- College of Medicine, The University of Arizona College of Medicine - Tucson, United States
- Department of Neurosurgery, The University of Arizona College of Medicine - Tucson, Tucson, United States.
Correspondence Address:
Martin Weinand, Department of Neurosurgery, The University of Arizona College of Medicine - Tucson, Tucson, United States.
DOI:10.25259/SNI_901_2022
Copyright: © 2022 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: James Christopher Mamaril-Davis1, Neil Joshi2, Ryan Palsma2, Pedro Aguilar-Salinas2, Christina M. Walter2, Shamam Hashim2, Martin Weinand2. Spinal cord stimulation for genitofemoral neuropathy: A case report and review of the literature. 18-Nov-2022;13:533
How to cite this URL: James Christopher Mamaril-Davis1, Neil Joshi2, Ryan Palsma2, Pedro Aguilar-Salinas2, Christina M. Walter2, Shamam Hashim2, Martin Weinand2. Spinal cord stimulation for genitofemoral neuropathy: A case report and review of the literature. 18-Nov-2022;13:533. Available from: https://surgicalneurologyint.com/surgicalint-articles/12013/
Abstract
Background: Chronic testicular pain due to genitofemoral neuropathy often becomes refractory to conservative medical therapy. Neurostimulation is a potentially useful treatment option, should the neuropathic pain remain refractory to more invasive procedures such as orchiectomy. We provide a case report of spinal cord stimulation (SCS) for successful treatment of genitofemoral neuropathy and have also reviewed the literature to find similar cases which required a similar treatment paradigm.
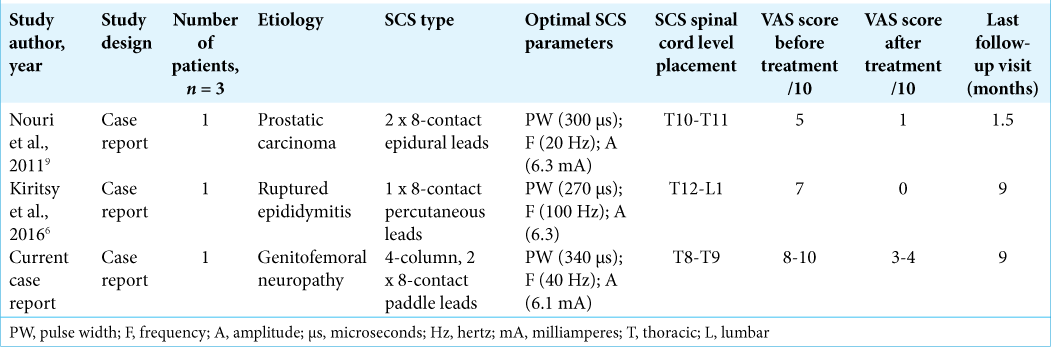
Case Description: A 42-year-old male underwent SCS for refractory testicular and groin pain. SCS through a four-column, 2 × 8 contact neurostimulator paddle lead, was implanted in the mid-thoracic-9 (T9) vertebral level, providing > 50% testicular pain relief with a decrease in visual analog scale scores from 8–10/10 to 3–4/10. The patient required one adjustment to the stimulation parameters at the time of the 6 weeks follow-up visit due to over-stimulation. He then continued to experience >50% resolution in pain 9 months later. A review of the literature yielded only two similar cases that successfully utilized SCS for treatment of chronic testicular pain.
Conclusion: SCS should be considered as a possible treatment option for patients with chronic testicular pain localized to the genitofemoral nerve distribution.
Keywords: Genitofemoral, Orchalgia, Spinal cord stimulation, Testicular pain
INTRODUCTION
Chronic testicular pain, or orchalgia, although typically idiopathic, may also be associated with genitofemoral neuropathy.[
CASE REPORT
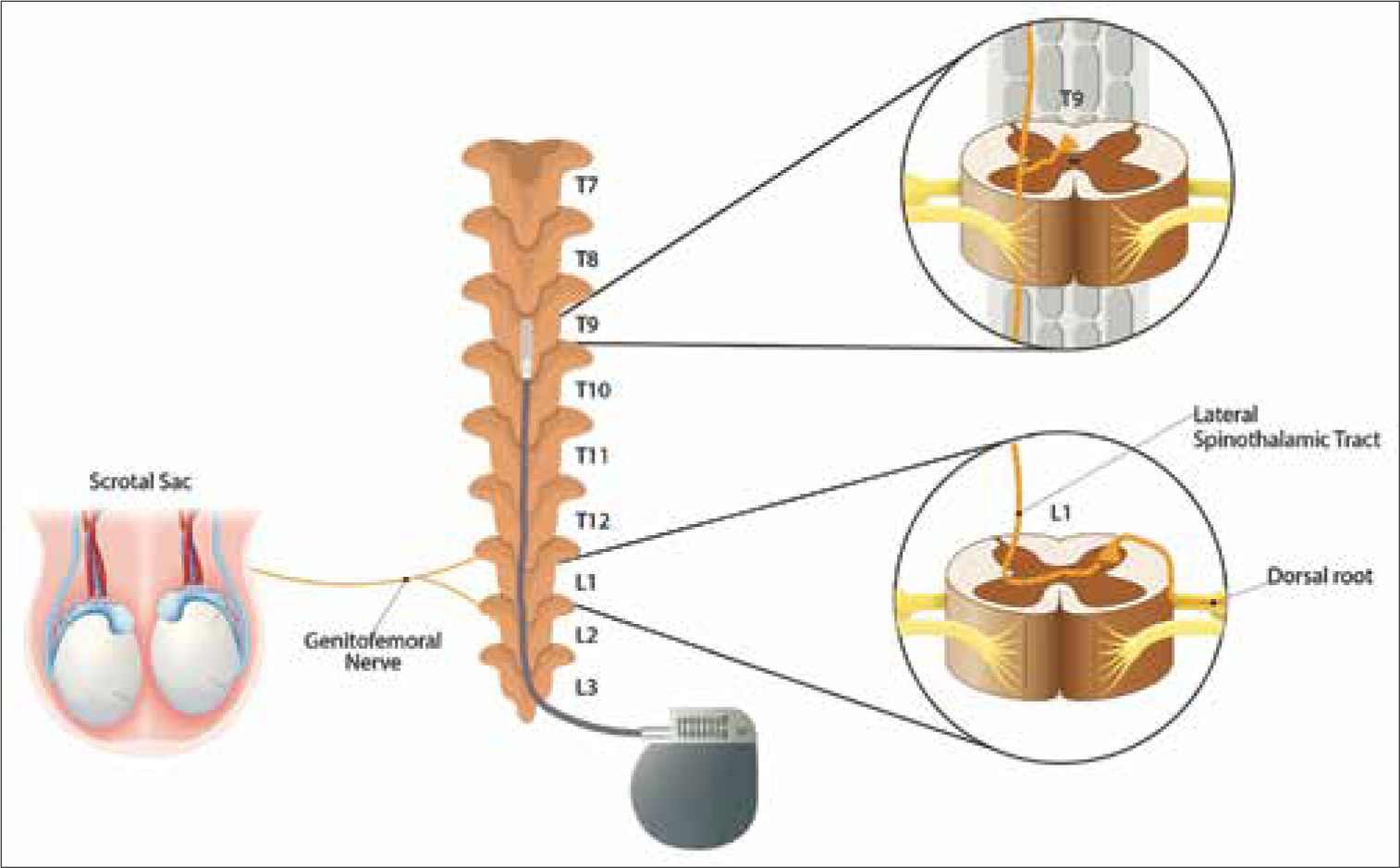
A 42-year-old male presented with a 3.5-year history of chronic right groin and testicular pain (refractory genitofemoral neuropathy characterized by constant and dull burning sensations) after a gunshot injury to the groin. The pain failed to resolve with several anti-inflammatory and pain medications: ibuprofen, gabapentin, ketorolac, and opioids (e.g., hydromorphone, oxycodone, morphine, and fentanyl). The patient initially benefitted from a genitofemoral nerve block, a right ilioinguinal nerve block, and a right spermatic cord block. As his pain then became refractory to these treatment modalities, and his MR studies of the thoracic and lumbar spine were negative for causative pathology, he underwent a trial of genitofemoral SCS. His visual analog scale (VAS) scores before the SCS trial ranged from 8 to 10/10.
SCS for genitofemoral neuropathy
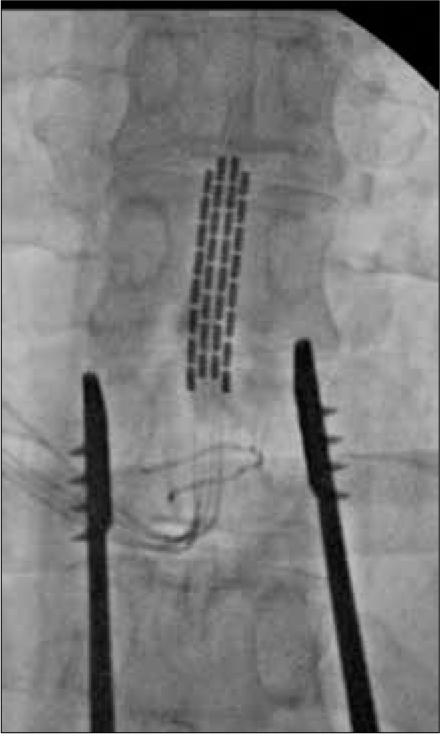
For the SCS trial, two percutaneous 16-contact, midline cylindrical leads were temporarily implanted, and positional adjustments were made until the patient reported adequate capture of the typical painful areas. The optimal level of stimulation was at the mid-T9 level. The SCS trial lasted for 1 week, at which point he reported >50% reduction in both right groin and testicular pain, with his VAS score decreasing to 3/10. He also noted improvement in sleep, mobility, and functional activity. He underwent permanent SCS placement with a 4-column, 2 × 8-contact epidural paddle lead (at mid-T9 through a laminotomy at T10) [
Postoperative course following SCS implantation
Six weeks after the permanent SCS was implanted, the patient continued to report >50% pain relief, VAS scores remained stable at 3/10, and he was able to return to work. At the 6 week follow-up visit, the patient reported over-stimulation symptoms; therefore, the stimulation parameters were adjusted to a pulse width of 340 μs, frequency of 40 Hz, and current of 6.1 mA. At the 9 months follow-up, the patient continued to report >50% pain relief with VAS scores in the 3–4/10 range.
DISCUSSION
The first-line treatment for chronic testicular pain is conservative medical therapy that often proves inadequate with success rates ranging from only 4.2% to 15.2%.[
Two prior cases of SCS successfully treating chronic testicular pain
A literature review including PubMed, Ovid MEDLINE, and Ovid EMBASE databases yielded just two prior reports of successful utilization of SCS for chronic testicular pain.[
Testicular pain as a complication of SCS
Huang et al. reported two cases of acute, neuropathic testicular and scrotal pain following a percutaneous SCS trial.[
Types of testicular pain
There are two proposed types of testicular pain: nociceptive and neuropathic.[
Role of SCS for neuropathic testicular pain
SCS is a relatively less invasive modality versus surgery for chronic orchalgia (e.g., SCS trial versus orchiectomy). However, perioperative complications occur in 0.7–38% of SCS cases and may be due to: device failure such as lead migration and over/under stimulation; biologic factors such as infection and seroma; and even complications of SCS placement itself.[
CONCLUSION
SCS is a valuable treatment modality for patients with chronic orchalgia, especially when the pain distribution can be adequately localized to the genitofemoral nerve region.
Declaration of patient consent
Institutional Review Board (IRB) permission obtained for the study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
References
1. Calixte N, Brahmbhatt J, Parekattil S. Chronic testicular and groin pain: Pathway to relief. Curr Urol Rep. 2017. 18: 83
2. Cameron T. Safety and efficacy of spinal cord stimulation for the treatment of chronic pain: A 20-year literature review. J Neurosurg. 2004. 100: 254-67
3. Chaudhari R, Sharma S, Khant S, Raval K. Microsurgical denervation of spermatic cord for chronic idiopathic orchialgia: Long-term results from an institutional experience. World J Mens Health. 2019. 37: 78-84
4. Huang M, Desai VR, Ho D, Simpson RK. Acute neuropathic orchalgia and scrotalgia after percutaneous spinal cord stimulator lead placement: Two cases with an unusual complication. Cureus. 2017. 9: e1003
5. Hunter JP, Ashby P. Segmental effects of epidural spinal cord stimulation in humans. J Physiol. 1994. 474: 407-19
6. Kiritsy MP, Siefferman JW. Spinal cord stimulation for intractable testicular pain: Case report and review of the literature. Neuromodulation. 2016. 19: 889-92
7. Leslie SW, Sajjad H, Siref LE, editors. Chronic Testicular Pain and Orchalgia. StatPearls. Treasure Island: StatPearls Publishing LLC; 2022. p.
8. Mekhail NA, Mathews M, Nageeb F, Guirguis M, Mekhail MN, Cheng J. Retrospective review of 707 cases of spinal cord stimulation: Indications and complications. Pain Pract. 2011. 11: 148-53
9. Nouri KH, Brish EL. Spinal cord stimulation for testicular pain. Pain Med. 2011. 12: 1435-8
10. Vallejo R, Bradley K, Kapural L. Spinal cord stimulation in chronic pain: Mode action. Spine (Phila Pa 1976). 2017. 42: S53-60