- Department of Neurosurgery, Azienda Ospedaliera Universitaria di Sassari, Via Enrico De Nicola, Sassari, Italy,
- Medical Oncology Unit, University Hospital of Parma, Parma, Italy.
- Department of Anatomy and Pathological Histology, Azienda Ospedaliera Universitaria (AOU) di Sassari, Italy.
Correspondence Address:
Giosué Dipellegrini, Department of Neurosurgery, Azienda Ospedaliero Universitaria di Sassari, Via Enrico De Nicola 1, Sassari 07100, Italy.
DOI:10.25259/SNI_34_2023
Copyright: © 2023 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Giosué DiPellegrini1, Riccardo Boccaletti1, Anna Mingozzi2, Antonella Fara3, Domenico Policicchio1. Single thalamic localization of brain toxoplasmosis mimicking brain tumors: Radiological and clinical findings. 10-Mar-2023;14:82
How to cite this URL: Giosué DiPellegrini1, Riccardo Boccaletti1, Anna Mingozzi2, Antonella Fara3, Domenico Policicchio1. Single thalamic localization of brain toxoplasmosis mimicking brain tumors: Radiological and clinical findings. 10-Mar-2023;14:82. Available from: https://surgicalneurologyint.com/surgicalint-articles/12187/
Abstract
Background: Cerebral toxoplasmosis is a relatively rare disorder that usually affects immunocompromised patients. The most common scenario occurs among human immunodeficiency virus (HIV)-positive patients. In those patients, toxoplasmosis is the most frequent cause of expansive brain lesion and continues to cause elevated morbidity and mortality. In typical cases of toxoplasmosis, both computed tomography and magnetic resonance imaging reveal single/ multiple nodular or ring-enhancing lesions with surrounding edema. Nevertheless, cases of cerebral toxoplasmosis with atypical radiological features have been reported. Diagnosis can be obtained by finding organisms in the cerebrospinal fluid or in stereotactic biopsy samples of the brain lesion. If untreated, cerebral toxoplasmosis is uniformly fatal, so prompt diagnosis is mandatory. A prompt diagnosis is necessary, as untreated cerebral toxoplasmosis is uniformly fatal.
Case Description: We discuss imaging and clinical findings of a patient – not aware of being HIV-positive – with a solitary atypical brain localization of toxoplasmosis mimicking a brain tumor.
Conclusion: Although relatively uncommon, neurosurgeons should be aware of the potential occurrence of cerebral toxoplasmosis. High index of suspicion is needed for timely diagnosis and prompt initiation of therapy.
Keywords: Brain magnetic resonance imaging, Brain tumor, Cerebral toxoplasmosis, Stereotactic biopsy
INTRODUCTION
Acute Toxoplasma gondii infection is typically subclinical in the vast majority of immunocompetent subjects and it is very seldom associated with serious clinical events. In contrast, cerebral toxoplasmosis is caused almost exclusively by reactivating latent cerebral cysts and can have devastating consequences in host immunocompromised patients, particularly in people living with human immunodeficiency virus (HIV) disease or acquired immune deficiency syndrome (AIDS). In these patients, toxoplasmosis is the most common cause of expansive brain lesions and continues to cause high morbidity and mortality.[
In an HIV-positive patient, the discovery of single or multiple brain lesions should immediately lead to a suspicion of cerebral toxoplasmosis to rapidly begin the treatment and reduce the risk of progression.[
We discuss imaging and clinical findings of a 25-year-old patient – not aware of being HIV positive – with a single right thalamic localization of toxoplasmosis mimicking a brain tumor.
CASE DESCRIPTION
A 25-year-old woman was admitted to our emergency department with a subacute onset of mild confusion and left hemiparesis (Medical Research Council grade 4/5). She was fully alert and orientated, complaining about headaches (Visual Analogic Scale 7/10). She had no history of drug abuse or cigarette smoking.
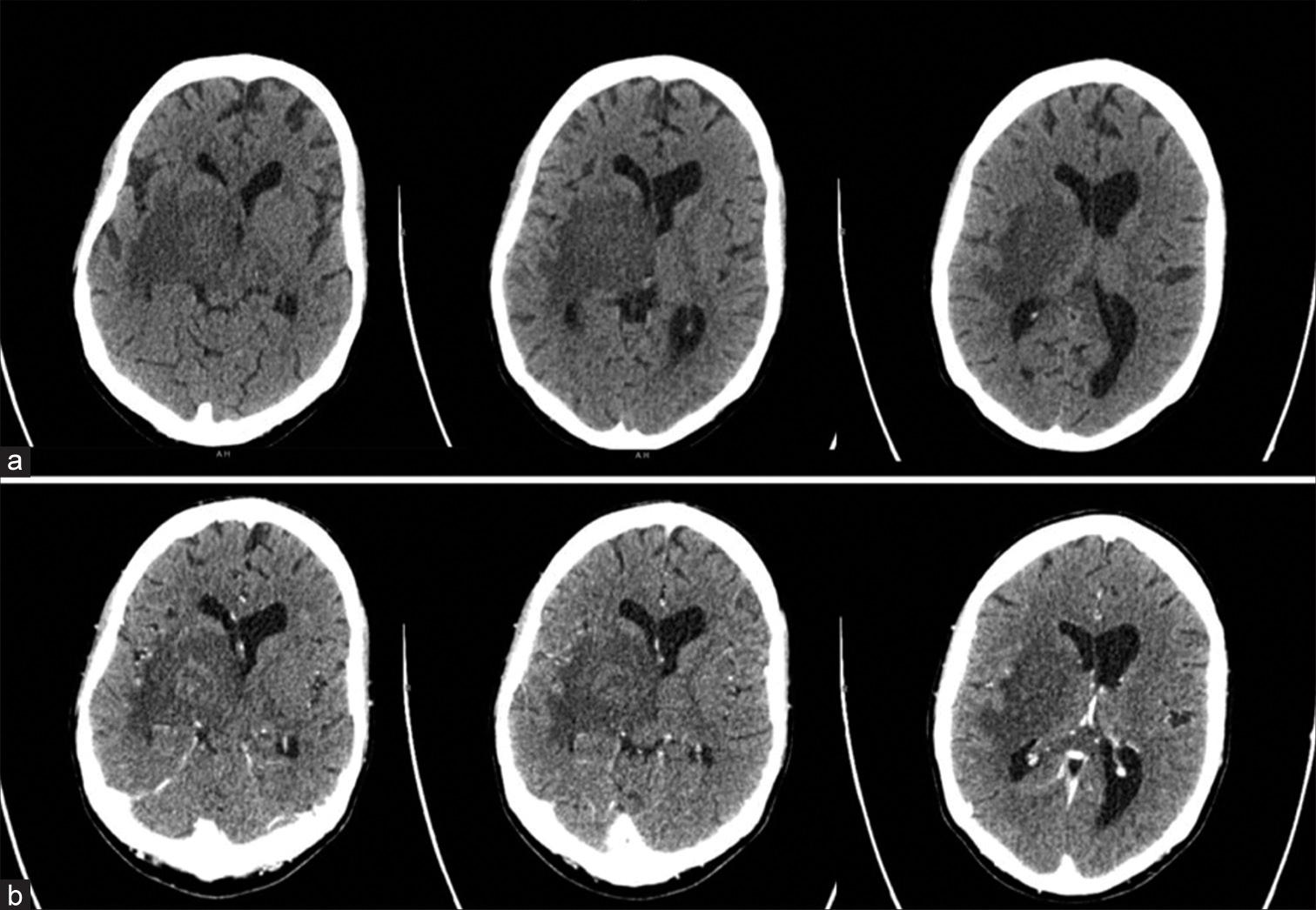
Pre- and postcontrast computed tomography (CT) scans of the brain [
Figure 2:
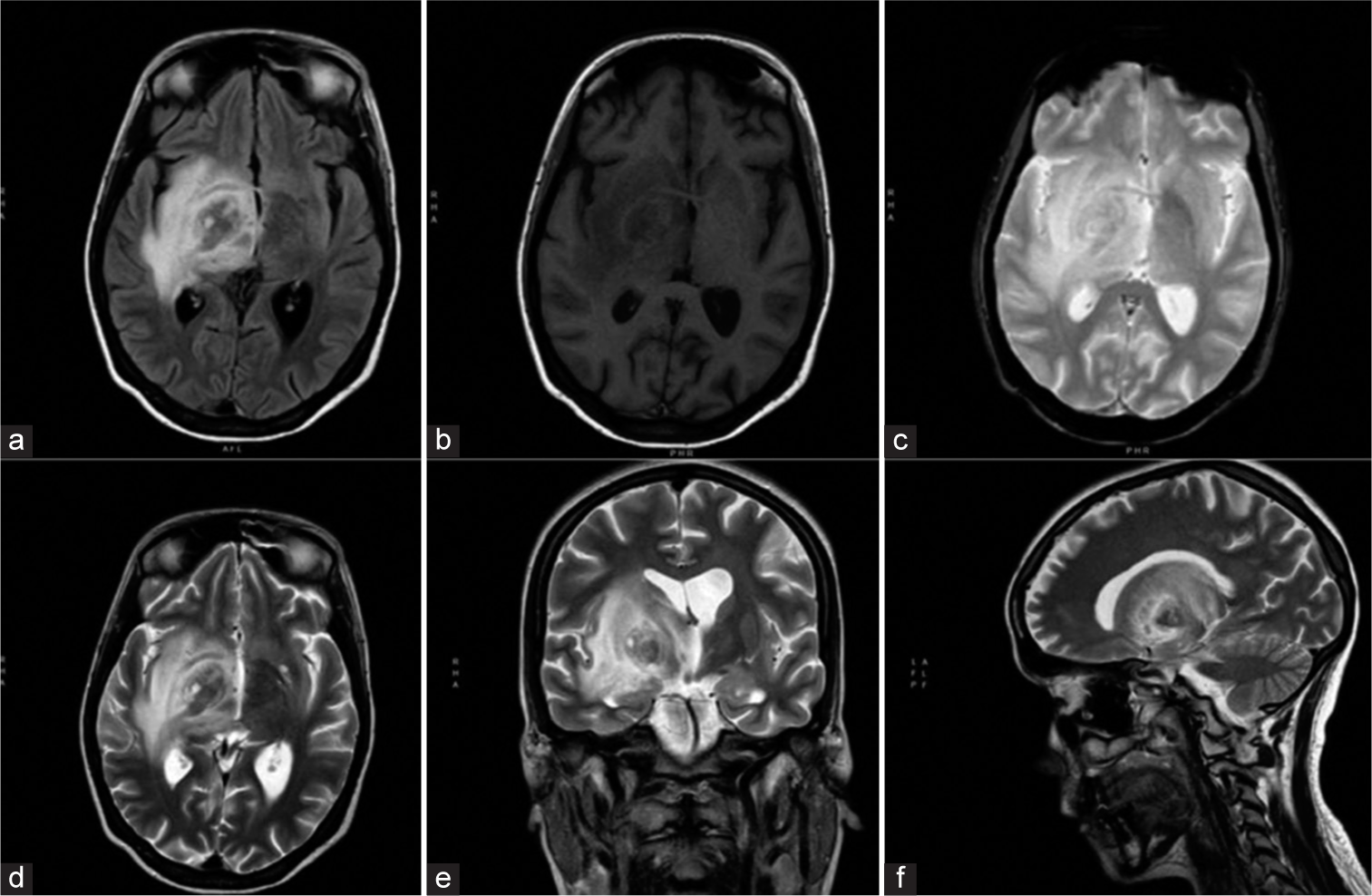
Brain magnetic resonance imaging without contrast showing a right thalamic nodular lesion measuring 25 × 22 mm, with perilesional edema. The nodule appeared hypointense in fluid-attenuated inversion recovery sequences (a), isointense on T1-weighted (b) and reg-diffusion-weighted imaging sequences (c), and hypointense on T2-weighted images (d-f). The nodule and associated edema resulted in slight compression of the ipsilateral ventricle.
The clinical presentation (without any known history of immune suppression) and the neuroimaging features of the solitary brain lesion primarily suggested a brain tumor, even if other diagnostic hypotheses could not be rejected.
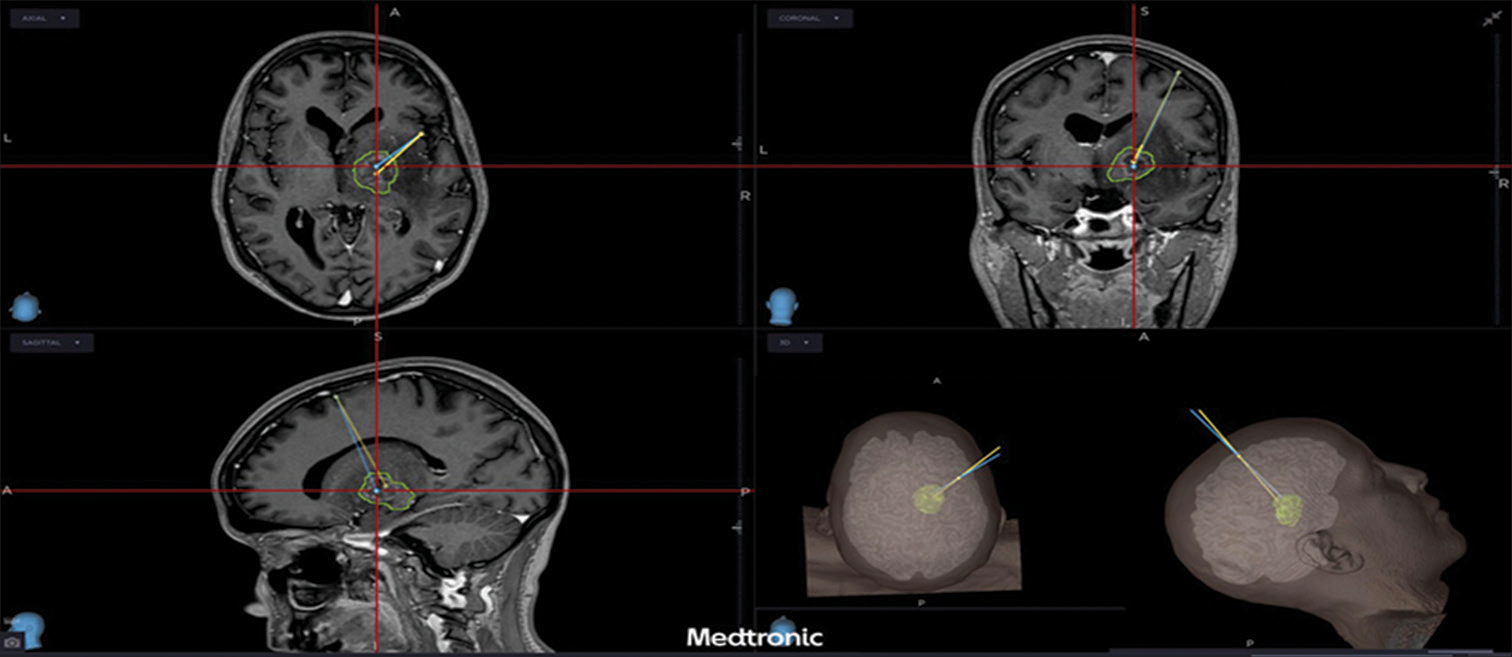
Considering the deep location in an eloquent area, it was decided to perform a neuronavigated stereotaxic biopsy [
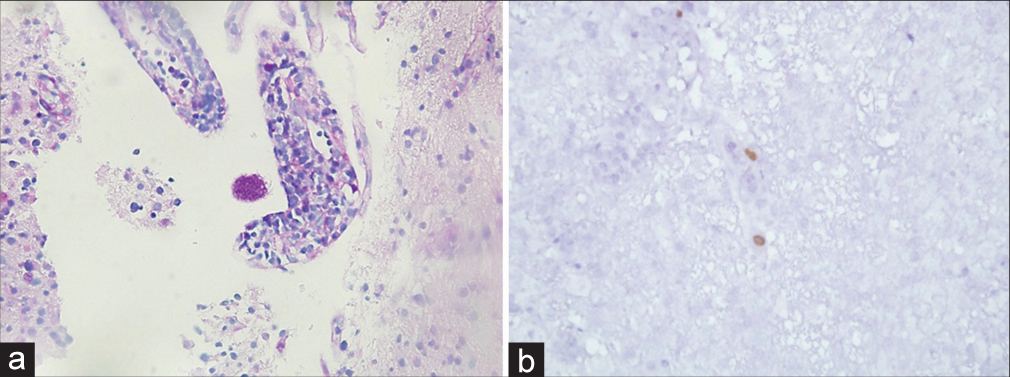
A coronal trajectory was used passing lateral to the lateral ventricle wall. Histopathology revealed extensive areas of necrosis, vascular thrombosis phenomena and aspects of vasculitis with lymphohistiocytic and granulocytic infiltrate at the level of the vascular walls. Rounded structures containing eosinophilic corpuscles inside the infiltrate were found, suggesting a protozoal infection, and allowing the diagnosis of cerebral toxoplasmosis [
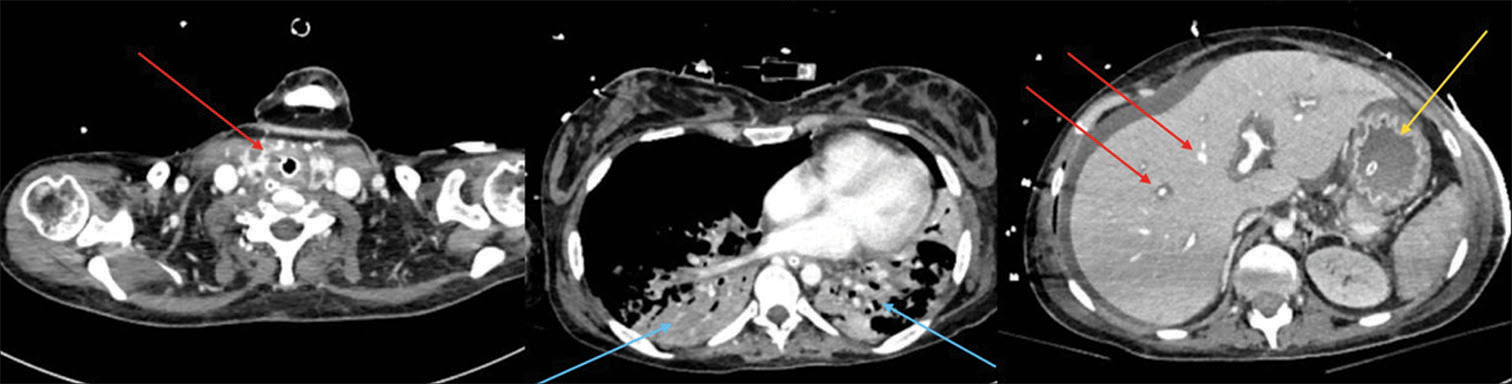
The immediate postoperative course was uneventful without complications. Nevertheless, in the following days, the patient presented with fever and abdominal pain with rapid clinical worsening and septic shock. Therefore, the patient was then transferred to intensive care unit. A total-body CT scan revealed multiple abscesses (liver, kidney, and thyroid gland), intestinal walls thickening, and pleural effusion [
Due to the septic shock and the diagnosis of toxoplasmosis, the patient was evaluated by the infectious disease specialist, who initiated the treatment of toxoplasmosis and performed a serologic test which revealed an HIV infection.
Despite therapy, due to HIV-related immunosuppression, the patient suffered multiorgan failure and died.
DISCUSSION
The clinical presentation of cerebral toxoplasmosis is notably heterogeneous, nonspecific, and overlapping. Cerebral toxoplasmosis usually presents neurological subacute manifestations. Nevertheless, the disease may progress rapidly with fatal diffuse encephalitis or ventriculitis, with or without focal brain lesions in imaging studies.[
Clinical signs suggesting encephalitis in an immunocompromised patient should raise suspicions of toxoplasma. In contrast, in an immunocompetent patient or in a patient who is unaware of immunosuppression, clinical presentation alone is not sufficient for proper diagnosis and may be confused with other types of meningoencephalitis or brain tumor.[
In typical cases of toxoplasmosis, both CT and MRI reveal varying numbers of nodular or ring-enhancing lesions with surrounding edema. The lesions are scattered throughout the brain, and probably, those adjacent to the subarachnoid space causes the meningitis component in meningoencephalitis.[
The intensity of enhancement varies and can be weaker in patients with advanced HIV disease and reduced immune response. Lesions occur more frequently at the gray-white matter interface and in the basal ganglia and thalamus. Although signal characteristics vary, the lesions are often hyperintense on T2-weighted sequences. In our case, contrary to the most common cases reported in the literature,[
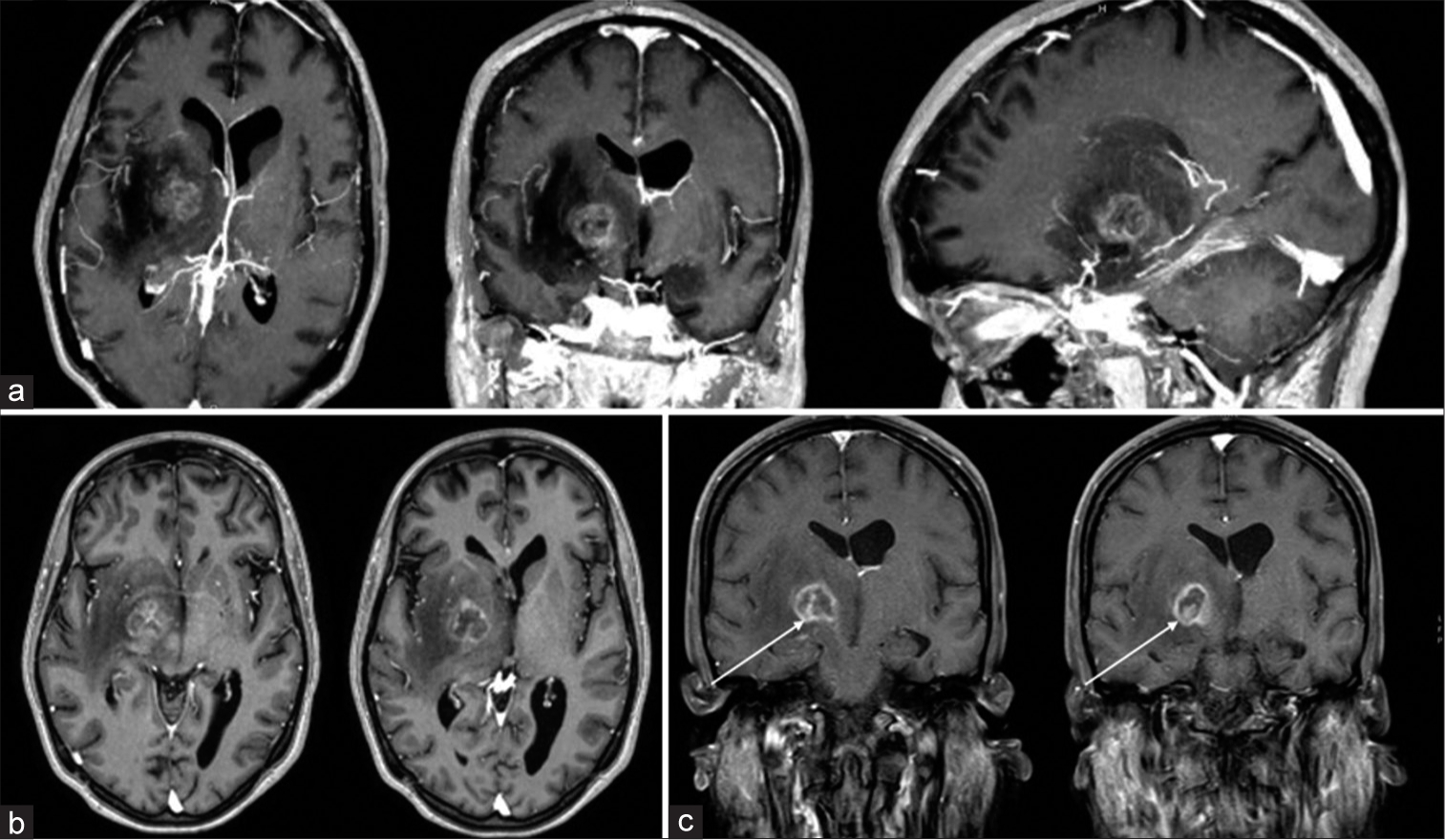
One of the most frequently described findings of CNS toxoplasmosis is the postcontrast T1 “eccentric target sign” consisting of three alternating zones: an innermost eccentric enhancing core, an intermediate hypointense zone, and an outer peripheral hyperintense enhancing rim.[
In our case, there was an enhanced intranodular component that, however, did not show the typical features of the abovementioned “eccentric target sign” [
In our patient, the intranodular component showed homogeneous enhancement without the intermediate hypointense component, as in certain neoplasms with intracystic nodule. A more specific imaging pattern is the more recently described “concentric target sign” on T2-weighted MR imaging. This focal lesion presents an alternation of concentric layers of hypo- and T2-weighted hyperintensities.[
In our patient, the radiological signs were quite atypical. In the postcontrast T1 sequences [
Furthermore, the patient was unaware that she was infected with HIV and the remaining clinical and radiological features were indicative of a brain tumor, which delayed diagnosis and treatment. Unfortunately, after surgery, the patient developed severe HIV-related immune suppression that resulted in sepsis and multiple infections leading to death.
Empirical treatment is often initiated in patients with characteristic enhancing mass lesions, due to the high incidence of toxoplasma encephalitis in HIV-infected patients and the difficulty in making a noninvasive diagnosis.
Toxoplasmosis often responds rapidly to therapy with pyrimethamine and sulfadiazine. Radiologic improvement is usually apparent within 2–4 weeks, but, in rare cases, patients may take up to 6 months to respond.[
When diagnostic uncertainty persists or in patients with radiological lesions suggestive of toxoplasmosis and lack of risk factors as HIV/AIDS and immunosuppression, a biopsy is needed to confirm the diagnosis.
CONCLUSION
MRI findings of single or multiple brain lesions suggesting cerebral toxoplasmosis should lead to screening for immunosuppression, when unknown, and early introduction of specific medical therapy, also for ex adiuvantibus purposes. A biopsy may be needed for differential diagnosis with tumors.
Although cerebral toxoplasmosis is rather rare, this diagnosis cannot be excluded, since an early diagnosis can significantly improve the prognosis of these patients.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
References
1. Collazos J. Opportunistic infections of the CNS in patients with AIDS: Diagnosis and management. CNS Drugs. 2003. 17: 869-87
2. Kastrup O, Wanke I, Maschke M. Neuroimaging of infections of the central nervous system. Semin Neurol. 2008. 28: 511-22
3. Mazzucchi E, La Rocca G, Hiepe P, Pignotti F, Galieri G, Policicchio D. Intraoperative integration of multimodal imaging to improve neuronavigation: A technical note. World Neurosurg. 2022. 164: 330-40
4. Nicholas MK, Collins J, Lukas RV, Winn HR, editors. Acquired immunodeficiency syndrom. Youmans and Winn Neurological Surgery. Philadelphia, PA: Elsevier; 2017. p. 223-41 Ch. 41
5. Policicchio D, Boccaletti R, Casu G, Dipellegrini G, Doda A, Muggianu G. Utility and feasibility of a low-cost system to simulate clipping strategy for cerebral aneurysms using three-dimensional computed tomography angiography with virtual craniotomy. World Neurosurg. 2022. 168: 155-64
6. Policicchio D, Dipellegrini G, Muggianu G, Pintus A, Sgaramella E, Santonio FV. Flexible fiber CO2 laser in microsurgical treatment of intraventricular tumors: Usefulness and limitations. World Neurosurg. 2019. 122: e427-35
7. Policicchio D, Ticca S, Dipellegrini G, Doda A, Muggianu G, Boccaletti R. Multimodal surgical management of cerebral lesions in motor-eloquent areas combining intraoperative 3D ultrasound with neurophysiological mapping. J Neurol Surg A Cent Eur Neurosurg. 2021. 82: 344-56
8. Roche AD, Rowley D, Brett FM, Looby S. Concentric and eccentric target MRI signs in a case of HIV-associated cerebral toxoplasmosis. Case Rep Neurol Med. 2018. 2018: 9876514
9. Smith AB, Smirniotopoulos JG, Rushing EJ. From the archives of the AFIP: Central nervous system infections associated with human immunodeficiency virus infection: Radiologicpathologic correlation. Radiographics. 2008. 28: 2033-58
10. Vidal JE. HIV-related cerebral toxoplasmosis revisited: Current concepts and controversies of an old disease. J Int Assoc Provid AIDS Care. 2019. 18: 2325958219867315