- Department of Neurosurgery, University Hospital Dr. Negrín, Gran Canaria, Canary Islands, Spain
- Department of Neurosurgery, Stroke Center, Teishinkai Hospital, Sapporo, Hokkaido, Japan
Correspondence Address:
Aruma J-O’Shanahan
Department of Neurosurgery, University Hospital Dr Negrín, Gran Canaria, Canary Islands, Spain
DOI:10.4103/2152-7806.179581
Copyright: © 2016 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Aruma J-O’Shanahan, Noda K, Tsuboi T, Ota N, Kamiyama H, Tokuda S, Tanikawa R. Radical surgical treatment for recurrent giant fusiform thrombosed vertebral artery aneurysm previously coiled. Surg Neurol Int 01-Apr-2016;7:
How to cite this URL: Aruma J-O’Shanahan, Noda K, Tsuboi T, Ota N, Kamiyama H, Tokuda S, Tanikawa R. Radical surgical treatment for recurrent giant fusiform thrombosed vertebral artery aneurysm previously coiled. Surg Neurol Int 01-Apr-2016;7:. Available from: http://surgicalneurologyint.com/surgicalint_articles/radical-surgical-treatment-for-recurrent-giant-fusiform-thrombosed-vertebral-artery-aneurysm-previously-coiled/
Abstract
Background: Fusiform aneurysms are rare (less than 1%) and the underlying pathophysiology is not well known. Endovascular coiling is the standard of treatment; however, a surgical procedure with vascular reconstruction by excluding the pathological segment of the vessel and restoring the blood flow, seems to be the most effective and definitive treatment.
Case Description: We report a patient who presented a fusiform vertebral artery aneurysm previously coiled which developed a giant enlargement and a new contralateral fusiform aneurysm. Hemodynamic changes resulting in the formation of contralateral aneurysm might be the result of aneurysm occlusion without revascularization. In addition, continued blood flow to the aneurysmal wall through the vasa vasorum might result in aneurysm recanalization or regrowth. In order to account for these possible sources of complications, we performed a vascular reconstruction with high and low flow bypasses after trapping the aneurysm.
Conclusions: We hypothesize that, in this and similar cases, surgical vascular reconstruction should be the first and definitive treatment under experienced cerebrovascular surgeons.
Keywords: Endovascular coiling, fusiform aneurysm, revascularization, vasa vasorum, vertebral artery
INTRODUCTION
Fusiform aneurysms are rare (<1%)[
CASE REPORT
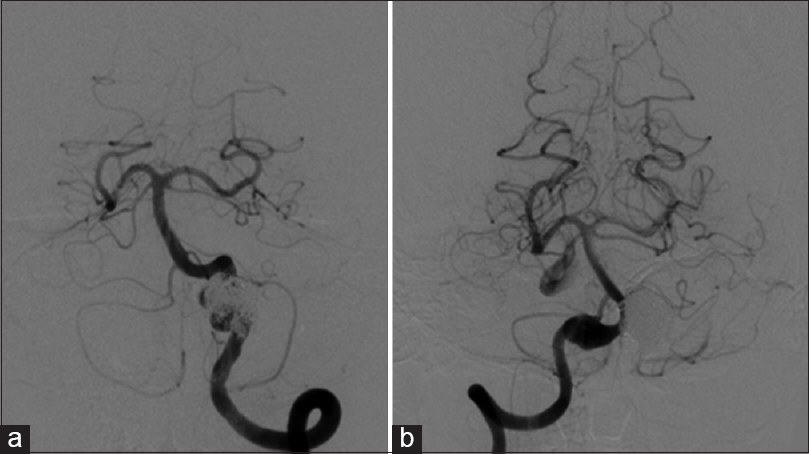
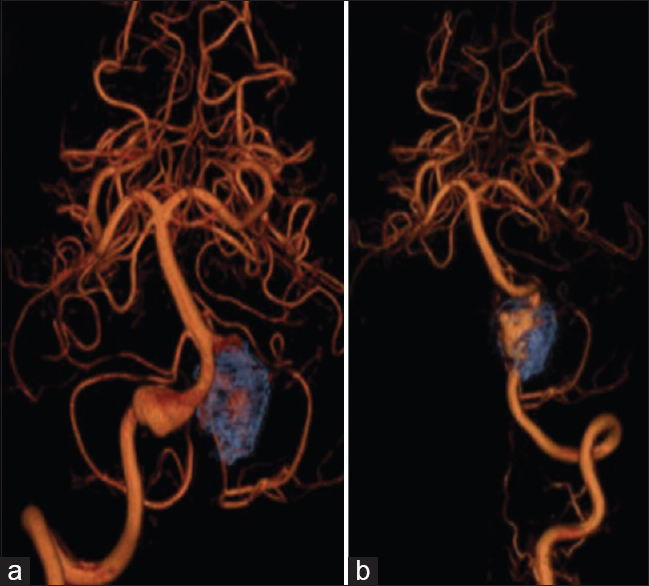
An incidental left fusiform vertebral artery (VA) aneurysm was coiled in a 60-year-old man [
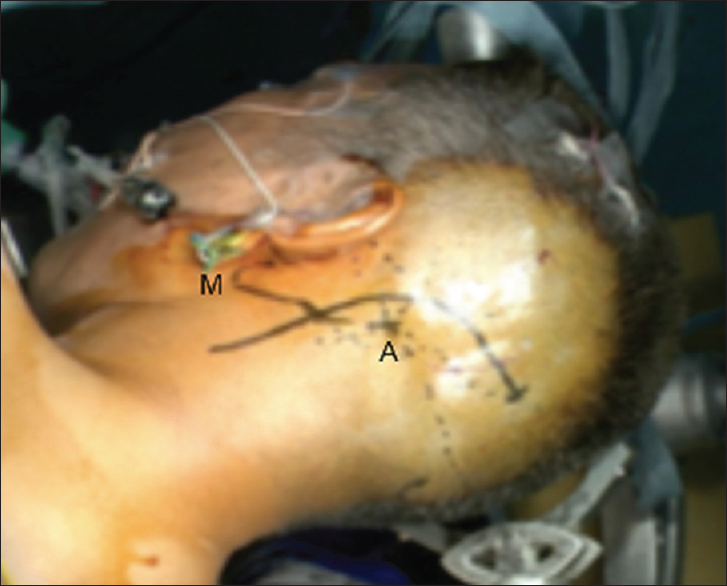
The surgery was performed under neurophysiological monitoring. The patient was placed in a right park bench position. A handheld Doppler was used to find the subcutaneous occipital artery (OA). An L-shape postauricular skin incision was made [
Figure 6
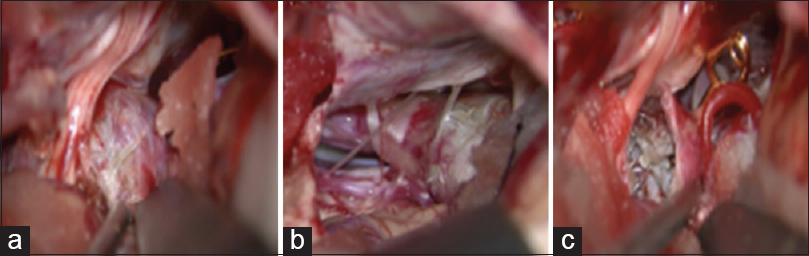
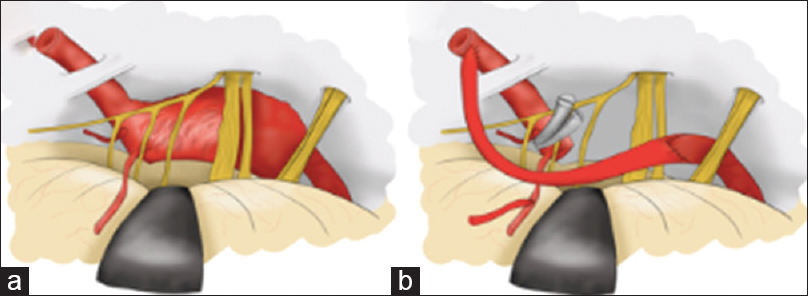
Illustration depicting giant fusiform coiled aneurysm located ventral to the XI, X, IX and VII, VIII cranial nerves (a). Surgical proximal VA trapping. Giant coiled aneurysm was removed. Low flow bypass: Occipital artery-small branch arising proximal from the aneurysm, end-to-side anastomosis. High flow bypass: Radial artery graft-intracranial V4 end-to-end and radial artery graft-V3-extracranial end-to-side fashion (b)
A fish mouth shape trimming was performed on the donor vessel to ensure endothelial layers of donor and recipient attached together.
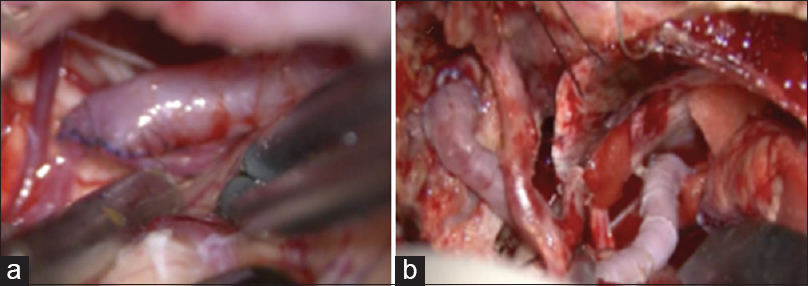
After temporary trapping, the aneurysm coil mass was incised by monopolar coagulator [
Intraoperative indocyanine green video angiography, Doppler sonography, and a transit-time blood flow meter were done to confirm bypass patency and perforator arteries integrity. Proximal aneurysm clipped reinforced was applied [
The patient could walk without assistance after 7 days, although left thermal hypoalgesia, due to Wallenberg's syndrome appeared because of small infarction of medulla oblongata [
Figure 9
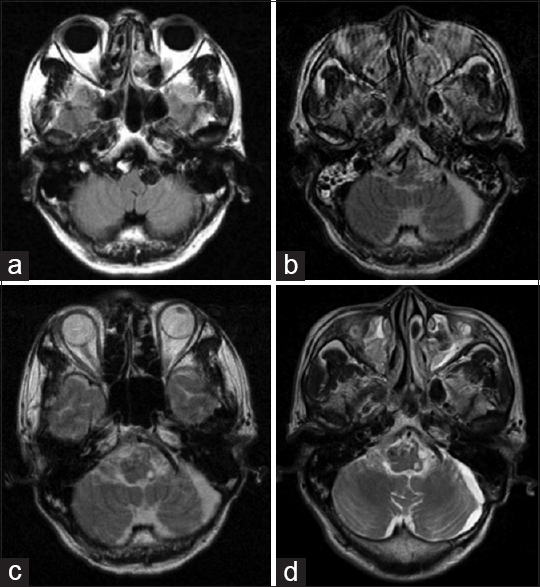
Magnetic resonance imaging (MRI) shows enlargement of the left fusiform VA aneurysm and a new contralateral right fusiform VA aneurysm (2013) (a). MRIs immediately after surgery (b), 6 months (c) and 18 months (d) later show small left infarction of medulla oblongata and no changes in the right fusiform VA aneurysm
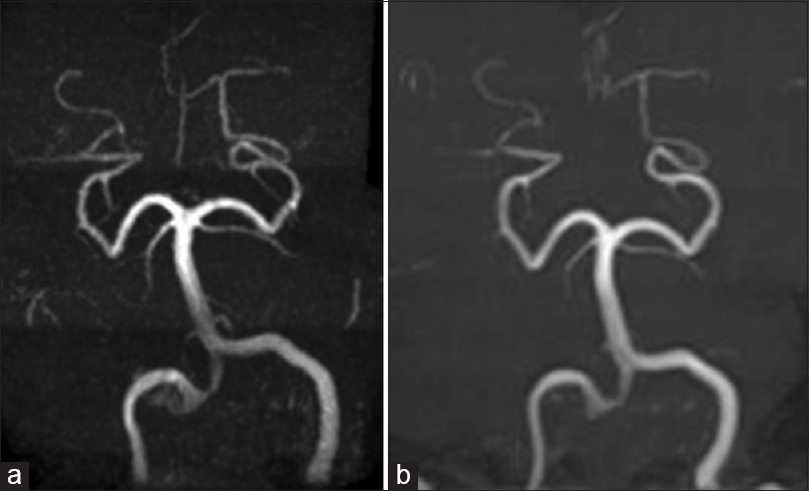
Radiological images confirmed no changes on the right fusiform VA aneurysm size [Figures
DISCUSSION
Vertebral fusiform aneurysm may be caused by dissection or an atherosclerotic change. Many classifications have been proposed to differentiate between dolichoectatic, fusiform, transitional, giant serpentine, or dissecting aneurysms,[
Endothelial damage on a preexisting weakness of the vessel wall (with/without an atherosclerotic plaque) and the presence of hemodynamic changes may lead to the fragmentation of the internal elastic lamina and to intimal hyperplasia with neovascularization of the wall.[
Symptoms commonly result from compression of neuronal structures, ischemic stroke or aneurysm rupture.[
Endovascular procedures involve few periprocedural complications[
More data and understanding regarding the VB fusiform aneurysm nature and behavior are needed before we can consider it a safe treatment option.[
It is logical to assume that an internal trapping by endovascular procedures cannot stop the blood feeding into the aneurysmal wall through the vasa vasorum.[
Surgical procedures are the most curative and definitive treatments.[
Series combining endovascular and surgical treatment have reported a mortality rate of 12.5–45%.[
We removed the aneurysm and performed a cerebrovascular reconstruction to avoid excessive hemodynamic load to the contralateral VA aneurysm. Unfortunately, because a perforator artery was arising from the VA close to the aneurysm, it may have been occluded after trapping the aneurysm. It could have been prevented if the ante grade flow through the intracranial VA had been maintained by an interposed graft reconstruction between intracranial proximal V4 and distal V4 segment after removing the aneurysm.
Hemodynamic stress and preexisting arterial wall weakness might be responsible for contralateral aneurysm formation after sacrificing the left VA.[
CONCLUSIONS
Summarizing, the vascular wall pathophysiology on fusiform aneurysms makes endovascular treatment a noncurative option. A previously coiled fusiform aneurysm may grow and become giant. The surgical excision on a coiled aneurysm becomes more difficult and the prognosis worsens. A first microsurgical reconstruction approach in experienced hands can prevent recurrence of the aneurysm and improve the patients’ prognosis.
Financial support and sponsorship
This manuscript was written while the main author was visiting Teishinkai Hospital in Sapporo, Japan. Her visit was supported by a grant from the Doctor Manuel Morales Foundation (La Palma, Canary Islands. Spain).
Conflicts of interest
There are no conflicts of interest.
Acknowledgements
I would like to thank Yeray Nóvoa and Ferzat Hijazy for their help in editing this manuscript and Eder J-O΄Shanahan for his help with the illustration.
References
1. Ahn JY, Han IB, Kim TG, Yoon PH, Lee YJ, Lee BH. Endovascular treatment of intracranial vertebral artery dissections with stent placement or stent-assisted coiling. J Neuroradiol. 2006. 27: 1514-20
2. Anson JA, Lawton MT, Spetzler RF. Characteristics and surgical treatment of dolichoectatic and fusiform aneurysms. J Neurosurg. 1996. 84: 185-93
3. Aydin F. Do human intracranial arteries lack vasa vasorum? A comparative immunohistochemical study of intracranial and systemic arteries. Acta Neuropathol. 1998. 96: 22-8
4. Aymard A, Gobin YP, Hodes JE, Bien S, Rüfenacht D, Reizine D. Endovascular occlusion of vertebral arteries in the treatment of unclippable vertebrobasilar aneurysms. J Neurosurg. 1991. 74: 393-8
5. Byrne JV, Beltechi R, Yarnold JA, Birks J, Kamran M. Early experience in the treatment of intra-cranial aneurysms by endovascular flow diversion: A multicentre prospective study. PLoS One. 2010. 5: pii: E12492-
6. Coert BA, Chang SD, Do HM, Marks MP, Steinberg GK. Surgical and endovascular management of symptomatic posterior circulation fusiform aneurysms. J Neurosurg. 2007. 106: 855-65
7. Cohen JE, Gomori JM, Moscovici S, Itshayek E. Successful endovascular treatment of a growing megadolichoectasic vertebrobasilar artery aneurysm by flow diversion using the “diverter-in-stent” technique. J Clin Neurosci. 2012. 19: 166-70
8. Dandy WE. Intracranial arterial aneurysms. Am J Med Sci. 1944. 208: 415-
9. Drake CG, Peerless SJ. Giant fusiform intracranial aneurysms: Review of 120 patients treated surgically from 1965 to 1992. J Neurosurg. 1997. 87: 141-62
10. Fiorella D, Hsu D, Woo HH, Tarr RW, Nelson PK. Very late thrombosis of a pipeline embolization device construct: Case report. Neurosurgery. 2010. 67: onsE313-4
11. Fiorella D, Woo HH, Albuquerque FC, Nelson PK. Definitive reconstruction of circumferential, fusiform intracranial aneurysms with the pipeline embolization device. Neurosurgery. 2008. 62: 1115-20
12. Flemming KD, Wiebers DO, Brown RD, Link MJ, Nakatomi H, Huston J. Prospective risk of hemorrhage in patients with vertebrobasilar nonsaccular intracranial aneurysm. J Neurosurg. 2004. 101: 82-7
13. Fujita K, Yamashita H, Masumura M, Nishizaki T, Tamaki N, Matsumoto S. Natural history of giant intracranial aneurysms. No Shinkei Geka. 1988. 16: 225-31
14. Gobin YP, Viñuela F, Gurian JH, Guglielmi G, Duckwiler GR, Massoud TF. Treatment of large and giant fusiform intracranial aneurysms with Guglielmi detachable coils. J Neurosurg. 1996. 84: 55-62
15. Guglielmi G, Viñuela F, Duckwiler G, Dion J, Lylyk P, Berenstein A. Endovascular treatment of posterior circulation aneurysms by electrothrombosis using electrically detachable coils. J Neurosurg. 1992. 77: 515-24
16. Hayes WT, Bernhardt H, Young JM. Fusiform arteriosclerotic aneurysm of the basilar artery. Five cases including two ruptures. Vasc Surg. 1967. 1: 171-8
17. Hoh BL, Putman CM, Budzik RF, Carter BS, Ogilvy CS. Combined surgical and endovascular techniques of flow alteration to treat fusiform and complex wide-necked intracranial aneurysms that are unsuitable for clipping or coil embolization. J Neurosurg. 2001. 95: 24-35
18. Hosoya T, Adachi M, Yamaguchi K, Haku T, Kayama T, Kato T. Clinical and neuroradiological features of intracranial vertebrobasilar artery dissection. Stroke. 1999. 30: 1083-90
19. Housepian EM, Pool JL. A systematic analysis of intracranial aneurysms from the autopsy file of the Presbyterian hospital, 1914 to 1956. J Neuropathol Exp Neurol. 1958. 17: 409-23
20. Iihara K, Murao K, Sakai N, Soeda A, Ishibashi-Ueda H, Yutani C. Continued growth of and increased symptoms from a thrombosed giant aneurysm of the vertebral artery after complete endovascular occlusion and trapping: The role of vasa vasorum. Case report. J Neurosurg. 2003. 98: 407-13
21. Iihara K, Sakai N, Murao K, Sakai H, Higashi T, Kogure S. Dissecting aneurysms of the vertebral artery: A management strategy. J Neurosurg. 2002. 97: 259-67
22. Inui Y, Oiwa Y, Terada T, Nakakita K, Kamei I, Hayashi S. De novo vertebral artery dissecting aneurysm after contralateral vertebral artery occlusion – Two case reports. Neurol Med Chir (Tokyo). 2006. 46: 32-6
23. Kalani MY, Zabramski JM, Nakaji P, Spetzler RF. Bypass and flow reduction for complex basilar and vertebrobasilar junction aneurysms. Neurosurgery. 2013. 72: 763-75
24. Katsuno M, Mizunari T, Kobayashi S, Takahashi H, Teramoto A. Rupture of a vertebral artery dissecting aneurysm developing immediately after trapping of a dissecting aneurysm on the contralateral vertebral artery: Case report. Neurol Med Chir (Tokyo). 2009. 49: 468-70
25. Kondo S, Hashimoto N, Kikuchi H, Hazama F, Nagata I, Kataoka H. Cerebral aneurysms arising at nonbranching sites. An experimental Study. Stroke. 1997. 28: 398-403
26. Kubota H, Tanikawa R, Katsuno M, Noda K, Ota N, Miyata S. Reconstruction of intracranial vertebral artery with radial artery and occipital artery grafts for fusiform intracranial vertebral aneurysm not amenable to endovascular treatment: Technical note. Acta Neurochir (Wien). 2013. 155: 1517-24
27. Lanzino G, Wakhloo AK, Fessler RD, Hartney ML, Guterman LR, Hopkins LN. Efficacy and current limitations of intravascular stents for intracranial internal carotid, vertebral, and basilar artery aneurysms. J Neurosurg. 1999. 91: 538-46
28. Leibowitz R, Do HM, Marcellus ML, Chang SD, Steinberg GK, Marks MP. Parent vessel occlusion for vertebrobasilar fusiform and dissecting aneurysms. J Neuroradiol. 2003. 24: 902-7
29. Lylyk P, Miranda C, Ceratto R, Ferrario A, Scrivano E, Luna HR. Curative endovascular reconstruction of cerebral aneurysms with the pipeline embolization device: The Buenos Aires experience. Neurosurgery. 2009. 64: 632-42
30. Mangrum WI, Huston J, Link MJ, Wiebers DO, McClelland RL, Christianson TJ. Enlarging vertebrobasilar nonsaccular intracranial aneurysms: Frequency, predictors, and clinical outcome of growth. J Neurosurg. 2005. 102: 72-9
31. Mericle RA, Wakhloo AK, Lopes DK, Lanzino G, Guterman LR, Hopkins LN. Delayed aneurysm regrowth and recanalization after Guglielmi detachable coil treatment. Case report. J Neurosurg. 1998. 89: 142-5
32. Milandre L, Bonnefoi B, Pestre P, Pellissier JF, Grisoli F, Khalil R. Vertebrobasilar arterial dolichoectasia. Complications and prognosis. Rev Neurol (Paris). 1991. 147: 714-22
33. Miyamoto S, Funaki T, Iihara K, Takahashi JC. Successful obliteration and shrinkage of giant partially thrombosed basilar artery aneurysms through a tailored flow reduction strategy with bypass surgery. J Neurosurg. 2011. 114: 1028-36
34. Mizutani T, Aruga T, Kirino T, Miki Y, Saito I, Tsuchida T. Recurrent subarachnoid hemorrhage from untreated ruptured vertebrobasilar dissecting aneurysms. Neurosurgery. 1995. 36: 905-11
35. Mizutani T, Miki Y, Kojima H, Suzuki H. Proposed classification of nonatherosclerotic cerebral fusiform and dissecting aneurysms. Neurosurgery. 1999. 45: 253-9
36. Nagahiro S, Takada A, Goto S, Kai Y, Ushio Y. Thrombosed growing giant aneurysms of the vertebral artery: Growth mechanism and management. J Neurosurg. 1995. 82: 796-801
37. Nagasawa S, Kawanishi M, Tada Y, Kawabata S, Ohta T. Simulation of therapeutic parent artery occlusion for basilar head aneurysms. Hemodynamic effect of occlusion sites and diameters of collateral arteries. Neurol Res. 1999. 21: 180-4
38. Nakatomi H, Segawa H, Kurata A, Shiokawa Y, Nagata K, Kamiyama H. Clinicopathological study of intracranial fusiform and dolichoectatic aneurysms: Insight on the mechanism of growth. Stroke. 2000. 31: 896-900
39. Nelson PK, Lylyk P, Szikora I, Wetzel SG, Wanke I, Fiorella D. The pipeline embolization device for the intracranial treatment of aneurysms trial. J Neuroradiol. 2011. 32: 34-40
40. Nijensohn DE, Saez RJ, Reagan TJ. Clinical significance of basilar artery aneurysms. Neurology. 1974. 24: 301-5
41. Nishizaki T, Tamaki N, Takeda N, Shirakuni T, Kondoh T, Matsumoto S. Dolichoectatic basilar artery: A review of 23 cases. Stroke. 1986. 17: 1277-81
42. Otawara Y, Ogasawara K, Ogawa A, Kogure T. Dissecting aneurysms of the bilateral vertebral arteries with subarachnoid hemorrhage: Report of three cases. Neurosurgery. 2002. 50: 1372-4
43. Pia HW. Classification of vertebro-basilar aneurysms. Acta Neurochir (Wien). 1979. 47: 3-30
44. Sacks JG, Lindenburg R. Dolicho-ectatic intracranial arteries: Symptomatology and pathogenesis of arterial elongation and distention. Johns Hopkins Med J. 1969. 125: 95-106
45. Saito N, Kamiyama H, Takizawa K, Takebayashi S, Asano T, Kobayashi T. Usefulness of V3-radial artery graft-V4 bypass in bilateral fusiform aneurysms of vertebral artery: Case report. Neurol Med Chir (Tokyo). 2014. 54: 189-91
46. Schubiger O, Valavanis A, Wichmann W. Growth-mechanism of giant intracranial aneurysms; demonstration by CT and MR imaging. Neuroradiology. 1987. 29: 266-71
47. Siddiqui AH, Abla AA, Kan P, Dumont TM, Jahshan S, Britz GW. Panacea or problem: Flow diverters in the treatment of symptomatic large or giant fusiform vertebrobasilar aneurysms. J Neurosurg. 2012. 116: 1258-66
48. Steinberg GK, Drake CG, Peerless SJ. Deliberate basilar or vertebral artery occlusion in the treatment of intracranial aneurysms. Immediate results and long-term outcome in 201 patients. J Neurosurg. 1993. 79: 161-73
49. Takikawa S, Kamiyama H, Nomura M, Abe H, Saitoh H. Vertebral dissecting aneurysm treated with trapping and bilateral posterior inferior cerebellar artery side-to side anastomosis; case report. No Shinkei Geka. 1991. 19: 571-6
50. Wakui K, Kobayashi S, Takemae T, Kamijoh Y, Nagashima H, Muraoka S. Giant thrombosed vertebral artery aneurysm managed with extracranial-intracranial bypass surgery and aneurysmectomy. Case report. J Neurosurg. 1992. 77: 624-7
51. Yasui T, Komiyama M, Iwai Y, Yamanaka K, Nishikawa M, Morikawa T. Evolution of incidentally-discovered fusiform aneurysms of the vertebrobasilar arterial system: Neuroimaging features suggesting progressive aneurysm growth. Neurol Med Chir (Tokyo). 2001. 41: 523-7
52. Yasui T, Komiyama M, Nishikawa M, Nakajima H, Kobayashi Y, Inoue T. Fusiform vertebral artery aneurysms as a cause of dissecting aneurysms. Report of two autopsy cases and a review of the literature. J Neurosurg. 1999. 91: 139-44
53. Yoshimoto T, Kamiyama H, Abe H, Takikawa S, Ito T. Proximal clipping and bypass between bilateral vertebral arteries using a radial arterial graft for the treatment of a dissecting aneurysm of the vertebral artery. Surg Neurol. 1991. 36: 476-81
54. Yu YL, Moseley IF, Pullicino P, McDonald WI. The clinical picture of ectasia of the intracerebral arteries. J Neurol Neurosurg Psychiatry. 1982. 45: 29-36