- Department of Neurosurgery, Wakayama Rosai Hospital, Wakayama, Wakayama, Japan
Correspondence Address:
Nobuhide Hayashi
Department of Neurosurgery, Wakayama Rosai Hospital, Wakayama, Wakayama, Japan
DOI:10.4103/2152-7806.200578
Copyright: © 2017 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Nobuhide Hayashi, Takahiro Sasaki, Nagatsuki Tomura, Hideo Okada, Toshikazu Kuwata. Removal of a malignant cystic brain tumor utilizing pyoktanin blue and fibrin glue: Technical note. 20-Feb-2017;8:24
How to cite this URL: Nobuhide Hayashi, Takahiro Sasaki, Nagatsuki Tomura, Hideo Okada, Toshikazu Kuwata. Removal of a malignant cystic brain tumor utilizing pyoktanin blue and fibrin glue: Technical note. 20-Feb-2017;8:24. Available from: http://surgicalneurologyint.com/surgicalint_articles/removal-of-a-malignant-cystic-brain-tumor-utilizing-pyoktanin-blue-and-fibrin-glue-technical-note/
Abstract
Background:The leakage of cystic fluid during metastatic cystic brain tumor resection may cause tumor dissemination. When the cyst wall is thin, excision without removing the wall is often difficult.
Methods:We were able to perform an en bloc resection of a cystic malignant brain tumor after aspirating the cystic fluid, injecting pyoktanin blue into the cyst to stain the cyst walls, and solidifying the empty cyst cavity by filling it with fibrin glue.
Results:Pyoktanin blue readily stained the thin cystic walls and enabled visualization of mural damage. Solidification of the tumor made it easier to grasp and facilitated the dissection of tumor margins.
Conclusions:This method has the potential to become a useful technique for the resection of malignant cystic brain tumors.
Keywords: Cystic malignant brain tumor, fibrin glue, operative technique, pyoktanin blue
INTRODUCTION
Leakage of cystic fluid during resection of a metastatic cystic brain tumor may cause tumor dissemination.[
CASE REPORT
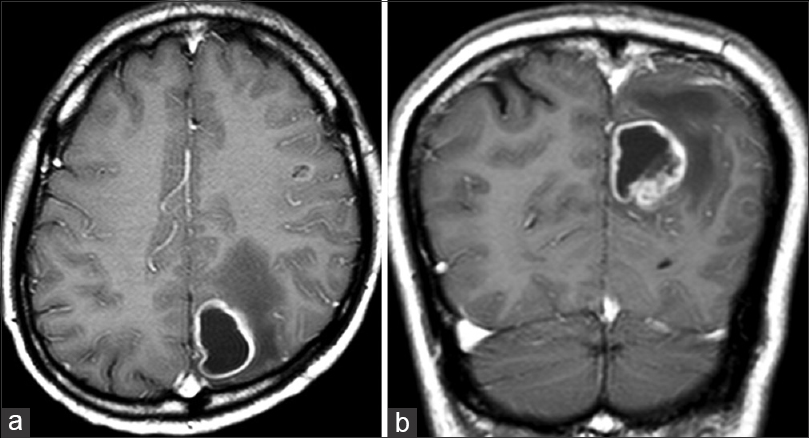
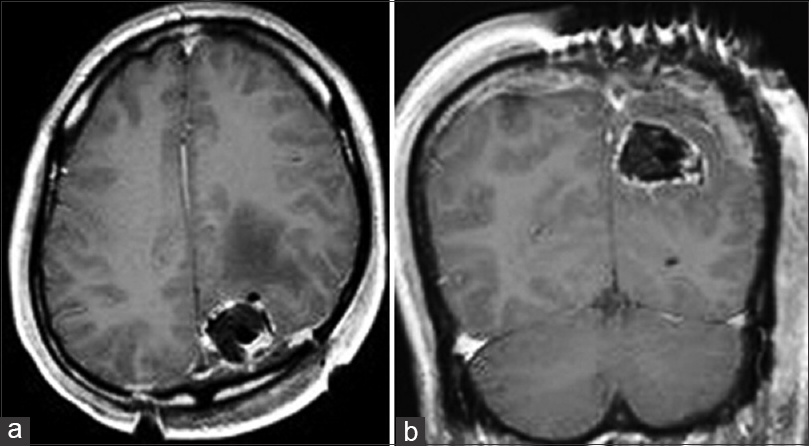
The patient, a 58-year-old man, suffered an epileptic seizure. Contrast-enhanced magnetic resonance imaging (MRI) revealed ring-enhancement lesions in the left parietal lobe and peritumoral cerebral edema associated with a cystic mass lesion [
Surgical operative technique
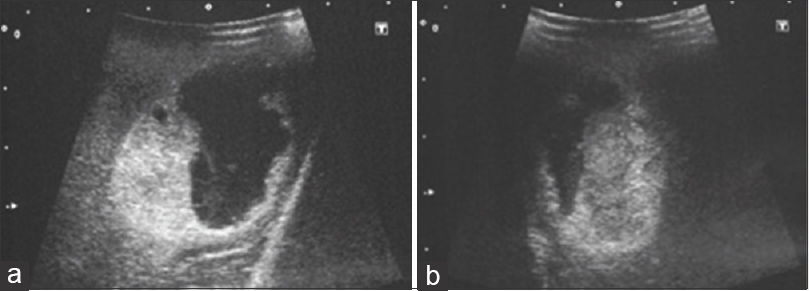
After the craniotomy, we determined the tumor cyst location and depth by ultrasonography [
Postoperative clinical course
At 19 months postoperatively, no adverse events due to this procedure were noted, and the clinical course of the patient was favorable with no neurological deficit and no epilepsy noted.
DISCUSSION
Thin cystic walls make dissection difficult during the resection of metastatic cystic brain tumors, and there is a danger that the leakage of cystic fluid will cause dissemination.[
The advantage of using pyoktanin blue[
Two methods have been reported for solidifying cysts: A method using autologous fibrin glue[
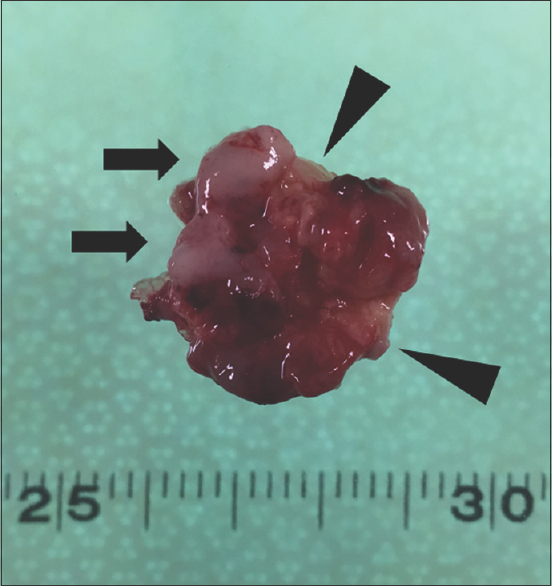
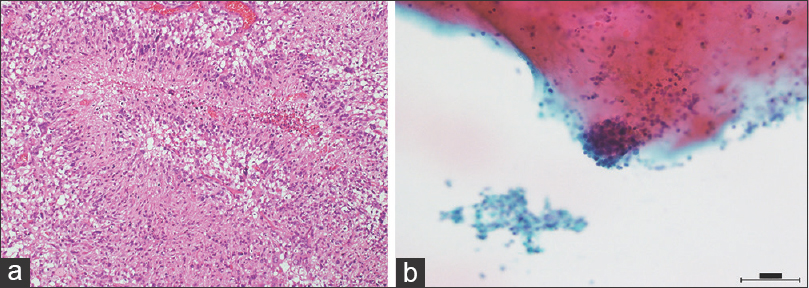
In the present case, frozen section pathology demonstrated a cystic glioblastoma and not a metastatic cystic brain tumor. To our knowledge, there are no reports concerning dissemination caused by cyst fluid leakage of cystic glioblastoma, nevertheless, the technique described was useful in insuring complete resection of the thin portions of the tumor cyst wall. If a tumor has been solidified by being filled with fibrin glue, the cystic fluid is aspirated, a working space is secured, and the tumor around the cystic wall is dissected away from the normal tissue. In contrast, when the working space is reduced, there is increased compression of the normal tissues, and the surrounding functional cerebral regions may also be affected. However, injection of fibrin glue into the collapsed cyst does not increase the tumor volume remarkably. In the present case, the neighboring cerebral surface was shaped like the tumor, although it was relatively easy to aspirate the cystic fluid and solidify the cyst lumen.
With regard to benign brain tumors that form cysts, in the cases of hemangioblastoma and pilocytic astrocytoma, excision of the mural nodule is sufficient and excision of the entire cyst is unnecessary. In addition, the method is difficult to adapt for deep lesions such as Rathkes's cleft cyst, cystic pituitary adenoma, craniopharyngioma and endodermal cyst. Therefore, this method may only be useful for metastatic cystic brain tumors with well-defined borders, and may be limited to comparatively superficial lesions. The technique we used to facilitate resection in this tumor case may also be useful for the management of brain abscess. Regarding the abscess capsule, if the wall is thick and pyoktanin blue staining is generally ineffective, depending on the bacterial species, pyoktanin blue exhibits antibacterial effects;[
CONCLUSIONS
Here, we report our experience with an excision method utilizing pyoktanin blue and fibrin glue for a cystic malignant brain tumor. Excision of the tumor as a lump could be performed without leakage of cyst fluid and removal of the cyst wall; this simple method may be a useful technique.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Acknowledgements
The authors thank Dr. Okuda for providing us the documents.
References
1. Glasgow BJ, Brown HH, Zargoza AM, Foos RY. Quantification of tumor seeding from needle aspiration of ocular melanomas. Am J Ophthalmol. 1988. 105: 538-46
2. Hoffmann CE, Rahn O. The bactericidal and bacteriostatic action of crystal violet. J Bacteriol. 1944. 47: 177-86
3. Mastronardi L, Cacciotti G, Caputi F, Roperto R, Tonelli MP, Carpineta E. Underlay hourglass-shaped autologous pericranium duraplasty in “key-hole” retrosigmoid approach surgery: Technical report. Surg Neurol Int. 2016. 7: 25-
4. Nakagawa H, Kimura S, Kubo S, Fujita T, Tsuruzono K, Hayakawa T. Prognostic Factors in Patients Surviving for more than 1 or 5 years after removal of metastatic brain tumors. Neurol Med Chir. 1992. 32: 947-51
5. Okuda T, Fujita M, Yoshioka H, Tasaki T, Kato A. Novel surgical technique to solidify cyst-type metastatic brain tumors using autologous fibrin glue for complete resection. Surg Neurol Int. 2014. 5: 100-
6. Okuda T, Teramoto Y, Yugami H, Kataoka K, Kato A. Surgical technique for a cystic-type metastatic brain tumor: Transformation to a solid-type tumor using hydrofiber dressing. Surg Neurol. 2009. 72: 703-6
7. Tomita Y, Sasaki T, Tanabe T, Idei M, Muraoka K, Terada K. Pyoktanin blue injection for resection of cystic brain tumor: A case report. No Shinkei Geka. 2013. 418: 687-91