may have similar efficacy to Gamma Knife SRS (GK-SRS), but the preponderance of data comes from patients treated with GK-SRS. Our objective was to analyze the outcomes for LINAC-based treatment of TN in patients at our institution.
Methods: We retrospectively analyzed data for patients who underwent LINAC-based SRS for TN from 2006 to 2018. Data were collected from the patients’ medical records. Nonparametric statistics were used for the analysis.
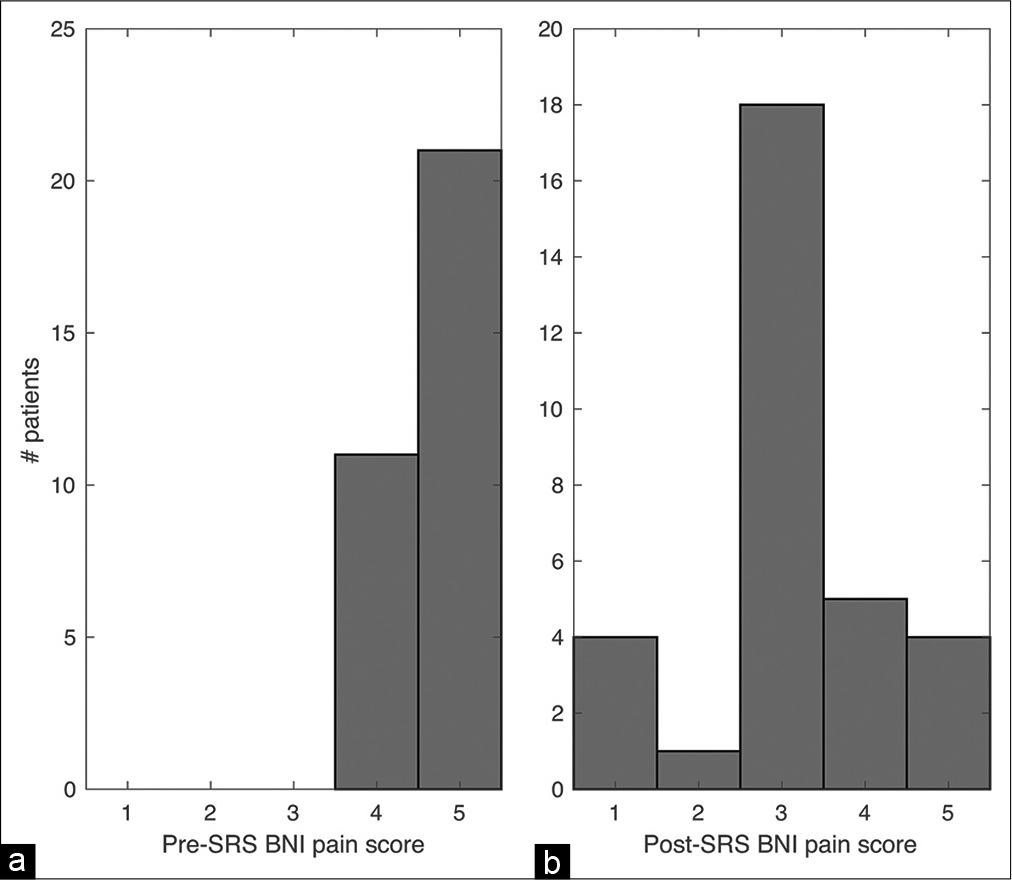
Results: Of the 41 patients treated with LINAC-based SRS (typically 90 Gy dosed using a 4 mm collimator for one fraction) during that time, follow-up data of >3 weeks post-SRS were available for 32 patients. The median pretreatment Barrow Neurological Institute (BNI) pain score was 5 (range 4–5). The follow-up period ranged from 0.9 to 113.2 months (median 5 months). There was significant improvement in postradiation BNI pain score (P 3). Survival analysis showed a median time to pain recurrence of 30 months. There was no relationship between prior microvascular decompression (MVD) surgery and change in BNI pain score pre- to posttreatment.
Conclusion: The results demonstrate that LINAC-based SRS is an effective means to treat TN. Prior MVD surgery did not affect efficacy of SRS in lowering the BNI score from pre- to posttreatment in this patient cohort.
Keywords: BNI pain score, Linear accelerator, Outcomes, Pain, Stereotactic radiosurgery, Trigeminal neuralgia
INTRODUCTION
Trigeminal neuralgia (TN) is classically characterized by episodic facial pain (Type 1). Other subtypes have also been recognized that involve continuous facial pain (Type 2) or pain related to demyelinating disorders.[
The two most common methods of SRS are Gamma Knife (GK) and linear accelerator (LINAC). A retrospective systematic review of the techniques suggested comparable efficacies for the treatment of TN,[
In this study, we retrospectively analyzed outcomes for LINAC-based treatment of TN in a group of patients at our institution. Importantly, we consistently used a radiation dose of 80–90 Gy delivered to the root entry zone (REZ) to treat our patients, which has been reported as an efficacious dose and target for the treatment of TN.[
MATERIALS AND METHODS
Patients
We retrospectively analyzed data for consecutive patients treated at our institution who underwent LINAC-based radiation treatment for TN from 2006 to 2018. For this Institutional Review Board (IRB)-approved case series, data were collected from the electronic medical record. Explicit patient consent was waived for this retrospective data review by the IRB. All data were deidentified for analysis. Data regarding age, sex, time of pain onset, and time of SRS procedure relative to other interventions (including MVD, percutaneous balloon rhizotomy, radiofrequency ablation, and glycerol rhizotomy) were recorded. A medical history of multiple sclerosis was also noted. Time to pain relief was determined based on clinical follow-up visit notes. Brainstem dosimetry data were also collected when available for patients.
TN type was delineated using the Burchiel classification system.[
Treatment protocol
All patients underwent frame-based stereotaxis using a stereotactic Leksell head frame (Elekta Instruments AB Stockholm, Sweden). Patients then underwent a CT scan acquired with a 1–1.25 mm slice thickness for dose calculation and treatment planning purposes. Constructive interference in steady-state or 3D-fast imaging employing steady-state acquisition magnetic resonance imaging had been performed previously, and these scans were imported into the iPlan treatment planning system (Brainlab AG, Munich, Germany). These were used in combination with stereotactic T1-weighted images to identify the trigeminal nerve target and place the isocenter. After fusion with the CT scan, a treatment plan with seven arcs using a 4 mm collimator was devised. The target for all patients was the REZ of the TN in the prepontine cistern such that the brainstem contour was at the ~50% isodose line (IDL).[
Pain scale
The Barrow Neurological Institute (BNI) scoring system was used to quantify pain before and after the procedure.[
Statistical analysis
Data analysis was done using Matlab (version 2019b, Natick, MA). The nonparametric Wilcoxon rank-sum and signed-rank tests were used for the analysis. The Kaplan–Meier estimator was used to estimate the probability of pain control (defined as a BNI <4) from time of SRS treatment in a cohort of patients who had initial pain control.[
This case series has been reported in line with the PROCESS guidelines.[
RESULTS
Demographics
Forty-one patients with a mean age of 67.4 (range 30–89) years were identified for inclusion. Of these, 25 (61%) had Type 1 TN, 3 (7%) had Burchiel Type 2 TN, 1 (2%) had TN related to trauma (neuropathic TN), 8 (20%) had multiple sclerosis-related TN (symptomatic TN), and 4 (10%) had atypical TN [
Outcomes after SRS
Of the 41 patients, 32 patients had sufficient follow-up data. The follow-up period varied from 0.9 to 113.2 months (median 5 months). Of these 32 patients, 23 (72%) reached a BNI pain score of 1–3. There was significant improvement in postradiation BNI score (z = 4.38, P < 0.001, Wilcoxon signed-rank test, [
Brainstem dosimetry was retrieved for 33 patients. The median maximal dose delivered was 4500 cGy (range 1530–7020 cGy). Volumetric analysis showed that median volume of tissue that received 4500 cGy was 0.001 ml, the median volume that received 2700 cGy was 0.009 ml, and the median volume that received 1800 cGy radiation was 0.029 ml. The maximum dose to the brainstem did not relate to pain persistence (i.e., BNI score >3; Kruskal–Wallis test, Chi-squared (1) = 1.17, P = 0.28). Because only one patient had a documented adverse effect, dosimetric analysis on toxicity could not be performed; however, the maximum dose delivered to that patient was 4050 cGy.
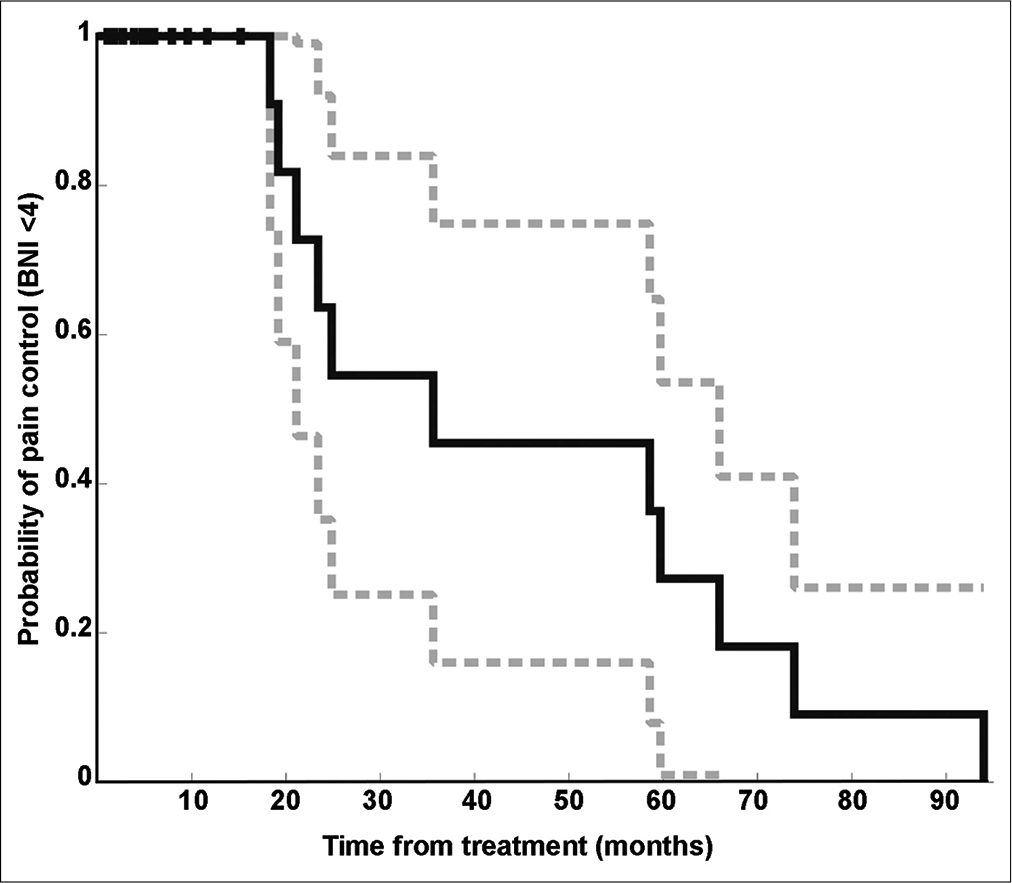
A total of 12 of 32 (38%) patients had recurrence of symptoms after SRS, and 11 underwent an additional intervention such as additional SRS, rhizotomy, or peripheral electrical stimulation. Kaplan–Meier analysis of the probability of pain control from time of SRS treatment was done using a subset of patients who had initial benefit from SRS (BNI score <4). The median time to recurrence was 30.2 months (range 18.3– 94 months; [
Figure 2:
Kaplan–Meier survival curve showing probability of pain control (BNI < 4) decreases with time. This analysis includes only patients who had initial pain control with LINAC-based SRS treatment. Censored data are those patients who did not reach the endpoint of interest, which was documented pain recurrence at last follow-up (denoted with a “|”). The curve shows decreasing probability of continued pain control from time of intervention. A 95% confidence interval shown in dashed line.
DISCUSSION
TN affects 4.3 in 100,000 people/year,[
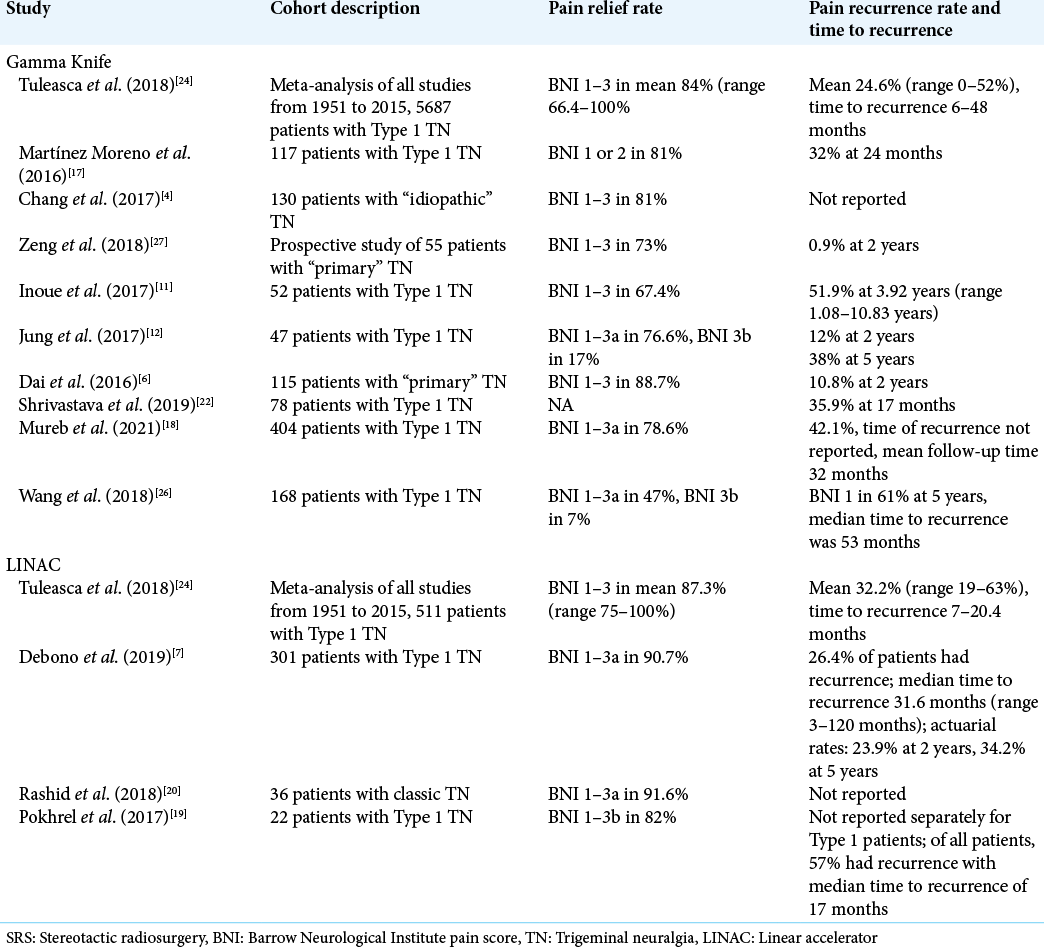
As for other types of TN, in Pokhrel et al.,[
Tuleasca et al.[
Interestingly, prior MVD surgery and prior surgical treatment history did not affect efficacy of SRS in lowering the BNI score from pre- to posttreatment in our cohort of patients. Tuleasca et al.[
Limitations
This study has a range of limitations including being retrospective and having a relatively small sample size, although only several other studies have larger cohorts of patients treated with LINAC-based SRS for TN.[
CONCLUSION
LINAC-based SRS is an effective means to treat TN for those with Type 1 as well as atypical TN and TN in the setting of multiple sclerosis. Prior MVD surgery did not affect efficacy of SRS in lowering the BNI score from pre- to posttreatment in this cohort of patients. A larger cohort of patients with prior MVD treatment followed by SRS treatment would help explore this result in future studies.
Declaration of patient consent
Institutional Review Board (IRB) permission obtained for the study.
Financial support and sponsorship
Neurosurgery Research and Education Fund (NREF).
Conflicts of interest
JDR has received consulting fees from Medtronic and Corlieve Therapeutics. RLJ has received consulting fees from Medtronic.
References
1. Agha RA, Sohrabi C, Mathew G, Franchi T, Kerwan A, O’Neill N. The PROCESS 2020 guideline: updating consensus preferred reporting of CasE series in surgery (PROCESS) guidelines. Int J Surg. 2020. 84: 231-5
2. Burchiel KJ. A new classification for facial pain. Neurosurgery. 2003. 53: 1164-6
3. Cardillo G.editors. KMPLOT: Kaplan-Meier Estimation of the Survival Function. San Francisco, California: GitHub; 2018. p.
4. Chang CS, Huang CW, Chou HH, Lin LY, Huang CF. Outcome of Gamma Knife radiosurgery for trigeminal neuralgia associated with neurovascular compression. J Clin Neurosci. 2018. 47: 174-7
5. Chen JC, Girvigian M, Greathouse H, Miller M, Rahimian J. Treatment of trigeminal neuralgia with linear accelerator radiosurgery: Initial results. J Neurosurg. 2004. 101: 346-50
6. Dai ZF, Huang QL, Liu HP, Zhang W. Efficacy of stereotactic gamma knife surgery and microvascular decompression in the treatment of primary trigeminal neuralgia: A retrospective study of 220 cases from a single center. J Pain Res. 2016. 9: 535-42
7. Debono B, Lotterie JA, Sol JC, Bousquet P, Duthil P, Monfraix S. Dedicated linear accelerator radiosurgery for classic trigeminal neuralgia: A single-center experience with long-term follow-up. World Neurosurg. 2019. 121: e775-85
8. Eller J, Raslan A, Burchiel K. Trigeminal neuralgia: Definition and classification. Neurosurg Focus. 2005. 18: E3
9. Friedman W. Linear accelerator radiosurgery. Clin Neurosurg. 1992. 38: 445-71
10. Goss BW, Frighetto L, DeSalles AA, Smith Z, Solberg T, Selch M. Linear accelerator radiosurgery using 90 gray for essential trigeminal neuralgia: Results and dose volume histogram analysis. Neurosurgery. 2003. 53: 823-30
11. Inoue T, Hirai H, Shima A, Suzuki F, Yamaji M, Fukushima T. Long-term outcomes of microvascular decompression and Gamma Knife surgery for trigeminal neuralgia: A retrospective comparison study. Acta Neurochir (Wien). 2017. 159: 2127-35
12. Jung HH, Park CK, Jung NY, Kim M, Chang WS, Chang JW. Gamma knife radiosurgery for idiopathic trigeminal neuralgia: Does the status of offending vessels influence pain control or side effects?. World Neurosurg. 2017. 104: 687-93
13. Katusic S, Beard CM, Bergstralh E, Kurland LT. Incidence and clinical features of trigeminal neuralgia, Rochester, Minnesota, 1945-1984. Ann Neurol. 1990. 27: 89-95
14. Königsmaier H, De Pauli-Ferch B, Hackl A, Pendl G. The costs of radiosurgical treatment: Comparison between gamma knife and linear accelerator. Acta Neurochir (Wien). 1998. 140: 1101-11
15. Kundu B, Rolston JD. Nationwide shift from percutaneous rhizotomy to microvascular decompression for treatment of trigeminal and other cranial nerve neuralgias. Headache. 2018. 58: 1675-9
16. Lee AT, Raygor KP, Elefant F, Ward MM, Wang DD, Barbaro NM. Comparison of stereotactic radiosurgery and radiofrequency ablation for trigeminal neuralgia in multiple sclerosis patients. Stereotact Funct Neurosurg. 2020. 98: 378-85
17. Moreno NE, Gutiérrez-Sárraga J, Rey-Portolés G, JiménezHuete A, Álvarez RM. Long-term outcomes in the treatment of classical trigeminal neuralgia by gamma knife radiosurgery: A retrospective study in patients with minimum 2-year follow-up. Neurosurgery. 2016. 79: 879-87
18. Mureb M, Golub D, Benjamin C, Gurewitz J, Strickland BA, Zada G. Earlier radiosurgery leads to better pain relief and less medication usage for trigeminal neuralgia patients: An international multicenter study. J Neurosurg. 2021. 135: 237-44
19. Pokhrel D, Sood S, Mcclinton C, Saleh H, Badkul R, Jiang H. Linac-based stereotactic radiosurgery (SRS) in the treatment of refractory trigeminal neuralgia: Detailed description of SRS procedure and reported clinical outcomes. J Appl Clin Med Phys. 2017. 18: 136-43
20. Rashid A, Pintea B, Kinfe TM, Surber G, Hamm K, Boström JP. LINAC stereotactic radiosurgery for trigeminal neuralgia-retrospective two-institutional examination of treatment outcomes. Radiat Oncol. 2018. 13: 1-11
21. Rogers CL, Shetter AG, Fiedler JA, Smith KA, Han P, Speiser B. Gamma knife radiosurgery for trigeminal neuralgia: Experience at the barrow neurological institute. Int J Radiat Oncol Biol Phys. 2000. 47: 1013-9
22. Shrivastava A, Mohammed N, Hung YC, Xu Z, Schlesinger D, Heinrichs T. Impact of integral dose on the maintenance of pain relief in patients with idiopathic trigeminal neuralgia treated with upfront gamma knife radiosurgery. World Neurosurg. 2019. 129: e375-80
23. Smith ZA, Gorgulho AA, Bezrukiy N, McArthur D, Agazaryan N, Selch MT. Dedicated linear accelerator radiosurgery for trigeminal neuralgia: A single-center experience in 179 patients with varied dose prescriptions and treatment plans. Int J Radiat Oncol Biol Phys. 2011. 81: 225-31
24. Tuleasca C, Régis J, Sahgal A, De Salles A, Hayashi M, Ma L. Stereotactic radiosurgery for trigeminal neuralgia: A systematic review. J Neurosurg. 2018. 130: 733-57
25. Wang DD, Ouyang D, Englot DJ, Rolston JD, Molinaro AM, Ward M. Trends in surgical treatment for trigeminal neuralgia in the United States of America from 1988 to 2008. J Clin Neurosci. 2013. 20: 1538-45
26. Wang DD, Raygor KP, Cage TA, Ward MM, Westcott S, Barbaro NM. Prospective comparison of long-term pain relief rates after first-time microvascular decompression and stereotactic radiosurgery for trigeminal neuralgia. J Neurosurg. 2018. 128: 68-77
27. Zeng YJ, Zhang H, Yu S, Zhang W, Sun XC. Efficacy and safety of microvascular decompression and gamma knife surgery treatments for patients with primary trigeminal neuralgia: A prospective study. World Neurosurg. 2018. 116: e113-7