- Department of Neurosurgery, Faculty of Medicine, Kasr Al-Ainy Medical College, Cairo, Egypt.
- Department of Neurosurgery, Al-Ahly Bank Hospital, Egypt.
Correspondence Address:
Hieder Al-Shami
Department of Neurosurgery, Al-Ahly Bank Hospital, Egypt.
DOI:10.25259/SNI_804_2020
Copyright: © 2021 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Ahmed Mohamed Alselisly1, Hieder Al-Shami2, Ahmed Mohamed Salah1. Surgical management of cerebral venous sinus thrombosis: Case series and literature review. 30-Mar-2021;12:133
How to cite this URL: Ahmed Mohamed Alselisly1, Hieder Al-Shami2, Ahmed Mohamed Salah1. Surgical management of cerebral venous sinus thrombosis: Case series and literature review. 30-Mar-2021;12:133. Available from: https://surgicalneurologyint.com/?post_type=surgicalint_articles&p=10676
Abstract
Background: Cerebral venous sinus thrombosis (CVST) is not a common type of stroke (5%) but still hazardous to be misdiagnosed or mistreated. Aggressive medical treatment is usually failed to hinder increase intracranial tension. Therefore, decompressive craniectomy (DC) is the final measure to mitigate the deleterious effect of supratentorial herniation. The purpose of the study is to illustrate our experience with the surgical treatment of CVST and reviewing the previous works of literature.
Methods: Forty-two patients were admitted to Kasr Al-Ainy University Hospital from June 2019 to March 2020. The admission was either to the neurology department or intensive care unit or neurosurgery department. Every patient who was diagnosed with CVST received an emergency neurosurgery consultation. Seven patients were operated on with DC according to the criteria mentioned above. Therapeutic heparin was given in addition to intracranial pressure lowering measures.
Results: The mean and standard deviation of the age was (25.14 ± 10.1) years. There were five females (71.45%) in our series. The mean and standard deviation of clinical manifestations are (8.5 ± 7.77) weeks with range (3– 14 weeks). Most of the cases were presented by a decreased level of consciousness (6/7) and anisocoria (6/7), followed by fits (3/7). Four cases out of seven had the previous history of oral contraceptive administration.
Conclusion: DC provides an urgent last arm for intractable increased intracranial tension. Patients with CVST need urgent consultation for neurosurgical intervention.
Keywords: Cerebral venous sinus thrombosis, Decompressive craniectomy, Hemorrhagic infarction
INTRODUCTION
The cerebral venous sinus thrombosis (CVST) is considered an ischemic episode due to blockage of main cerebral drainage of brain tissue with a resultant marvelous acute increment of intracranial pressure (ICP).[
The pathophysiology of CVST can involve any territory of the venous system but commonly found at sinus-vein junction.[
The clinical and radiological signs for which DC is indicated included: third cranial nerve palsy, deterioration of conscious level, uncal herniation, midline shift ≥5 mm, ICP over 20 cm H2O, and hypodensity of posterior cerebral artery territory.[
In this article, we are illustrating our experience with the surgical treatment of CVST and reviewing the previous works of literature.
PATIENTS AND METHODS
Study sample
It is retrospective study including 42 patients were admitted to Kasr Al-Ainy University Hospital from June 2019 to March 2020. The admission was either to the neurology department or intensive care unit (ICU) or neurosurgery department. Every patient who was diagnosed with CVST received an emergency neurosurgery consultation. Seven patients were operated upon with DC according to the criteria mentioned above after taking the consent from relatives. Therapeutic heparin was given in addition to ICP lowering measures. An ethical approval was retrieved from IRB of our college to start gathering information.
Perioperative treatment
For surgical patients, therapeutic heparin was discontinued 12 h before surgery and re-admitted again postsurgery with a prophylactic dose every 24 h. Prophylactic anticoagulant was given for more than a weak (11.48 ± 5.5 days).
DC
DC is simply bone excision and duraplasty. It acts by permitting space for edematous tissue to expand beyond intracranial anatomy with the creation of a closed space or room. It counteracts three models of herniation. The bone flap should include frontal, parietal, and temporal bones. The dural opening should guarantee maximal decompression. The dural edges are left with no approximated edges with the closure of overlying skin flaps only. A routine computed tomography (CT) scan should be obtained within 24 h of surgery. Re-implantation of the bone flap should not be discussed before 6 months.
Outcome analysis
Assessment of outcome included Glascow coma scale (GCS), motor deficit, and modified Rankin scale [
Statistical analysis
Data retrieved were processed using the Statistical Package for the Social Sciences (SPSS®) program version 25. Data of numerical values were compared using Chi-square t-tests while the categorical comparisons were tested by Fisher exact test. P = 0.05 was regarded as significant.
RESULTS
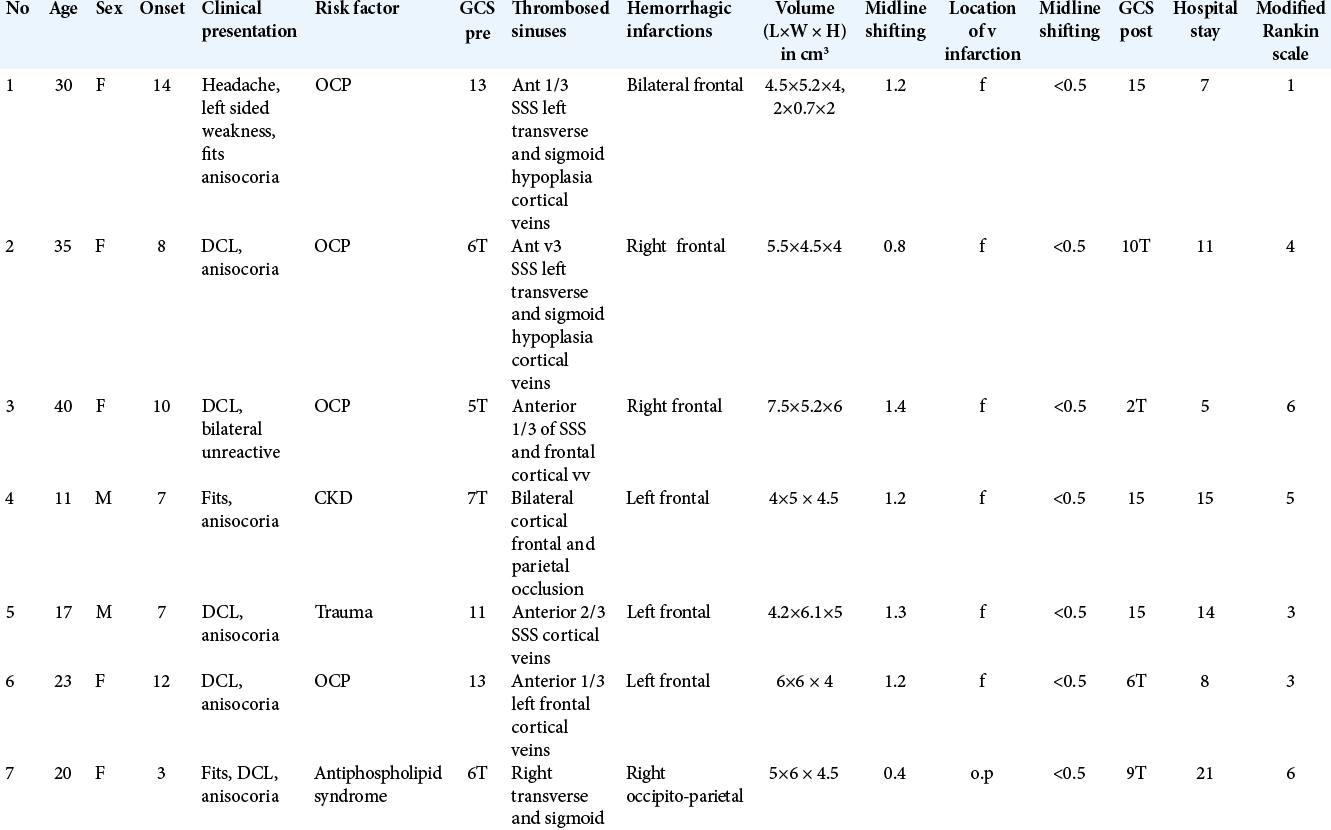
All patients’ criteria are illustrated in [
CASE PRESENATION
Case #1
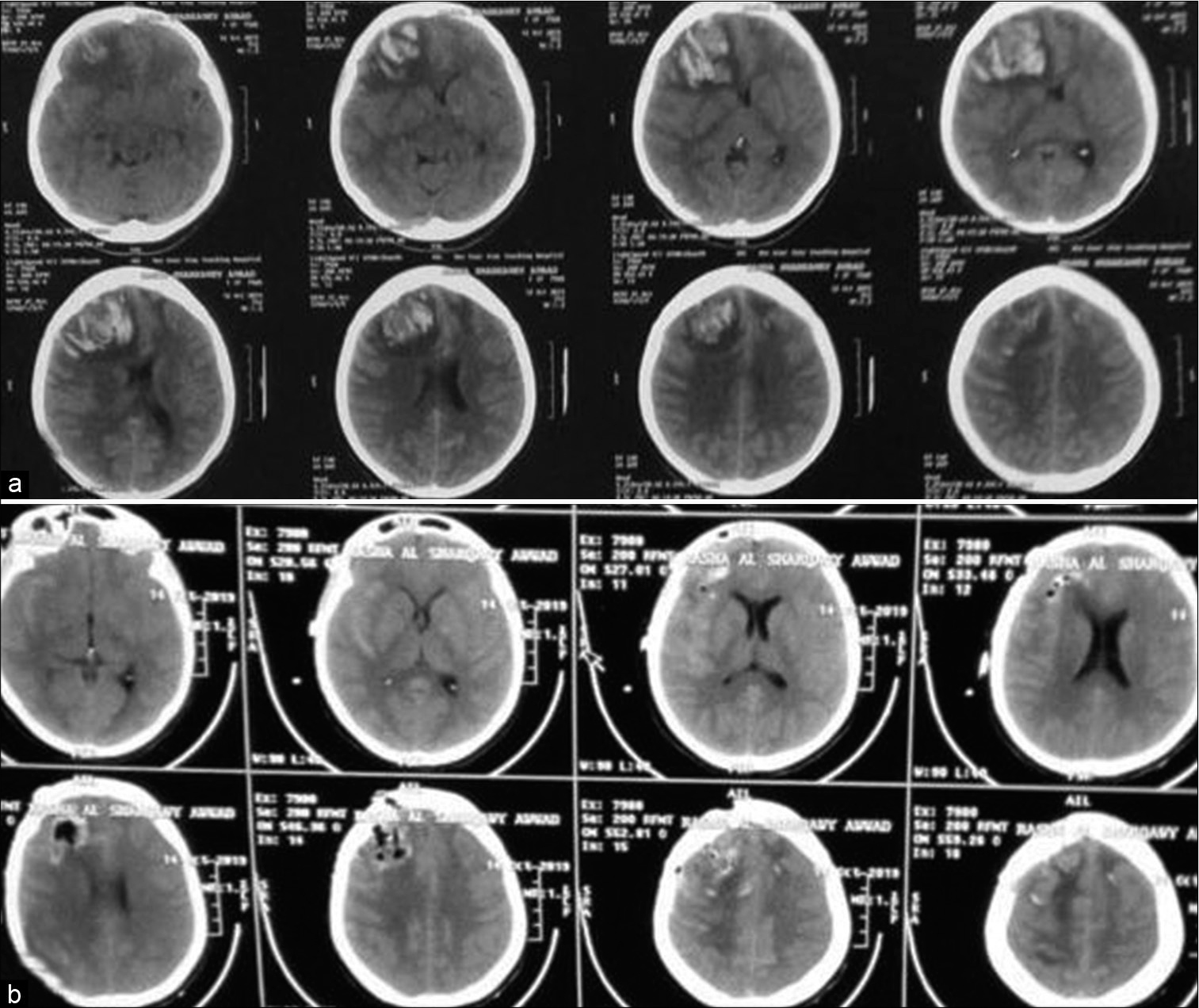
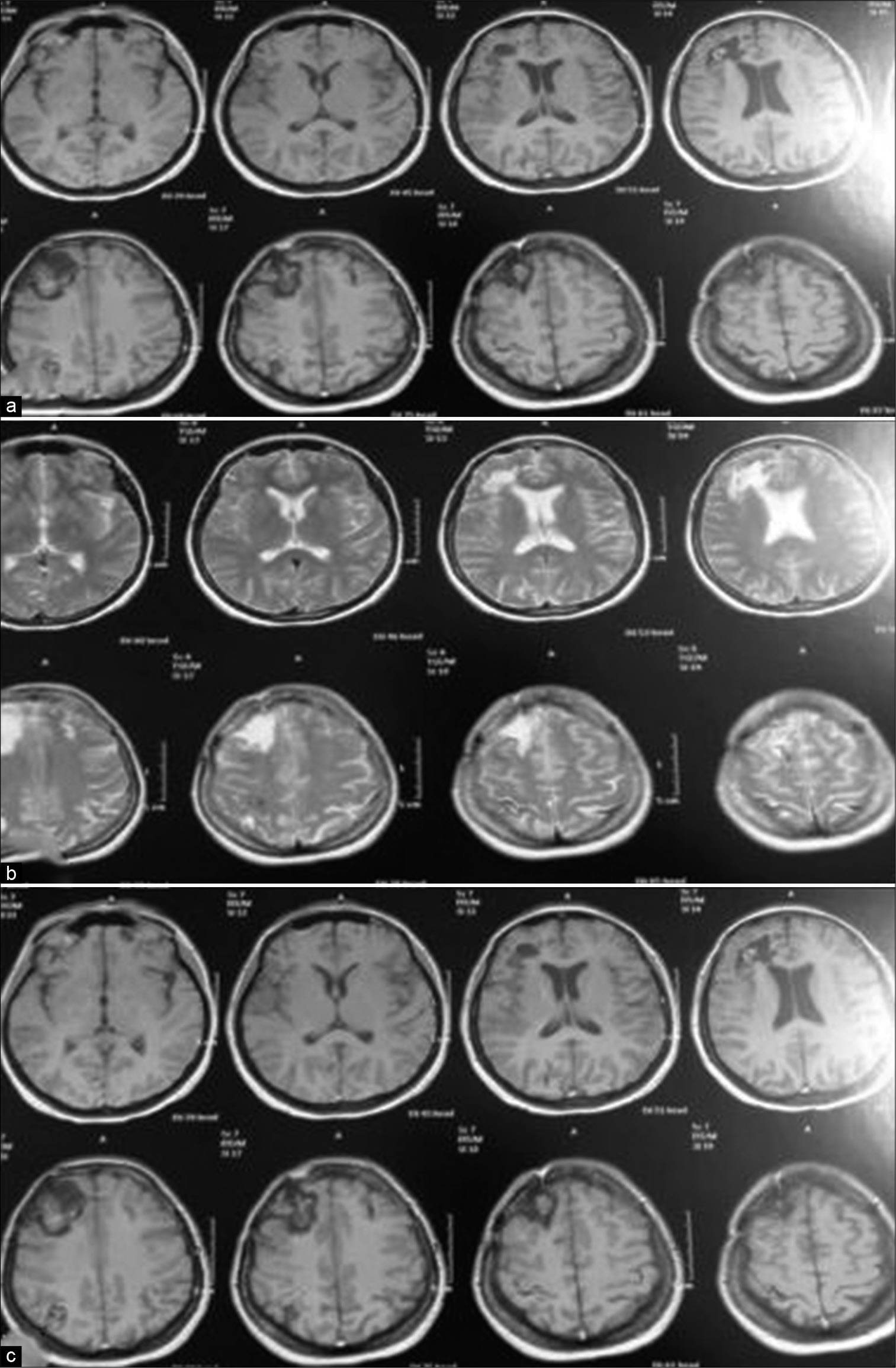
A 30-year-old female with a history of oral contraceptives administration for long time, experienced an episode of headache 2 weeks before ictus, presented to ER with a slight confusion and left-sided weakness grade 4/5. She underwent CT brain, magnetic resonance imaging (MRI), and magnetic resonance venography (MRV). CT findings were of right frontal intracerebral hemorrhage with midline shift [
Figure 1:
Case 1, noncontrast computed tomography (CT) brain. (a) preoperative CT showed right frontal intracerebral hemorrhage with compression of ipsilateral frontal horn and midline shift. (b) postoperative CT brain. Decompression of the mass effect exerted by the hematoma with pneumocephalus in the bed of surgical site.
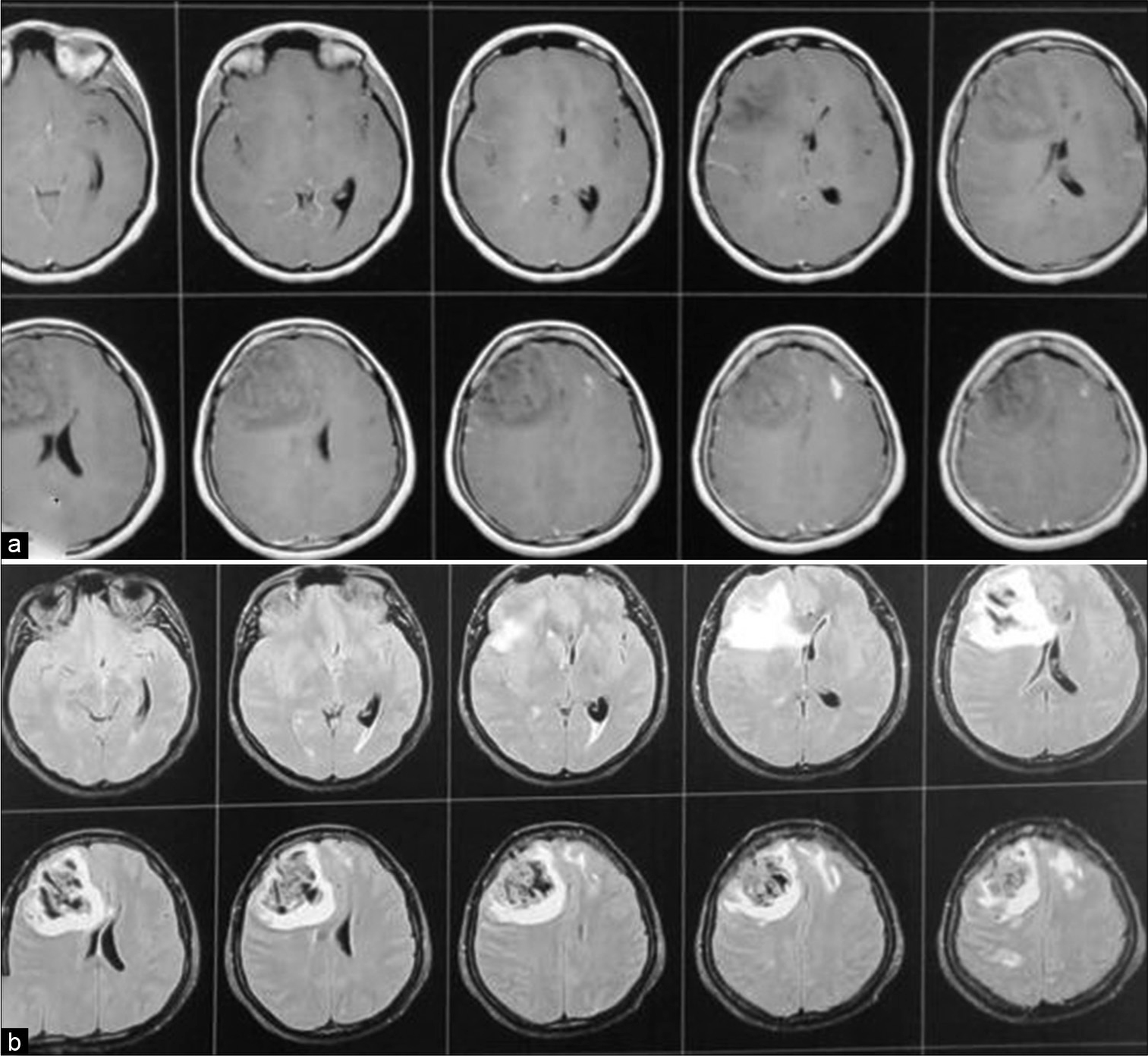
Figure 2:
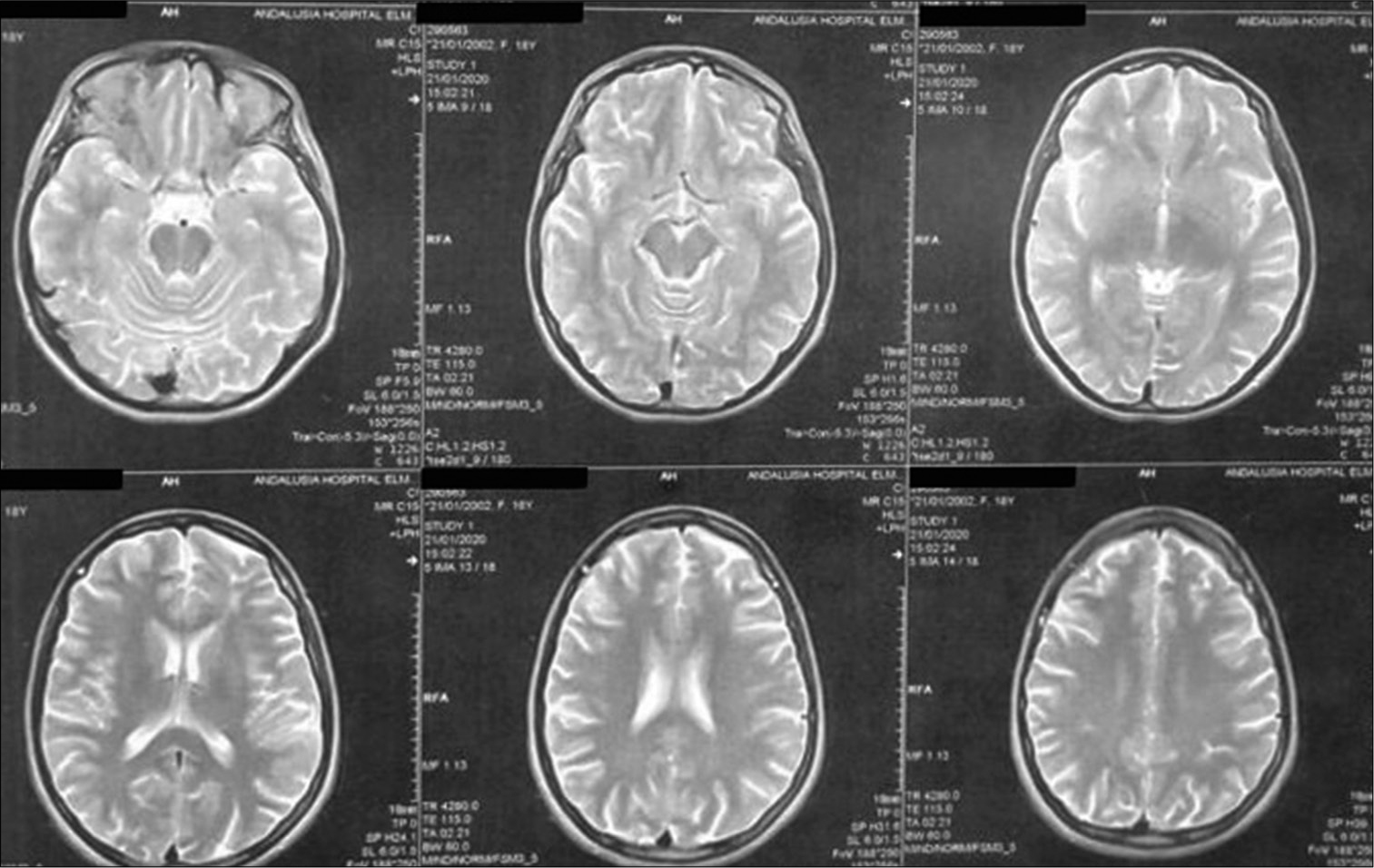
Case 1, (a) T1 WI with contrast showed right frontal intracerebral hemorrhage with surrounding edema, obliteration of ipsilateral frontal horn and displacement of contralateral horn with midline shift. (b) FLAIR image. The marked hypointensity of the ICH is most likely caused by the T2 shortening effects of intracellular methaemoglobin present abundantly in the clot at this stage.
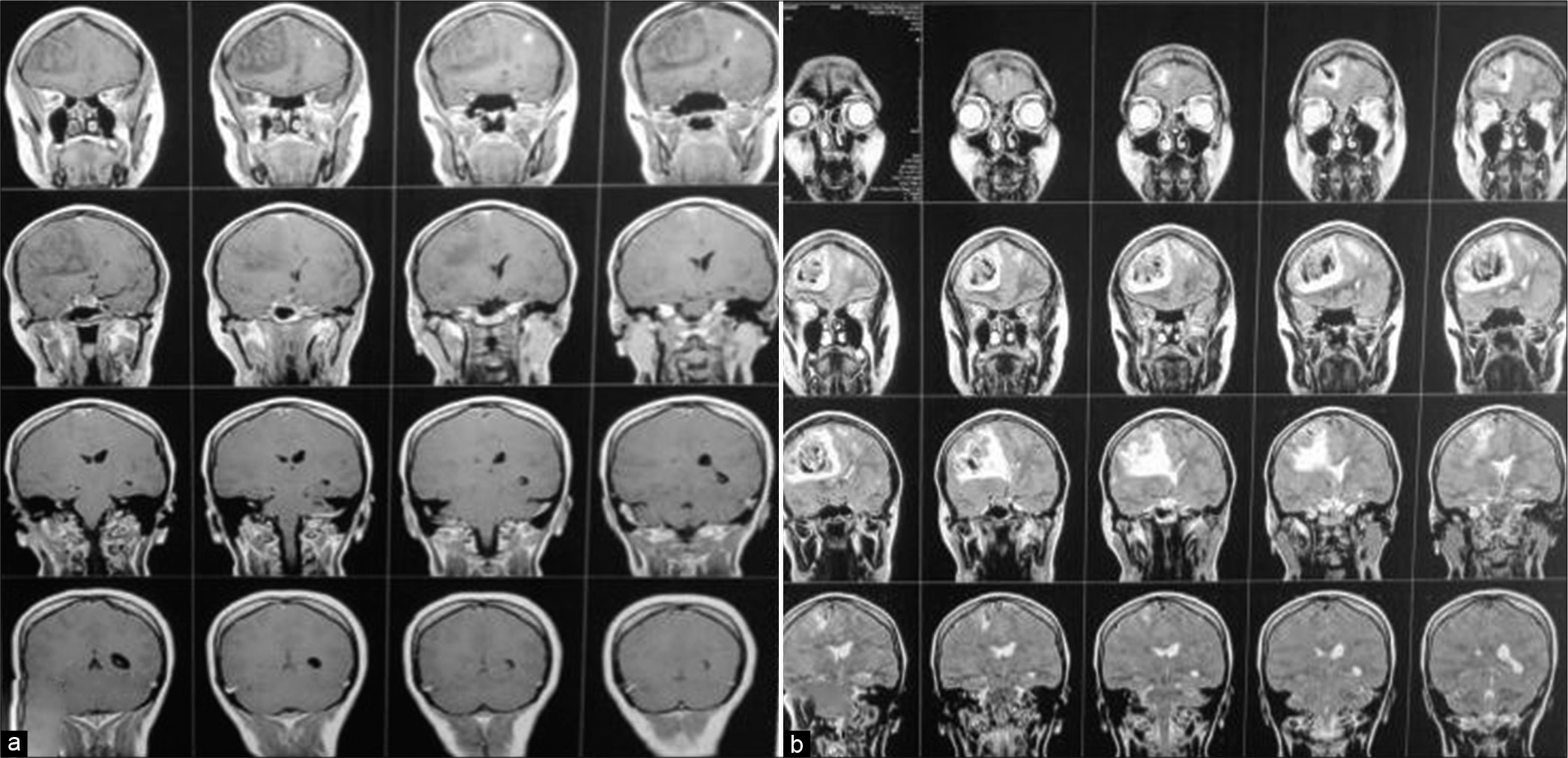
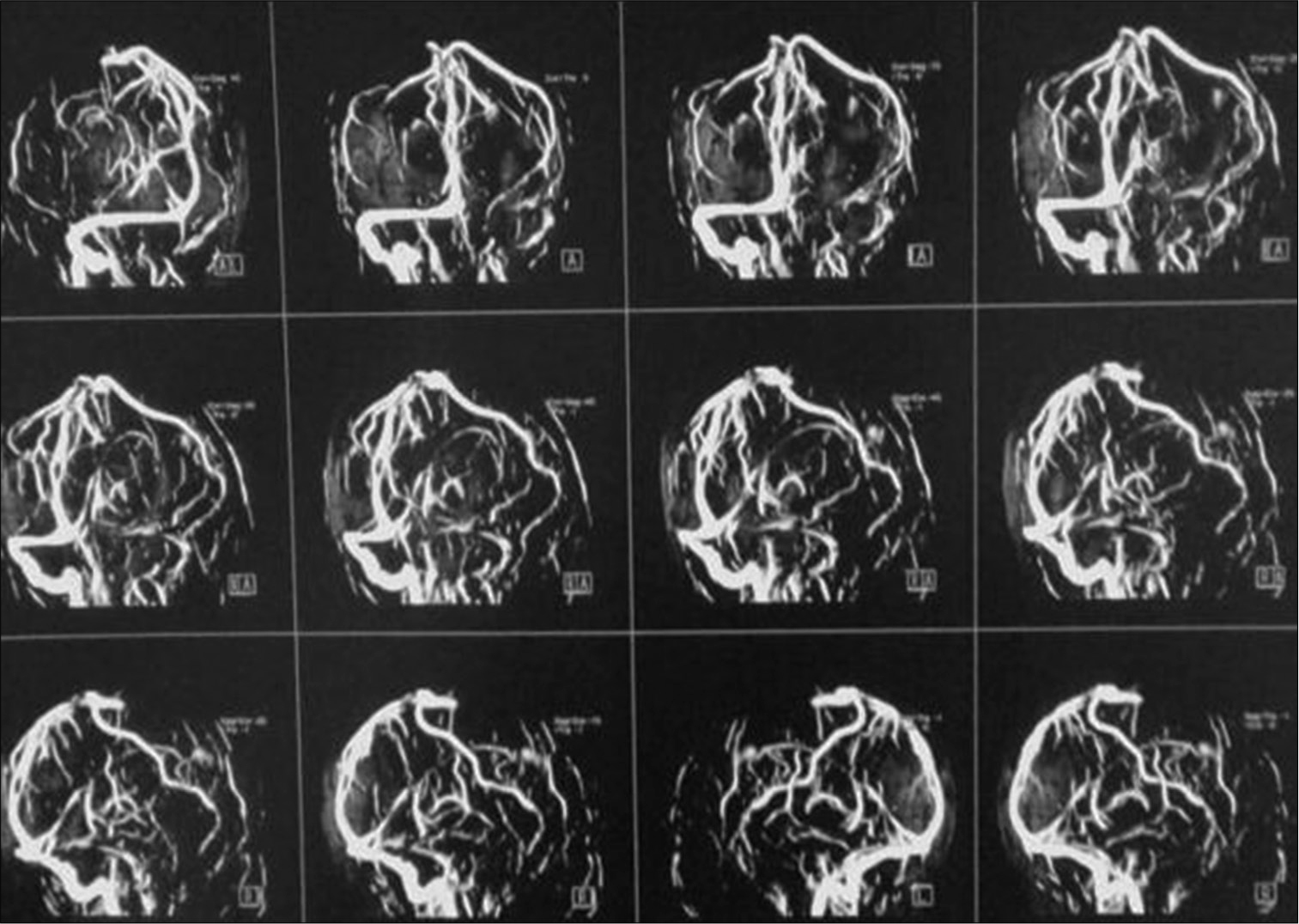
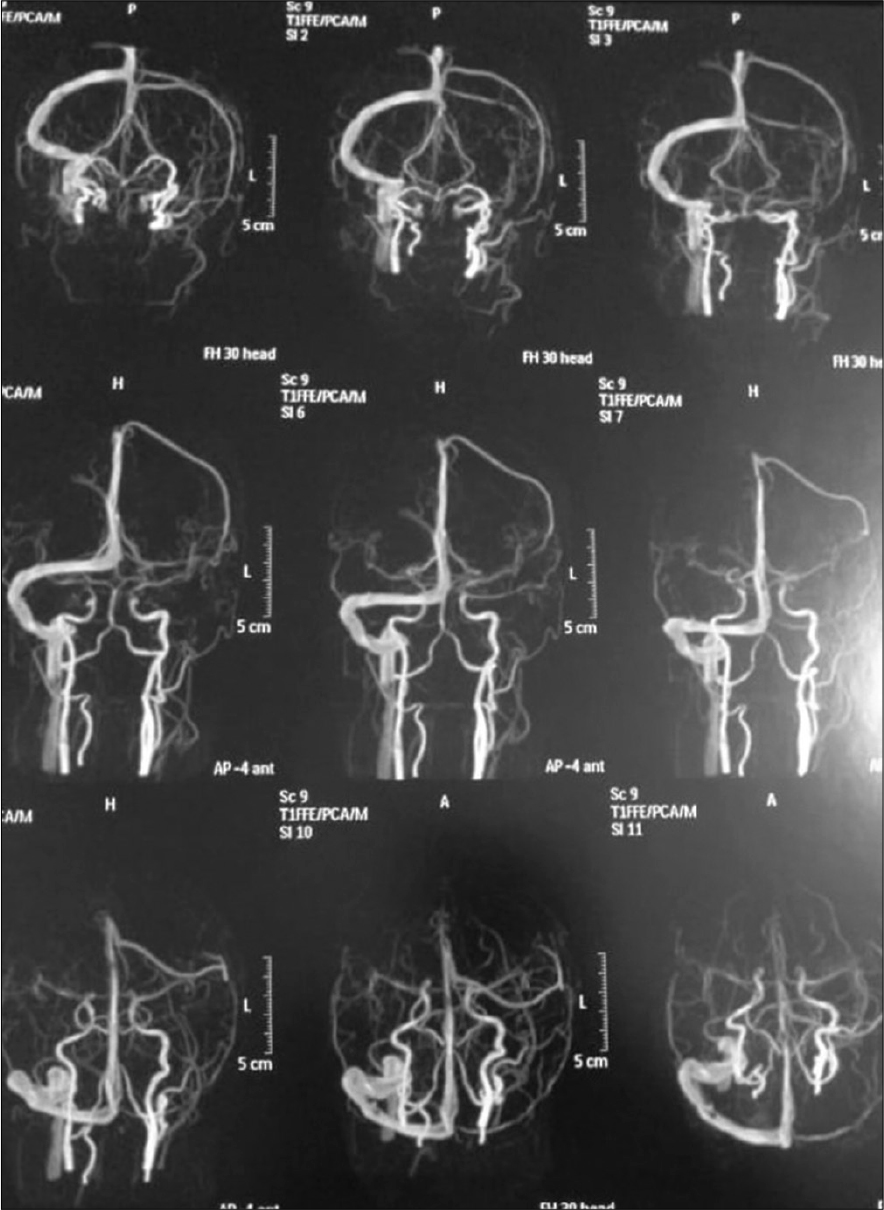
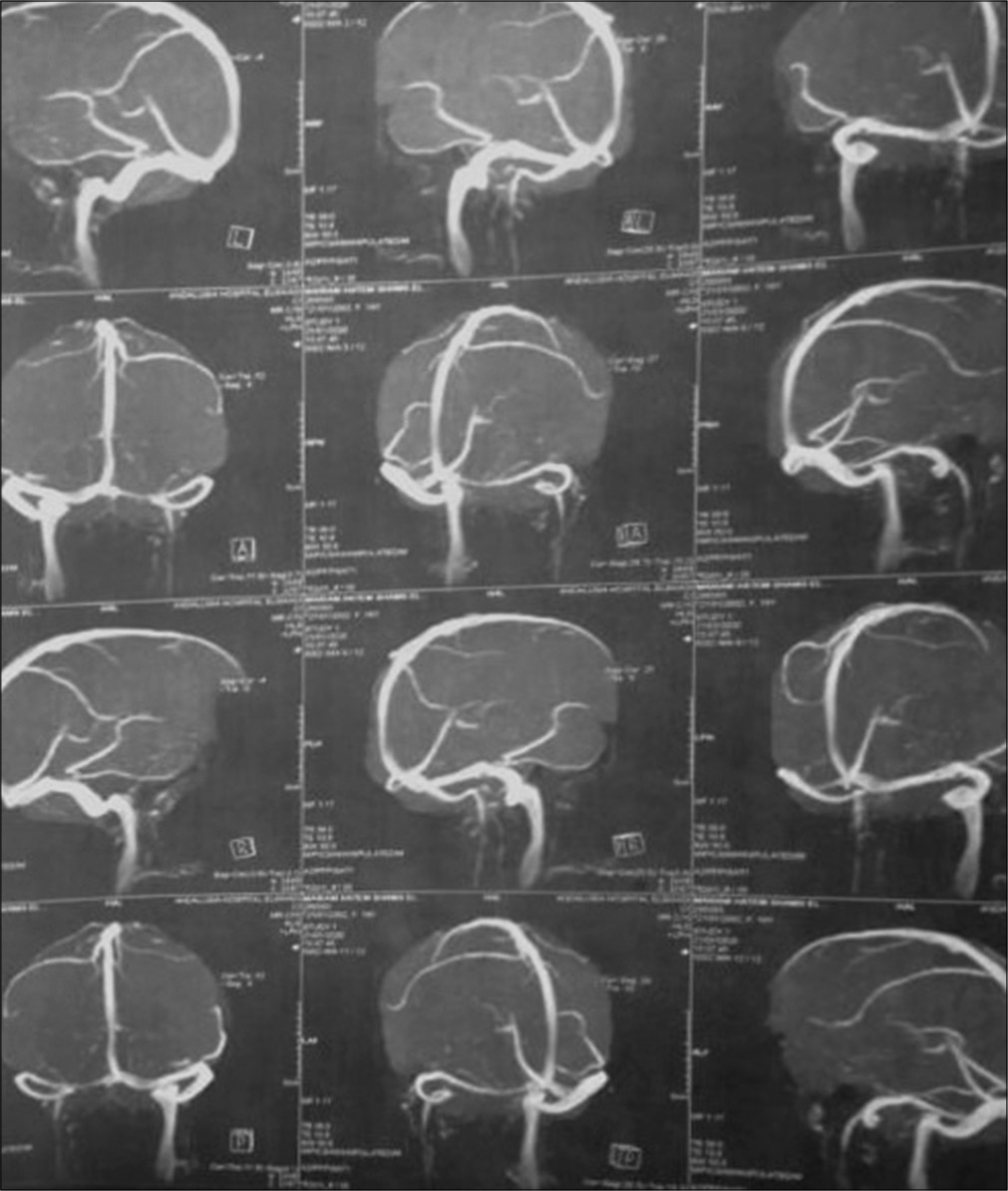
Patient was discharged after 1 week with GCS 15/15, with gradual improvement in motor power, she was maintained for four days on clexane, in addition to Apixaban and antiepileptic. Three months after surgery patient presented with an attack of tonic clonic convulsions, as she was not compliant to medical treatment. At follow-up visit, she was fully conscious with full motor power, no hypertonia, or exaggerated reflexes. MRI brain with/without contrast were done and revealed resolution of cortical and subcortical damage with small area of encephalomalacia, MRV showed obliteration of cortical veins and anterior half of superior sagittal sinus [
Figure 5:
Case 1 (a) Postoperative T1 WI without contrast, note disappearance of mass effect and replaced by encephalomalacic area filled with CSF appears as hypointense signal on T1 and hyperintense on T2 in (b) image. (c) T1 WI with contrast, note the slight enhancement of neural tissue and fill-defect in the superior sagittal sinus.
Case #7
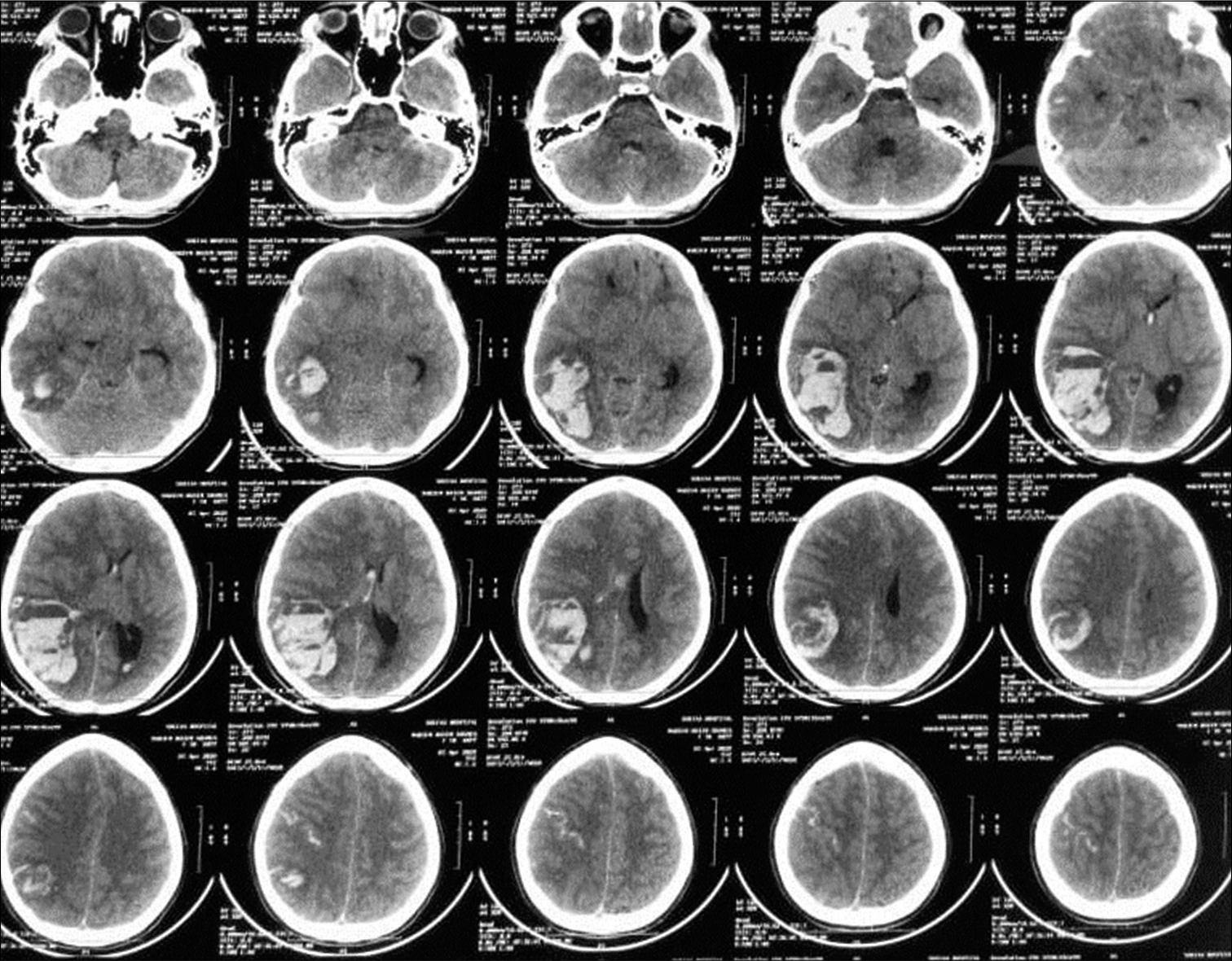
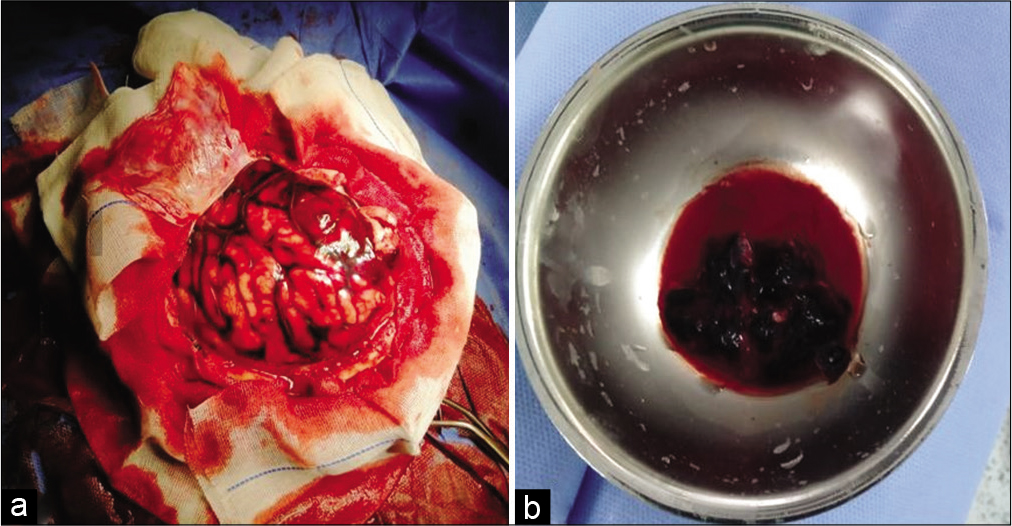
A 20-year-old female patient presented with known history of antiphospholipid syndrome. Three days ago, she started to have severe headache. One day later, she developed fits and decrease conscious level (E3V3M4). CT scan was done and a large occipitoparietal hematoma was seen on right side [
DISCUSSION
In our study, failure of usual medical treatment of elevated ICP in the management of hemorrhagic infarction of CVST is an indication for DC.[
Noncontrast CT scan was the first modality of choice to investigate patient with acute deficit or seizure. Thrombosed veins may manifest as elongated, cordlike hyperdense lesions (cord sign). The pathophysiology of this sign is clotted vessel. It is found in 20% of cases only. Empty delta sign is a triangular defect visualized on postcontrast imaging when SSS is filled with thrombus. This sign was seen in our series in postoperative MRIs for survived cases.
A DC is a good option for an enormous increment of intracranial tension, in comparison with data published before on implementation of DC in malignant edema due to MCA infarction, it has been found the survival rate was increased in the entity of “failed medical treatment” who underwent surgery versus sustained medical treatment.[
The cause of death in CVST is due to increased ICP with subsequent to transtentorial herniation. ICP rises due to a lot of mechanisms: (1) mass effect; (2) venous obstruction; (3) obstruction to CSF flow and absorption, especially if the dural sinuses with the arachnoid granulations are involved; and (4) brain swelling induced by ischemia.[
In [
In our series, we followed the indications of DC strictly.[
Endovascular treatment has no role in the management of hemorrhagic transformation with impending herniation.[
Stam et al.[
Giving anticoagulants in CVST are a crucial step in treatment. Anticoagulation prevents thrombus propagation, fasten spontaneous resolution, and helps in the prevention of deep vein thrombosis or embolism, without adversely promoting ICH.[
The time to restart therapeutic anticoagulation after DC is questionable.[
CONCLUSION
DC provides an urgent last arm for intractable increased intracranial tension. Patients with CVST need urgent consultation for neurosurgical intervention.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Acknowledgment
A great thanks to neurosurgery department of our institute for their help and support.
References
1. Aaron S, Alexander M, Moorthy RK, Mani S, Mathew V, Patil AK. Decompressive craniectomy in cerebral venous thrombosis: A single centre experience. J Neurol Neurosurg Psychiatry. 2013. 84: 995-1000
2. Armonda RA, Vo AH, Bell R, Neal C, Campbell WW. Multimodal monitoring during emergency hemicraniectomy for vein of labbe thrombosis. Neurocrit Care. 2006. 4: 241-4
3. Ashjazadeh N, Borhani A. Cerebral venous-sinus thrombosis : A case series analysis. Iran J Med Sci. 2011. 36: 178-82
4. Avanali R, Gopalakrishnan MS, Devi BI, Bhat DI, Shukla DP, Shanbhag NC. Role of decompressive craniectomy in the management of cerebral venous sinus thrombosis. Front Neurol. 2019. 10: 511
5. Barbati G, Montà GD, Coletta R, Blasetti AG. Post-traumatic superior sagittal sinus thrombosis. Case report and analysis of the international literature. Minerva Anestesiol. 2003. 69: 919-25
6. Coutinho JM, Majoie CB, Coert BA, Stam J. Decompressive hemicraniectomy in cerebral sinus thrombosis: Consecutive case series and review of the literature. Stroke. 2009. 40: 2233-5
7. Coutinho JM, Majoie CB, Coert BA, Stam J. Decompressive hemicraniectomy in cerebral sinus thrombosis: consecutive case series and review of the literature. Stroke. 2009. 40: 2233-5
8. Dohmen C, Galldiks N, Moeller-Hartmann W, Fink GR, Timmermann L. Sequential escalation of therapy in “malignant” cerebral venous and sinus thrombosis. Neurocrit Care. 2010. 12: 98-102
9. Ebke M, Jürgens KU, Tomandl B, Merten U, Kastrup A. Surgical treatment of space occupying edema and hemorrhage due to cerebral venous thrombosis during pregnancy. Neurocrit Care. 2011. 15: 166-9
10. Ferro JM, Crassard I, Coutinho JM, Canhão P, Barinagarrementeria F, Cucchiara B. Decompressive surgery in cerebrovenous thrombosis: A multicenter registry and a systematic review of individual patient data. Stroke. 2011. 42: 2825-31
11. Galarza M, Gazzeri R. Cerebral venous sinus thrombosis associated with oral contraceptives: The case for neurosurgery. Neurosurg Focus. 2009. 27: E5
12. Gioti I, Faropoulos K, Picolas C, Lambrou MA. Decompressive craniectomy in cerebral venous sinus thrombosis during pregnancy: A case report. Acta Neurochir (Wien). 2019. 161: 1349-52
13. Girot M, Ferro JM, Canhão P, Stam J, Bousser MG, Barinagarrementeria F. Predictors of outcome in patients with cerebral venous thrombosis and intracerebral haemorrhage. Stroke. 2007. 38: 337-42
14. Graf S, Prothmann S, Lehmberg J, Ilg R, Wunderlich S, Schneider J. Sinus thrombosis in a 15-year-old girl: An interdisciplinary case. Nervenarzt. 2015. 86: 743-7
15. Gupta RK, Jamjoom AA, Devkota UP. Superior sagittal sinus thrombosis presenting as a continuous headache: A case report and review of the literature. Cases J. 2009. 2: 9361
16. Keller E, Pangalu A, Fandino J, Könü D, Yonekawa Y. Decompressive craniectomy in severe cerebral venous and dural sinus thrombosis. Acta Neurochir Suppl. 2005. 94: 177-83
17. Kuroki K, Taguchi H, Sumida M, Onda J. Dural sinus thrombosis in a patient with protein S deficiency-case report. Neurol Med Chir (Tokyo). 1999. 39: 928-31
18. L Chefitz D. Cerebral venous sinus thrombosis: A case report with review of diagnosis and treatment strategies. J Clin Case Rep. 2014. 4: 1-3
19. Lanterna LA, Gritti P, Manara O, Grimod G, Bortolotti G, Biroli F. Decompressive surgery in malignant Dural sinus thrombosis: Report of 3 cases and review of the literature. Neurosurg Focus. 2009. 26: E5
20. Lath R, Kumar S, Reddy R, Boola GR, Ray A, Prabhakar S. Decompressive surgery for severe cerebral venous sinus thrombosis. Neurol India. 2010. 58: 392-7
21. Lechanoine F, Janot K, Herbreteau D, Maldonado IL, Velut S. Surgical thrombectomy combined with bilateral decompressive craniectomy in a life-threatening case of coma from cerebral venous sinus thrombosis: Case report and literature review. World Neurosurg. 2018. 120: 485-9
22. Prandoni P, Milan M, Sarolo L, Zanon E, Bilora F. Optimal duration of anticoagulation in patients with unprovoked venous thromboembolism: The impact of novel anticoagulants. Int Angiol. 2017. 36: 395-401
23. Lin HS, Lin JF, Chang CK, Tsai CC, Chen SJ. Cerebral sinus thrombosis with intracerebral hemorrhage in pregnancy: A case report. Acta Neurol Taiwan. 2008. 17: 189-93
24. Mallick AA, Sharples PM, Calvert SE, Jones RW, Leary M, Lux AL. Cerebral venous sinus thrombosis: A case series including thrombolysis. Arch Dis Child. 2009. 94: 790-4
25. Mohindra S, Umredkar A, Singla N, Bal A, Gupta SK. Decompressive craniectomy for malignant cerebral oedema of cortical venous thrombosis: An analysis of 13 patients. Br J Neurosurg. 2011. 25: 422-9
26. Nagpal RD. Dural sinus and cerebral venous thrombosis. Neurosurg Rev. 1983. 6: 155-60
27. Peng Y, Xuan JG, Yang YL, Wang SN. Endovascular thrombolysis for severe cerebral sinus thrombosis. Interv Neuroradiol. 2009. 15: 407-12
28. Pfeilschifter W, Neumann-Haefelin T, Hattingen E, Singer OC. Cortical venous thrombosis after a diagnostic lumbar puncture. Nervenarzt. 2009. 80: 1219-21
29. Vivakaran TT, Srinivas D, Kulkarni GB, Somanna S. The role of decompressive Craniectomy in cerebral venous sinus thrombosis. J Neurosurg. 2012. 117: 738-44
30. Raza E, Shamim MS, Wadiwala MF, Ahmed B, Kamal AK. Decompressive surgery for malignant cerebral venous sinus thrombosis: A retrospective case series from pakistan and comparative literature review. J Stroke Cerebrovasc Dis. 2014. 23: e13-22
31. Soyer B, Rusca M, Lukaszewicz AC, Crassard I, Guichard JP, Bresson D. Outcome of a cohort of severe cerebral venous thrombosis in intensive care. Ann Intensive Care. 2016. 6: 29
32. Stam J, Majoie CB, van Delden OM, van Lienden KP, Reekers JA. Endovascular thrombectomy and thrombolysis for severe cerebral sinus thrombosis. Stroke. 2008. 39: 1487-90
33. Stefini R, Latronico N, Cornali C, Rasulo F, Bollati A. Emergent decompressive craniectomy in patients with fixed dilated pupils due to cerebral venous and Dural sinus thrombosis: Report of three cases. Neurosurgery. 1999. 45: 626-9
34. Théaudin M, Crassard I, Bresson D, Saliou G, Favrole P, Vahedi K. Should decompressive surgery be performed in malignant cerebral venous thrombosis?: A series of 12 patients. Stroke. 2010. 41: 727-31
35. Wang Y, Wang Y, Zhang J, Du S, Wu J. Cerebral venous sinus incision for surgical thrombectomy combined with thrombolysis during decompressive craniectomy for malignant cerebral venous sinus thrombosis complicated with cerebral hernia. J Stroke Cerebrovasc Dis. 2019. 28: e60-3
36. Weber J, Spring A. Unilateral decompressive craniectomy for thrombosis of the left transverse and sigmoid sinuses. Zentralbl Neurochir. 2004. 65: 135-40
37. Zeng L, Derex L, Maarrawi J, Dailler F, Cakmak S, Nighoghossian N. Lifesaving decompressive craniectomy in “malignant” cerebral venous infarction. Eur J Neurol. 2007. 14: e27-8
38. Zhang S, Zhao H, Li H, You C, Hui X. Decompressive craniectomy in haemorrhagic cerebral venous thrombosis: Clinicoradiological features and risk factors. J Neurosurg. 2017. 127: 709-15
39. Zhen Y, Zhang N, He L, Shen L, Yan K. Mechanical thrombectomy combined with recombinant tissue plasminogen activator thrombolysis in the venous sinus for the treatment of severe cerebral venous sinus thrombosis. Exp Ther Med. 2015. 9: 1080-4
40. Zuurbier SM, Coutinho JM, Majoie CB, Coert BA, van den Munckhof P, Stam J. Decompressive hemicraniectomy in severe cerebral venous thrombosis: A prospective case series. J Neurol. 2012. 259: 1099-105