- Clinical Professor of Neurosurgery, Schoold of Medicine, State University of New York at Stony Brook, and % Dr. Marc Agulnick, 1122 Franklin Avenue Suite 106, Garden City, NY 11530, USA.
Correspondence Address:
Nancy E. Epstein, M.D. Clinical Professor of Neurosurgery, School of Medicine, State University of New York at Stony Brook, and % Dr. Marc Agulnick, 1122 Franklin Avenue Suite 106, Garden City, NY 11530, USA
DOI:10.25259/SNI_1003_2021
Copyright: © 2021 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Nancy E. Epstein. Perspective on the true incidence of bowel perforations occurring with extreme lateral lumbar interbody fusions. How should they be treated?. 23-Nov-2021;12:576
How to cite this URL: Nancy E. Epstein. Perspective on the true incidence of bowel perforations occurring with extreme lateral lumbar interbody fusions. How should they be treated?. 23-Nov-2021;12:576. Available from: https://surgicalneurologyint.com/surgicalint-articles/11239/
Abstract
Background: What is the risk of bowel perforation (BP) with open or minimally invasive (MI) extreme lateral lumbar interbody fusion (XLIF)? What is the truth? Further, if peritoneal symptoms/signs arise following XLIF/MI XLIF, it is critical to obtain an emergent consultation with general surgery who can diagnose and treat a potential BP.
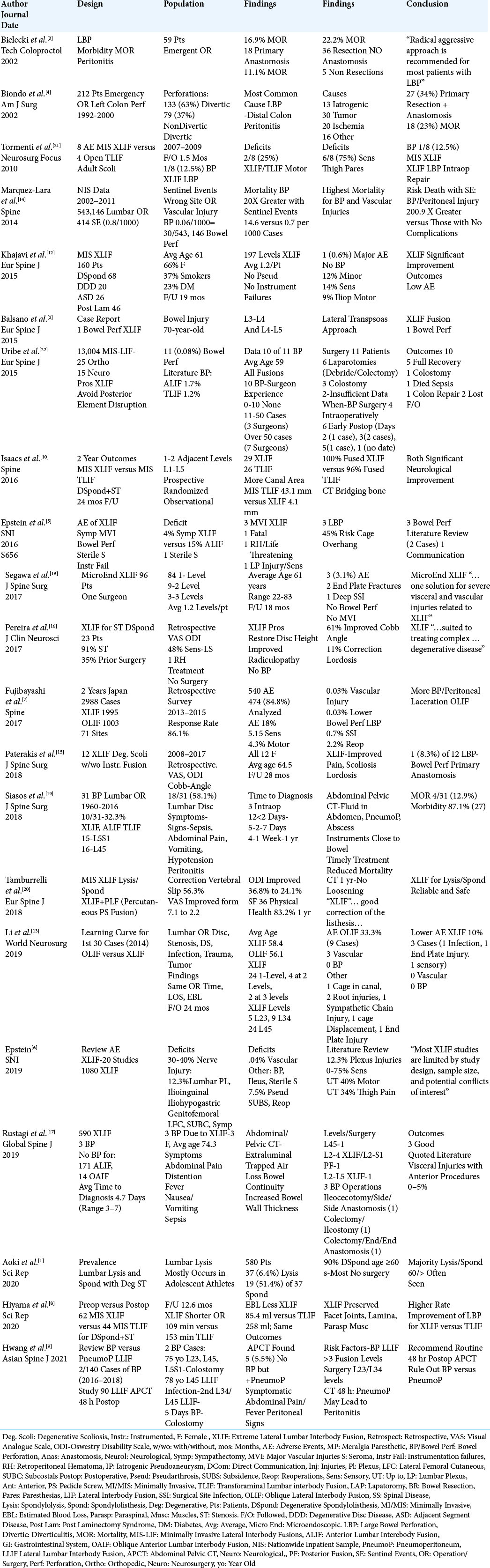
Literature Review: In multiple series, the frequency of BP ranged markedly from 0.03% (i.e. 1 of 2998 patients), to 0.08% (11/13,004), to 0.5%, to 8.3% (1 in 12 patients), up to 12.5% (1 in 8 patients). BPs attributed to different causes carry high mortality rates varying from 11.1% to 23%. For the 11 (0.08%) BP occurring out of 13,004 patients undergoing XLIF in one series, there was one (9.09%) death due to uncontrolled sepsis. In another series, where 31 BP were identified for multiple lumbar surgical procedures identified through PubMed (1960–2016), including 10 (32.2%) for lateral lumbar surgery including XLIF, the overall mortality rate was 12.9% (4/31).
Conclusion: The incidence of BPs occurring following XLIF/MI XLIF procedures ranged from 0.03% to 12.5% in various reports. What is the true incidence of these errors? Certainly, it is more critical that when spine surgeons’ patients develop acute peritoneal symptoms/signs following these procedures, they immediately consult general surgery to both diagnose, and treat potential BP in a timely fashion to avoid the high morbidity (87.1%) and mortality rates (12.9%) attributed to these perforations.
Keywords: Bowel, Extreme lateral lumbar interbody fusion, Perforation, Real frequency, Validation, XLIF, Minimally Invasive (MI) XLIF
INTRODUCTION
What is the true risk of bowel perforation (BP) following open or minimally invasive (MI) extreme lateral lumbar interbody fusion (XLIF) [
FREQUENCY OF SPONDYLOSIS WITH ISTHMIC SPONDYLOLYSIS AND SPONDYLOLISTHESIS
In 2020, Aoki et al. reviewed 580 cases of lumbar spondylosis; 37 (6.4%) patients had spondylolysis, with 19 of 37 additionally demonstrating spondylolisthesis (51.4%) [
XLIF SUPPLEMENTED WITH POSTERIOR LATERAL FUSION (PLF) WITH PEDICLE SCREW INSTRUMENTATION FOR SPONDYLOLYSIS/ SPONDYLOLISTHESIS
In select patients with isthmic spondylolysis/ spondylolisthesis, XLIF/MI XLIF may be combined with posterolateral instrumented pedicle screw fusions to provide simultaneous anterior indirect canal decompression with posterior stabilization/fusion (PLF).[
DIFFERENT FREQUENCIES OF BOWEL PERFORATIONS REPORTS FOR XLIF/MI XLIF
For multiple series, the incidence of BPs attributed to XLIF/MI XLIF ranged between 0.03% to 12.5%; this left us questioning the true incidence of this surgical error [
STUDIES NOT SPECIFICALLY REPORTING THE INCIDENCE OF BOWEL PERFORATIONS
Four clinical series, involving between 23 and 96 patients per study, did not discuss BP as occurring following XLIF/ MI XLIF [
SIGNIFICANT INCIDENCE OF NEUROLOGICAL, VASCULAR, AND OTHER INJURIES WITH XLIF/MI XLIF
Many XLIF/MI XLIF studies cited high frequencies of new sensory (i.e. 5.15–75% including thigh paresthesias), new motor (5.15–40% - typically iliopsoas), and vascular (0.03– 0.04%) injuries [
LEARNING CURVE FOR XLIF VERSUS OLIF
In Li et al. (2019), the learning curve for the first 30 cases of XLIF versus OLIF were studied in patients undergoing lumbar surgery for disc disease, stenosis, degenerative spondylolisthesis, infection, trauma, or tumor.[
FREQUENCY OF BOWEL PERFORATIONS WITH XLIF/MI XLIF
Vastly different frequencies of BPs occurred in patients undergoing XLIF/MI XLIF, ranging from 0.03% to 12.5% [
DIAGNOSTIC FINDINGS FOR BOWEL PERFORATIONS ON ABDOMINAL/PELVIC CT SCANS
Multiple studies also confirmed common findings on Abdominal/Pelvic CT scans (APCT) consistent with/diagnostic of BPs following XLIF/MI XLIF. [
TREATMENT OF BPs OF ALL ETIOLOGIES
High mortality rates are reported for BPs occurring due to many different factors (i.e. especially diveriticular disease, and non-diverticular disease exclusive of spinal surgery) [
HIGH MORTALITY RATE FOR BOWEL PERFORATIONS FOLLOWING XLIF/MI XLIF
High mortality rates (i.e. up to 12.9%) are reported for BPs occurring due to XLIF/MI XLIF.[
CONCLUSION
The incidence of BPs reported following XLIF/MI XLIF procedures ranged from 0.03% up to 12.5% in multiple studies.[
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Commentary
Bowel injury in spinal surgery is an uncommon event but well documented for traumatic SCI with associated thoracolumbar fracture/dislocations, interbody spacer migration, and prominent anterior instrumentation. As more lateral, anterior, and redo lateral surgery is performed on an aging population and more spinal surgeons are doing their own approaches, bowel injury may increase. A low threshold for postoperative CT-abdomen/pelvis as a routine precautionary measure may be needed to minimize the potential missed bowel injury or to provide an earlier diagnosis of bowel perforation versus paralytic ileus. Early mobilization, minimization of postoperative narcotics, and optimization of intraoperative anesthesia during surgery can minimize the incidence of ileus in this patient group, but the quick diagnosis and management of bowel perforation is the key to minimizing morbidity and mortality. Early general surgery consultation should be sought in any spine surgery cases with a questionable abdominal exam or postoperative radiographic findings.
Jamie Baisden, M.D.
Professor of Neurosurgery
MCW –Neurosurgery
HUB 4th Floor
8701. Watertown Plank Road
Milwaukee, WI 53226
Cell 262-902-0059
414-248-4997
References
1. Aoki Y, Takahashi H, Nakamina A, Kubota G, Watanabe A, Nakajima T. Prevalence of lumbar spondylolysis and spondylolisthesis in patients with degenerative spinal disease. Sci Rep. 2020. 10: 6739
2. Balsano M, Carlucci S, Ose M, Boriani L. A case report of a rare complication of bowel perforation in extreme lateral interbody fusion. Eur Spine J. 2015. 24: 405-8
3. Bielecki K, Kaminski P, Klukowski M. Large bowel perforation: Morbidity and mortality. Tech Coloproctol. 2002. 6: 177-82
4. Biondo S, Pares D, Rague JM, de Oca J, Toral D, Borobia FG. Emergency operations for nondiverticular perforation of the left colon. Am J Surg. 2002. 183: 256-60
5. Epstein NE. Non-neurological major complications of extreme lateral and related lumbar interbody fusion techniques. Surg Neurol Int. 2016. 7: S656-9
6. Epstein NE. Review of risks and complications of extreme lateral inter-body fusion (XLIF). Surg Neurol Int. 2019. 10: 237
7. Fujibayashi S, Kawakami N, Asazuma T, Ito M, Mitzutani J, Nagashima H. Complications associated with lateral inter-body fusion: Nationwide survey of 2998 cases during the first 2 years of its use in Japan. Spine (Phila Pa 1976). 2017. 42: 1478-84
8. Hiyama A, Katoh H, Sakai D, Tanaka M, Sto M, Watanabe M. Short-term comparison of preoperative and postoperative pain after indirect decompression surgery and direct decompression surgery in patients with degenerative spondylolisthesis. Sci Rep. 2020. 10: 18887
9. Hwang ES, Kim KJ, Lee CS, Lee MY, Yoon SJ, Park JW. Bowel injury and insidious pneumoperitoneum after lateral lumbar inter-body fusion. Asian Spine J. 2021. p.
10. Isaacs RE, Sembrano JN, Tohmeh AG. SOLAS Degenerative Study Group. Two-Year comparative outcomes of MIS lateral and MIS transforaminal interbody fusion in the treatment of degenerative spondylolisthesis: Part II: Radiographic findings. Spine (Phila Pa 1976). 2016. 41: S133-44
11. Januszewski J, Keem SK, Smith W, Beckman JM, Kanter AS, Oskuian RJ. The potentially fatal Ogilvie’s syndrome in lateral transpsoas access surgery: A multi-institutional experience with 2930 patients. World Neurosurg. 2017. 99: 302-7
12. Khajavi K, Shen A, Lagina M, Hutchison A. Comparison of clinical outcomes following minimally invasive lateral interbody fusion. Eur Spine J. 2015. 24: 322-30
13. Li J, Wang X, Sun Y, Zhang F, Gao Y, Li Z. Safety analysis of two anterior lateral lumbar inter-body fusions at the initial stage of learning curve. World Neurosurg. 2019. 127: E901-9
14. Marquez-Lara A, Nandyala SV, Hassanzadeh H, Sundberg E, Jorgensen A, Singh K. Sentinel events in lumbar spine surgery. Spine (Phila Pa 1976). 2014. 39: 900-5
15. Paterakis KN, Brotis AG, Paschalis A, Tzannis A, Fountas KN. Extreme lateral lumbar interbody fusion (XLIF) in the management of degenerative scoliosis: A retrospective case series. J Spine Surg. 2018. 4: 610-5
16. Pereira EAC, Farwana M, Lam KS. Extreme lateral interbody fusion relieves symptoms of spinal stenosis and low-grade spondylolisthesis by indirect decompression in complex patients. J Clin Neurosci. 2017. 35: 56-61
17. Rustagi T, Yilmaz E, Alonso F, Schmidt C, Oskouian R, Tubbs RS. Iatrogenic bowel injury following minimally invasive lateral approach to the lumbar spine: A retrospective analysis of 3 cases. Global Spine J. 2019. 9: 375-82
18. Segawa T Inanami H, Koga H. Clinical evaluation of microendoscopy-assisted extreme lateral interbody fusion. J Spine Surg. 2017. 3: 398-402
19. Siasos I, Vakharia K, Khan A, Meyers JE, Yavorck S, Pollina J. Bowel injury in lumbar spine surgery: A review of the literature. J Spine Surg. 2018. 4: 130-7
20. Tamburrelli FC, Meluzio MC, Burrofato A, Perna A, Proietti L. Minimally invasive surgery procedure in isthmic spondylolisthesis. Eur Spine J. 2018. 27: 237-43
21. Tormenti MJ, Maserati MB, Bonfield CM, Okonkwo DO, Kanter AS. Complications and radiographic correction in adult scoliosis following combined transpsoas extreme lateral interbody fusion and posterior pedicle screw instrumentation. Neurosurg Focus. 2010. 28: E7
22. Uribe JS, Deukmedjian AR. Visceral, vascular, and wound complications following over 13, 000 lateral inter-body fusions: A survey study and literature review. Eur Spine J. 2015. 24: S386-96