- Department of Anesthesiology, Reanimatology and Intensive Care, Division of Neuroanesthesia, Zagreb, Croatia
- Department of Neurosurgery, University Hospital Center Zagreb, Zagreb, Croatia
Correspondence Address:
Vasilije Stambolija
Department of Anesthesiology, Reanimatology and Intensive Care, Division of Neuroanesthesia, Zagreb, Croatia
DOI:10.4103/sni.sni_363_17
Copyright: © 2018 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Vasilije Stambolija, Martina Miklić Bublić, Marin Lozić, Jakob Nemir, Miroslav Ščap. PbtO2 monitoring in normobaric hyperoxia targeted therapy in acute subarachnoidal hemorrhage. 23-Feb-2018;9:46
How to cite this URL: Vasilije Stambolija, Martina Miklić Bublić, Marin Lozić, Jakob Nemir, Miroslav Ščap. PbtO2 monitoring in normobaric hyperoxia targeted therapy in acute subarachnoidal hemorrhage. 23-Feb-2018;9:46. Available from: http://surgicalneurologyint.com/surgicalint-articles/primary-intracerebral-ini1%e2%80%91deficient-rhabdoid-tumor-with-cd34-immunopositivity-in-a-young-adult/
Abstract
Background:Low brain tissue oxygen tension (PbtO2), or brain hypoxia, is an independent predictor of poor outcome. Increasing inspirational fraction of oxygen could have a significant influence on treating lower PbtO2. Combined PbtO2 therapy, compared to the approach that focus only on regulation of cerebral perfusion pressure and intracranial pressure, shows better patient outcomes. Monitoring of PbtO2 could be helpful in individualizing treatment, preventing or limiting secondary brain injury, and maintaining better patient outcome.
Case Description:We present a case of a patient with subarachnoidal hemorrhage to whom PbtO2 monitor was implanted, and normobaric hyperoxia treatment was adjusted according to PbtO2 measurement. The patient progressively recovered and was dismissed with Glasgow Coma Score 4/5/6.
Conclusion:The use of PbtO2 monitoring may be useful for monitoring the local tissue values that are useful for induction of normobaric hyperoxia and optimizing the therapy toward more target-defined values. It is an important part of multimodal neuromonitoring, and is the gold standard for brain oxygenation monitoring that can lead to better patient outcome.
Keywords: Anesthesia, Licox, neuromonitoring, normobaric hyperoxia, PbtO2 monitoring, subarachnoidal hemorrhage
INTRODUCTION
The main goals in neuroanesthesia are maintaining intracranial pressure (ICP), cerebral perfusion pressure (CPP), and aerobic brain metabolism, together with diminishing secondary brain injury.[
Normobaric hyperoxia enhances aerobic metabolism in brain and therefore may be neuroprotective. It is achieved by increasing inspirational fraction of oxygen (FiO2) resulting in supraphysiological arterial oxygen tension. Optimal arterial oxygen tension (PaO2) should be 13.3 kPa (100 mm Hg), although higher values of PaO2 in the first 24 h do not increase predicted 6 months mortality.[
Combined PbtO2 therapy, compared to the approach that focus only on regulation of CPP and ICP, shows better patient outcomes.[
The Licox is triple-lumen catheter that is inserted through intracranial bolt and measures ICP, PbtO2, and brain tissue temperature. It may detect poor PbtO2 before ICP rise. Likewise, it may help in defining optimal CPP values. Therefore, by individualizing the treatment of patients, monitoring of PbtO2 could be helpful in preventing or limiting secondary brain injury and maintaining better patient outcome.[
CASE DESCRIPTION
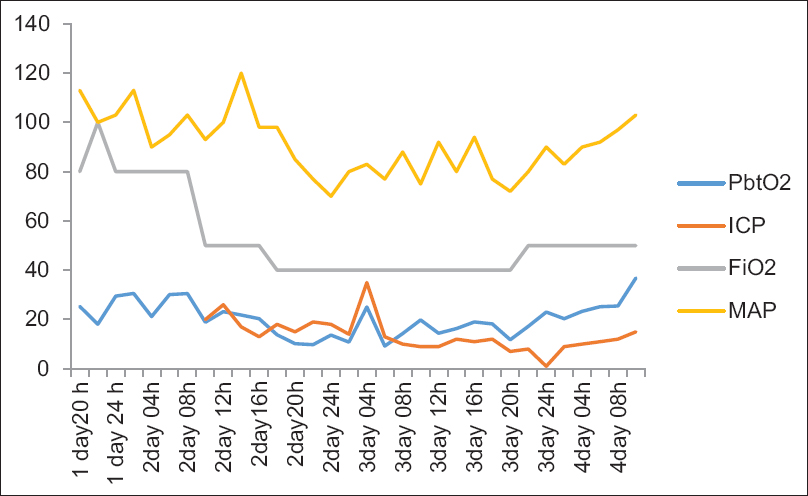
We present a case report of a patient with SAH, to whom Licox monitor was implanted. The patient, aged 35, was admitted to hospital after rupture of right medial cerebral artery aneurysm, scored 3 on the Hunt and Hess Scale. After neurosurgical procedure of aneurysmal clipping, a Licox monitor was implanted in her penumbra region, as well as the CODMAN® MICROSENSOR® ICP monitoring. She was transferred to the neuroanesthesia intensive care unit (ICU), intubated, and mechanically ventilated. During the first 24 h in the ICU, normobaric hyperoxia was applied (FiO2 80%) and the PbtO2 was kept between 20 and 35 mm Hg. On the second day, FiO2 was set down to 40% and a decline in PbtO2 from 20 to 10 mm Hg was noticed, so FiO2 was set to 50% with rise in PbtO2 again. During that time peripheral capillary oxygen saturation (SpO2) was 97–100%.
Invasive arterial pressure was monitored and maintained in adequate range and other means of brain relaxation and possible neuroprotection were already met by meticulous neurointensive treatment (normocapnia, hypertonic saline and high normal blood sodium, analgosedation and relaxation, addition of magnesium). By introducing Licox system of PbO2, we were able to monitor closely PbO2 of the affected brain that was reacting. After aforementioned neurointensive treatment was not giving any further result by not elevating further PbO2, we decided to elevate FiO2, which produced instantaneously positive results visible on the Licox monitor and hence positive clinical outcome on patient morbidity [
Progressive recovery was obvious, and the patient was dismissed with the Glasgow Coma Score (GCS) of 4/5/6. Glasgow Outcome Score after rehabilitation was 5, and Glasgow Outcome Score Extended was 8; no neurologic sequelae were present.
DISCUSSION
Evaluating brain aerobic metabolism and oxygenation based only on PaO2 or SpO2 values may be inadequate and may mislead to the conclusion based on the insufficient data. Also cerebral hypoxia and ischemia can develop in the normal range of CPP and ICP. Additional measurement of PbtO2 in penumbra region is therefore needed, and according to the results, it is possible to adjust the patient therapy toward better cerebral oxygen delivery and utilization ratio.
There is still much controversies about the clinical effect of normobaric hyperoxia in trauma brain injuries (lesser) and/or ischemic brain lesions (greater). Our goal was to circumvent the possible further brain injury and prevent any secondary complications by simply elevating PbO2 in the given delicate moment of the patient treatment, because at that point current standard neurointensive treatment was not met by the criteria given by the Licox system probe measurement of the PbO2.
Normobaric hyperoxia can be beneficial to specific group of patients, and the efficiency of its administration may be monitored with Licox. Therefore, the use of Licox may be useful for monitoring the local tissue values which are useful for induction of normobaric hyperoxia and optimizing the therapy toward more target defined values. PbtO2 monitoring is an important part of multimodal neuromonitoring in the perioperative and ICU settings, and is the gold standard for brain oxygenation monitoring.[
Most studies using tissue oxygen monitors treat initial desaturation episodes with 100% inspired oxygen rather than a transfusion of red blood cells or vasopressor administration to improve CPP.[
Both normobaric hyperoxia and hyperbaric hyperoxia may improve cellular redox state and cerebral metabolic rate for oxygen after TBI in the presence of PbtO2 values that are within or above the normal physiological range.[
In our experience, we have noticed the decrease of PbtO2 values during transport of the patient to the operating room, or to the computed tomography (CT) scan. We have also noticed the decrease of PbtO2 values during mechanical ventilation with PEEP. PEEP is known to obstruct cerebral venous return, which may cause higher ICP,[
For reliable results, proper positioning of PbtO2 monitoring system should always be confirmed with the CT brain scan, which is usually performed within 8 h from the placement of the probe, as well as performing the oxygen test.
CONCLUSION
In our experience with Licox, there was no complications in 5-year period in sense of intracerebral bleeding. In that time we had 20 patients to whom Licox was implanted. Only two of them had lethal outcome, but they were already admitted to the ICU with GCS 1/1/1 and dilated and fixed pupils. The management differs in patients with Licox compared to those who had only ICP monitoring. This case shows how PbtO2 values were low in spite of adequate PaO2 and SpO2 values. With individualizing treatment and increasing FiO2 according to PbtO2 values, subsequently those values have risen. Enhancing cerebral oxygenation may lead to better patient outcome.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form the patient(s) has/have given his/her/their consent for his/her/their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. . American Association of Neurological Surgeons, Congress of Neurological Surgeons: Guidelines for the management of severe traumatic brain injury. J Neurotrauma. 2007. 24: S1-S106
2. Chang JJ, Youn TS, Benson D, Mattick H, Andrade N, Harper CR. Physiologic and functional outcome correlates of brain tissue hypoxia in traumatic brain injury. Crit Care Med. 2009. 37: 283-90
3. Contant CF, Valadka AB, Gopinath SP, Hannay HJ, Robertson CS. Adult respiratory distress syndrome: A complication of induced hypertension after severe traumatic head injury. J Neurosurg. 2001. 95: 560-8
4. Eriksson EA, Barletta JF, Figueroa BE, Bonnell BW, Sloffer CA, Vanderkolk WE. The first 72 hours of brain tissue oxygenation predicts patient survival with traumatic brain injury. J Trauma Acute Care Surg. 2012. 72: 1345-9
5. Frost EA. Effects of positive end-expiratory pressure on intracranial pressure and compliance in brain-injured patients. J Neurosurg. 1977. 47: 195-200
6. Ghosh A, Highton D, Kolyva C, Tachtsidis I, Elwell CE, Smith M. Hyperoxia results in increased aerobic metabolism following acute brain injury. J Cereb Blood Flow Metab. 2017. 37: 2910-20
7. Kirkman MA, Smith M. Brain oxygenation monitoring. Anesthesiology Clin. 2016. 34: 537-56
8. Makarenko S, Griesdale DE, Gooderham P. Multimodal neuromonitoring for traumatic brain injury: A shift towards individualized therapy. J Clin Neurosci. 2016. 26: 8-13
9. Narotam PK, Morrison JF, Nathoo N. Brain tissue oxygen monitoring in traumatic brain injury and major trauma: Outcome analysis of a brain tissue oxygen-directed therapy. J Neurosurg. 2009. 111: 672-82
10. Raj R, Bendel S, Reinikainen M, Kivisaari R, Siironen J, Lang M. Hyperoxemia and long-term outcome after traumatic brain injury. Crit Care. 2013. 17: R177-
11. Rosenthal G, Hemphill JC, Sorani M, Martin C, Morabito D, Meeker M. The role of lung function in brain tissue oxygenation following traumatic brain injury. J Neurosurg. 2008. 108: 59-65