- Department of Neurosurgery, Pt. B.D. Sharma University of Health Sciences, Rohtak, Haryana, India
- Department of Radiodiagnosis, Pt. B.D. Sharma University of Health Sciences, Rohtak, Haryana, India
- Department of Anaesthesiology and Critical Care, Pt. B.D. Sharma University of Health Sciences, Rohtak, Haryana, India
Correspondence Address:
Ishwar Singh
Department of Neurosurgery, Pt. B.D. Sharma University of Health Sciences, Rohtak, Haryana, India
DOI:10.4103/2152-7806.166752
Copyright: © 2015 Singh I. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.How to cite this article: Singh I, Rohilla S, Kumar P, Sharma S. Spinal dorsal dermal sinus tract: An experience of 21 cases. Surg Neurol Int 07-Oct-2015;6:
How to cite this URL: Singh I, Rohilla S, Kumar P, Sharma S. Spinal dorsal dermal sinus tract: An experience of 21 cases. Surg Neurol Int 07-Oct-2015;6:. Available from: http://surgicalneurologyint.com/surgicalint_articles/spinal-dorsal-dermal-sinus-tract-an-experience-of-21-cases/
Abstract
Background:Spinal dorsal dermal sinus is a rare entity, which usually comes to clinical attention by cutaneous abnormalities, neurologic deficit, and/or infection. The present study was undertaken to know the clinical profile of these patients, to study associated anomalies and to assess the results of surgical intervention.
Methods:Medical records of 21 patients treated for spinal dorsal dermal sinus from September 2007 to December 2013 were reviewed.
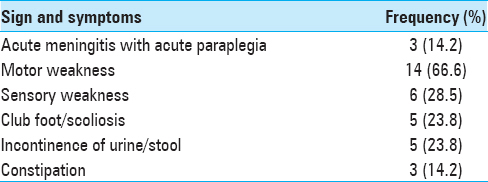
Results:We had 21 patients with male: female ratio of 13:8. Only 2 patients were below 1-year of age, and most cases (15) were between 2 and 15 years (mean age = 8.2 years). Lumbar region (11 cases) was most frequently involved, followed by thoracic (4 cases), lumbosacral, and cervical region in 3 patients each. All of our patients presented with neurological deficits. Three patients were admitted with acute meningitis with acute onset paraplegia and had intraspinal abscess. The motor, sensory, and autonomic deficits were seen in 14, 6, and 8 patients, respectively. Scoliosis and congenital talipes equinovarus were the common associated anomalies. All patients underwent surgical exploration and repair of dysraphic state and excision of the sinus. Overall, 20 patients improved or neurological status stabilized and only 1 patient deteriorated. Postoperative wound infection was seen in 2 cases.
Conclusions:All patients with spinal dorsal dermal sinuses should be offered aggressive surgical treatment in the form of total excision of sinus tract and correction of spinal malformation, as soon as diagnosed.
Keywords: Complication, dermal sinus, dysraphism, presentation, spine
INTRODUCTION
Spinal dorsal dermal sinus tract (DST) is a rare congenital dysraphism that occurs in approximately one in every 2500 live births.[
PATIENTS AND METHODS
This is a retrospective study conducted in Pt. B.D. Sharma University of Health Sciences, Rohtak from September 2007 to December 2013. Medical records of all patients treated for spinal DST were reviewed. Information regarding patients’ demographic variables, type of presentation, symptoms, physical examination, radiological and surgical findings, and histopathological evaluation were collected. Magnetic resonance imaging (MRI) was the investigation of choice and was performed in all cases. MRI revealed the relationship of the dermal sinus to the dural sac and also gave information regarding associated abnormalities in the cord like dysraphic state of spine or inclusion tumor.
Surgical intervention
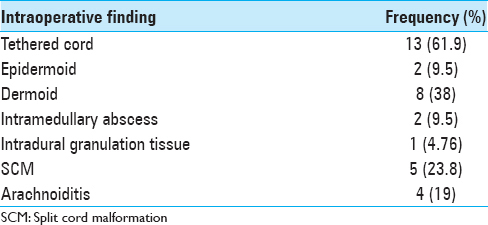
The aim of surgery was to excise the sinus tract completely and to correct the dysraphic state in the same sitting. Surgery was performed in all cases through midline incision with encircling the sinus. DST was followed through the subcutaneous tissue and muscle layer sinus tract was traced until its end and excised completely. The course of DST was invariably rostral through the incompletely formed lamina or underneath the normal lamina. After doing the laminectomy, dura was opened in all cases irrespective of end of DST. In cases where DST was intradural, part of dura encircling the DST was excised. Intraspinal pathologies like SCM were dealt accordingly that is, dermoid and epidermoid were decompressed or excised; myelocele and lipomeningomyelocele were repaired; drainage of abscess in intramedullary abscess, removal of arachnoid adhesion in arachnoiditis and detethering of the cord was done in case of tethered cord. Those patients presenting with infectious complications were managed with appropriate antibiotics and then after recovery surgery for resection of DST, and correction of associated anomalies was performed. Postoperative follow-up ranged from 6 months to 5 years (mean - 2.8 years).
RESULTS
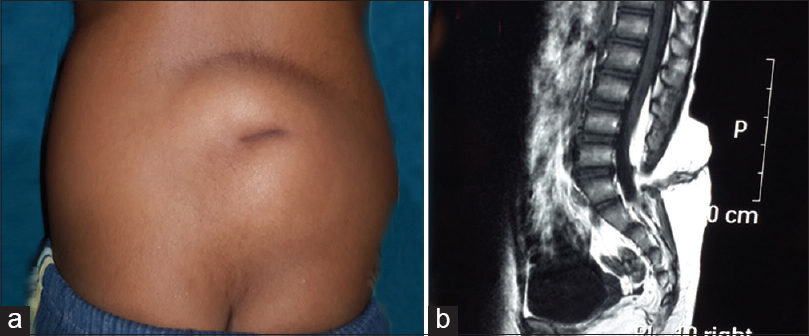
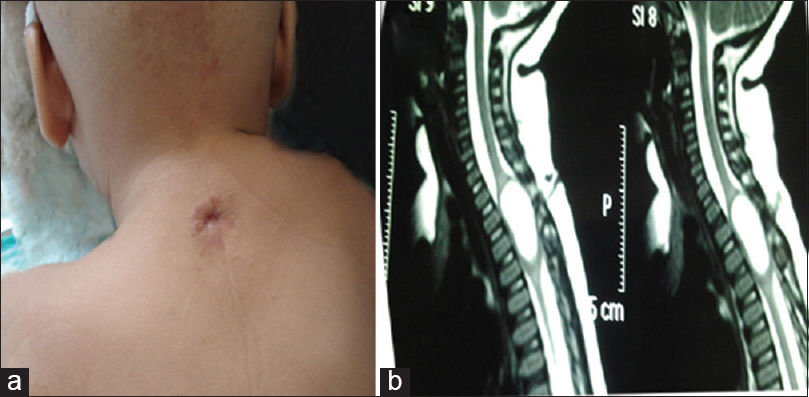
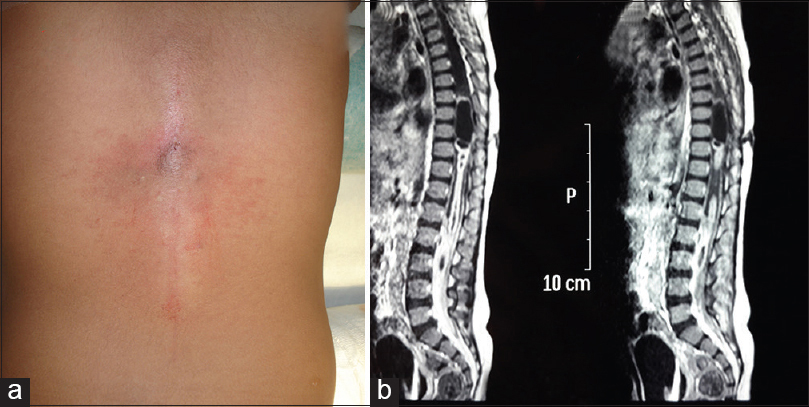
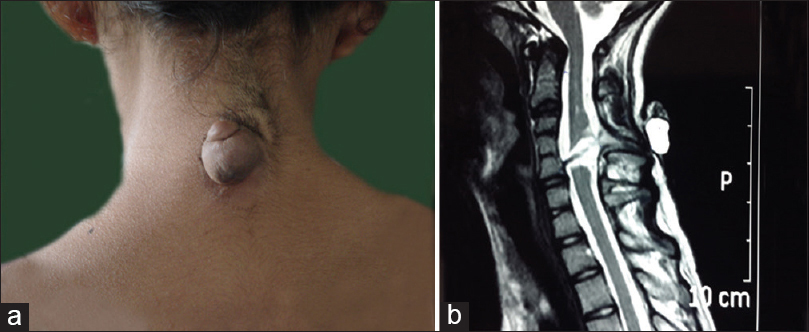
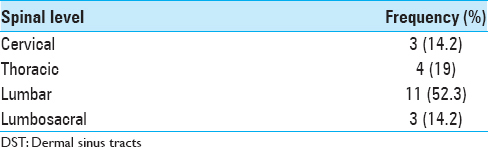
Records of total 21 patients were analyzed, of which 13 were male, and 8 were female. Patients’ age on admission ranged from 9 months to 15 years (mean - 8.2 years). Every patient underwent a detailed neurological examination and a complete radiological workup to delineate any underlying/associated spinal abnormalities. DST was located most frequently in lumbar region (11 cases) [
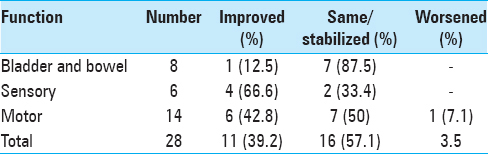
Overall, 11 patients showed neurological improvement, 9 patients stabilized neurologically while 1 patient deteriorated [
DISCUSSION
A spinal DST consists of a tract lined by stratified squamous epithelium found on or near the midline and is thought to result from the abnormal adhesions (or incomplete disjunction) between the neuroectoderm (destined to form the neural tube) and the cutaneous ectoderm.[
Dermal sinuses should be distinguished from the more common coccygeal pits. Dermal sinuses are located above the intergluteal cleft and have a cephalically oriented course and are often associated with other pathologies. On the contrary, coccygeal dimples are usually simple blind sinuses with no associated cutaneous abnormalities that lie within intergluteal cleft a few millimeters cranial to the tip of coccyx. They are oriented caudally or straight and are not associated with other intradural pathologies and thus do not warrant further evaluation.[
Dermal sinuses provide a portal of entry for bacterial agents into the intraspinal compartments that can cause meningitis or abscess formation that may be extradural, subdural, and intramedullary or infection of associated tumor. Also, aseptic meningitis can occur by spillage of inclusion tumor contents or other dermal elements into the cerebrospinal fluid.[
It has been said that nearly all children with spinal DSTs have intact neurological function at birth.[
Spinal dermal sinuses may be accompanied by other forms of spinal dysraphism such as lipomyelomeningocele and myelomeningocele, reflecting a possible common ontogenic pathway.[
Approximately, half of all dermal sinuses are associated with dermoid or epidermoid tumor, usually at the termination of these tracts, but they may be located anywhere between the skin and the neural tube.[
Postoperative complications were few and easy to manage. Our results indicate that once a patient developed bowel/bladder incontinence, there was about 12.5% chance of improvement in deficit while in patients with sensory or motor deficits; the chance of improvement was 66.6% and 42.8%, respectively [
Since none of imaging modalities can accurately show intraspinal details, all dermal sinuses above the sacrococcygeal region should be explored operatively regardless of neuroimaging findings.[
CONCLUSION
Spinal DST is an innocuous-appearing spinal dysraphism that may contribute to devastating morbidities if not timely addressed. Although there has been increased awareness about the impotence of dorsal midline cutaneous finding among primary health care physician, there still much more to be done especially in developing country. All patients with spinal DST should be offered aggressive surgical treatment in the form of total excision of sinus tract and correction of spinal malformation, as soon as diagnosed since chances of preserving and/or improving neural function are high (95%).
References
1. Ackerman LL, Menezes AH, Follett KA. Cervical and thoracic dermal sinus tracts. A case series and review of the literature. Pediatr Neurosurg. 2002. 37: 137-47
2. Ackerman LL, Menezes AH. Spinal congenital dermal sinuses: A 30-year experience. Pediatrics. 2003. 112: 641-7
3. Elton S, Oakes WJ. Dermal sinus tracts of the spine. Neurosurg Focus. 2001. 10: e4-
4. French BN. The embryology of spinal dysraphism. Clin Neurosurg. 1983. 30: 295-340
5. Gupta DK, Shastank RR, Mahapatra AK. An unusual presentation of lumbosacral dermal sinus with CSF leak and meningitis. A case report and review of the literature. Pediatr Neurosurg. 2005. 41: 98-101
6. Jindal A, Mahapatra AK. Spinal congenital dermal sinus: An experience of 23 cases over 7 years. Neurol India. 2001. 49: 243-6
7. Kaufman BA. Neural tube defects. Pediatr Clin North Am. 2004. 51: 389-419
8. Lew SM, Kothbauer KF. Tethered cord syndrome: An updated review. Pediatr Neurosurg. 2007. 43: 236-48
9. Martínez-Lage JF, Pérez-Espejo MA, Tortosa JG, Ros de San Pedro J, Ruiz-Espejo AM. Hydrocephalus in intraspinal dermoids and dermal sinuses: The spectrum of an uncommon association in children. Childs Nerv Syst. 2006. 22: 698-703
10. McComb JG, Chen TC, Tindall GT, Cooper PR, Barrow DL.editors. Closed neural tube defects. The Practice of Neurosurgery. Baltimore: William and Wilkins; 1996. p. 2753-78
11. McLone DG, Naidich TP, McLaurin RL, Venes JL.editors. The tethered spinal cord. Pediatric Neurosurgery. Philadelphia: WB Saunders; 1989. p. 71-96
12. Mete M, Umur AS, Duransoy YK, Barutçuoglu M, Umur N, Gurgen SG. Congenital dermal sinus tract of the spine: Experience of 16 patients. J Child Neurol. 2014. 29: 1277-82
13. Morandi X, Mercier P, Fournier HD, Brassier G. Dermal sinus and intramedullary spinal cord abscess. Report of two cases and review of the literature. Childs Nerv Syst. 1999. 15: 202-6
14. Naidich TP, Harwood-Nash DC, McLone DG. Radiology of spinal dysraphism. Clin Neurosurg. 1983. 30: 341-65
15. Pang D, Dias MS, Ahab-Barmada M. Split cord malformation: Part I: A unified theory of embryogenesis for double spinal cord malformations. Neurosurgery. 1992. 31: 451-80
16. Pang D. Split cord malformation: Part II: Clinical syndrome. Neurosurgery. 1992. 31: 481-500
17. Radmanesh F, Nejat F, El Khashab M. Dermal sinus tract of the spine. Childs Nerv Syst. 2010. 26: 349-57
18. Ramnarayan R, Dominic A, Alapatt J, Buxton N. Congenital spinal dermal sinuses: Poor awareness leads to delayed treatment. Childs Nerv Syst. 2006. 22: 1220-4
19. Tubbs RS, Frykman PK, Harmon CM, Oakes WJ, Wellons JC. An unusual sequelae of an infected persistent dermal sinus tract. Childs Nerv Syst. 2007. 23: 569-71