- The Brain Tumor Center and Pituitary Disorders Program, John Wayne Cancer Institute at Providence's Saint John's Health Center, 2200 Santa Monica Blvd., Santa Monica, CA 90404, USA
- Department of Otolaryngology, Pacific Eye and Ear Specialists, 11645 Wilshire Blvd, Los Angeles, CA 90025, USA
Correspondence Address:
Daniel F. Kelly
The Brain Tumor Center and Pituitary Disorders Program, John Wayne Cancer Institute at Providence's Saint John's Health Center, 2200 Santa Monica Blvd., Santa Monica, CA 90404, USA
DOI:10.4103/2152-7806.157442
Copyright: © 2015 Lobo B. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.How to cite this article: Lobo B, Heng A, Barkhoudarian G, Griffiths CF, Kelly DF. The expanding role of the endonasal endoscopic approach in pituitary and skull base surgery: A 2014 perspective. Surg Neurol Int 20-May-2015;6:82
How to cite this URL: Lobo B, Heng A, Barkhoudarian G, Griffiths CF, Kelly DF. The expanding role of the endonasal endoscopic approach in pituitary and skull base surgery: A 2014 perspective. Surg Neurol Int 20-May-2015;6:82. Available from: http://surgicalneurologyint.com/surgicalint_articles/expanding-role-endonasal-endoscopic-approach/
Abstract
Background:The past two decades have been the setting for remarkable advancement in endonasal endoscopic neurosurgery. Refinements in camera definition, surgical instrumentation, navigation, and surgical technique, including the dual surgeon team, have facilitated purely endonasal endoscopic approaches to the majority of the midline skull base that were previously difficult to access through the transsphenoidal microscopic approach.
Methods:This review article looks at many of the articles from 2011 to 2014 citing endonasal endoscopic surgery with regard to approaches and reconstructive techniques, pathologies treated and outcomes, and new technologies under consideration.
Results:Refinements in approach and closure techniques have reduced the risk of cerebrospinal fluid leak and infection. This has allowed surgeons to more aggressively treat a variety of pathologies. Four main pathologies with outcomes after treatment were identified for discussion: pituitary adenomas, craniopharyngiomas, anterior skull base meningiomas, and chordomas. Within all four of these tumor types, articles have demonstrated the efficacy, and in certain cases, the advantages over more traditional microscope-based techniques, of the endonasal endoscopic technique.
Conclusions:The endonasal endoscopic approach is a necessary tool in the modern skull base surgeon's armamentarium. Its efficacy for treatment of a wide variety of skull base pathologies has been repeatedly demonstrated. In the experienced surgeon's hands, this technique may offer the advantage of greater tumor removal with reduced overall complications over traditional craniotomies for select tumor pathologies centered near the midline skull base.
Keywords: Craniopharyngioma, chordoma, endoscopic skull base surgery, endoscopic endonasal surgery, endoscopic pituitary surgery, tuberculum sella meningioma
INTRODUCTION
In traditional transsphenoidal surgery, the operating microscope provides a clear view of the sella through a corridor created by a nasal speculum. While this approach has been a highly effective technique for most midline sellar pathology for over five decades, the “tunnel vision” and restricted maneuverability provided by this approach led neurosurgeons to seek better modes of visualizing and accessing the parasellar region.[
To date, the fully endonasal endoscopic approach (EEA) has been described to access the anterior middle fossa through the cribriform plate,[
Given the complex nature of many if not most midline skull base lesions and the fact that a majority may have sellar and pituitary gland involvement, the use of a multidisciplinary team is strongly advocated to optimize outcomes. This team approach should include specialists in neurosurgery, otorhinolaryngology (ENT), endocrinology, radiation oncology, medical oncology, neuro-ophthalmology, diagnostic and interventional neuroradiology, and neuropathology.[
ENDONASAL ENDOSCOPIC APPROACH AND SKULL BASE RECONSTRUCTION
History
Jho and Carrau's first description of their fully endoscopic endonasal technique utilized a single nostril, single surgeon approach with an endoscope holder in the majority of cases.[
Approach
In our center, we utilize a bimanual-binasal, two surgeon technique for all endonasal surgeries. All operations are performed using neuronavigation with face mask registration (Stryker Inc, Kalamazoo, Michigan, USA).[
The initial transnasal approach is performed by a single surgeon, usually an otolaryngologist with skull base endoscopic experience, by out-fracturing the middle and inferior turbinates bilaterally. The right middle turbinate is usually preserved to maintain endonasal physiologic function but in select rare cases removal is performed to facilitate movement of instruments and expose more lateral pathology. If a high flow CSF leak is expected, a full nasoseptal flap is raised at the beginning of the operation and placed in the nasopharynx during the main portion of the operation. Some centers raise a unilateral “rescue” flap and sacrifice the contralateral posterior nasoseptal pedicle prior to performing sphenoidotomies. A modification of the “rescue” flap may be made by placing the horizontal cut lower along the septum to spare the olfactory fibers within the superior strip of septum. We recently published a technique in which we perform bilateral olfactory fiber sparing modified “rescue” flaps in cases when a CSF leak is not expected. This technique preserves both posterior septal artery pedicles and promotes rapid healing while reducing the incidence of postoperative epistaxis and olfactory dysfunction.[
Reconstruction
High rates of postoperative CSF leakage following initial expanded endonasal approaches lead to the trials of several multilayered closure techniques[
Since its advent, the nasoseptal flap has become the work-horse for skull base reconstruction following expanded EEAs. Its centralized vascular pedicle and large surface area makes it ideal for coverage of nearly all central skull base defects. However, in some patients, the nasoseptal flap may not be an option because of prior surgery or involvement of the flap by tumor. In cases of an anterior fossa defect, the endoscopicaly harvested pericranial flap[
INDICATIONS AND OUTCOMES OF THE FULLY ENDONASAL ENDOSCOPIC APPROACH BY PATHOLOGY
Pituitary adenomas
Pituitary adenomas are the third most common intracranial neoplasm in adults after gliomas and meningiomas.[
Interestingly, a recent study published by Dallapiazza et al.[
Dallapiazza et al.[
Regarding functional adenomas, a recent paper by Starke et al. retrospectively reviewed a series of 113 patients treated by a fully endoscopic removal versus microscopic removal of a growth hormone secreting adenomas by two experienced pituitary surgeons.[
At this time, one can conclude the results from the endoscopic transsphenoidal surgical technique for pituitary adenomas is comparable to microscopic transsphenoidal surgery for the majority of endocrine-active and endocrine-inactive adenomas. The benefit of the endoscope may be for macroadenomas over 2 cm in maximal diameter and those with significant lateral and/or suprasellar extension: Areas for which the microscope has limited visualization. Such a case is demonstrated by [
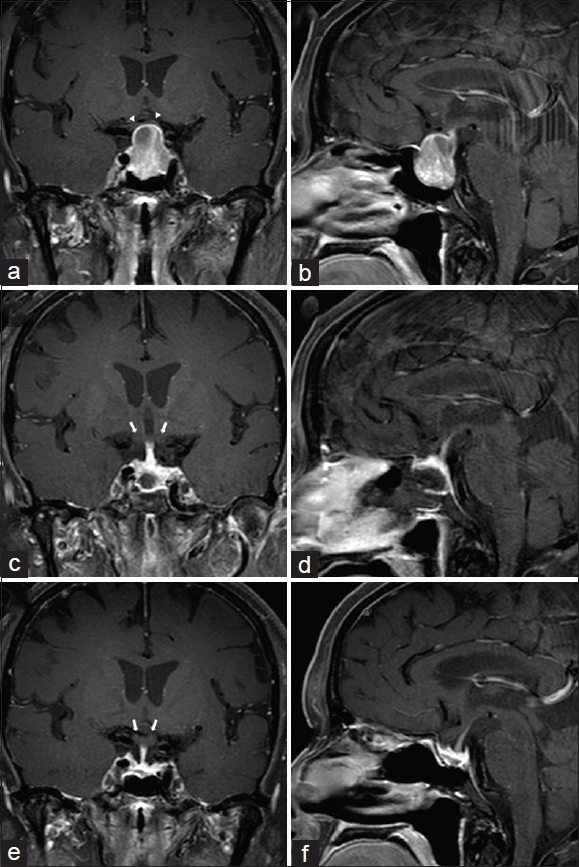
Figure 1
A 71-year-old female who developed progressive vision loss in the left eye over 6 months. Because of her worsening vision she underwent a brain MRI with gadolinium (a and b) revealing a 24 × 19 × 16 mm sellar mass with suprasellar extension. The chiasm was markedly compressed (white arrowheads). The normal gland was thinned and pushed superiorly toward the right. Formal neuro-ophthalmologic visual field testing confirmed severe global decreased acuity in the left eye, and a superior temporal quadrant defect in the right eye. Her preoperative prolactin level was 41.5 ng/ml indicating the tumor was not a prolactinoma. The patient underwent endonasal endoscopic tumor removal. The patient's immediate postoperative MRI (c and d) and 1 year postoperative MRI (e and f) revealed a GTR of the tumor and decompression of the optic apparatus (white arrows)
At our center, most pituitary adenoma patients are hospitalized for 1–2 nights depending on the extent of the procedure and have outpatient neurosurgical follow-up and their first nasal debridement at 7–10 days after surgery. They then typically have two additional nasal debridements at 2-week intervals and magnetic resonance imaging (MRI) at 3 months. Additional MRI and clinical follow-up are typically completed at 6 months or 12-month intervals to monitor for possible tumor recurrence or progression. All patients also have follow-up pituitary hormonal testing typically within 4–6 weeks of surgery and long-term endocrinology follow-up is arranged depending upon perioperative pituitary hormonal function and tumor subtype.
Craniopharyngiomas
Craniopharyngiomas represent the second most common neoplasm in the sellar/suprasellar region after pituitary adenomas.[
Both transcranial[
Recently, two larger case series have demonstrated the efficacy of the EEA for treatment of craniopharyngiomas.[
Komotar et al.[
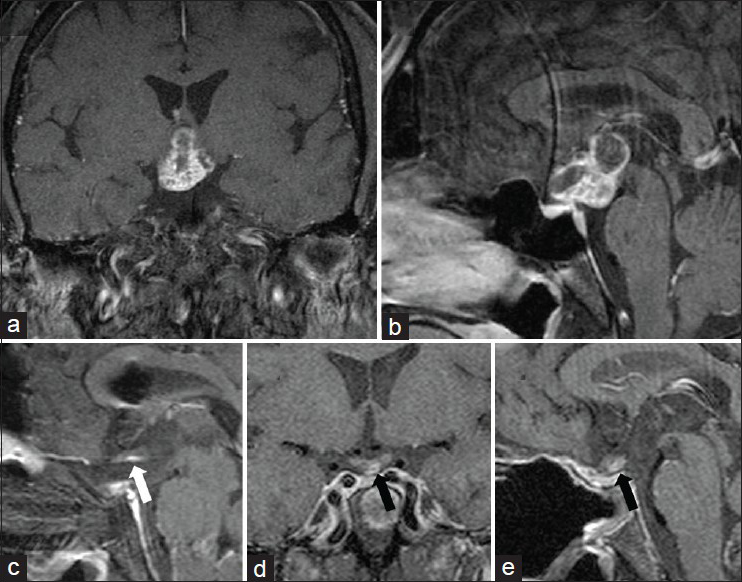
Figure 2
A 52-year-old male who presented with worsening bouts of cold intolerance, 18 kg weight gain over 2 years, fatigue, decreased libido, polyuria, and 6 months of worsening vision. Endocrinological workup revealed central hypothyroidism, hypogonadotropic hypotestosteronism. T1 MRI with gadolinium (a and b) demonstrated a heterogeneously enhancing cystic suprasellar retrochiasmatic mass. The patient underwent an endonasal endoscopic transsellar, transplanum resection of the tumor. The pathology was consistent with craniopharyngioma. The postoperative MRI with gadolinium 1 day after surgery (c) demonstrated near total removal of the tumor with a small amount of tumor purposefully left along the posterior edge of the optic chiasm given its dense adhesions to the optic apparatus (white arrow). MRI with gadolinium 6 months after surgery (d and e) reveals residual tumor along the chiasm that was followed with serial MRIs
Anterior skull base meningiomas
After the experience gained with endonasal endoscopic pituitary surgery, surgeons recognized several theoretical benefits of the EEA for anterior skull base meningiomas: Bilateral access to the tumor, removal of involved hypertrophied bone at the origin site of the tumor, early dural devascularization, and no required brain retraction. Koutourousiou et al. recently published the largest single institution series on olfactory groove[
Komotar et al. performed a meta-analysis of 60 studies published between 2001 and 2010 to assess the EEA versus traditional transcranial approaches for midline anterior cranial fossa meningiomas.[
Based on this information and our own experience [
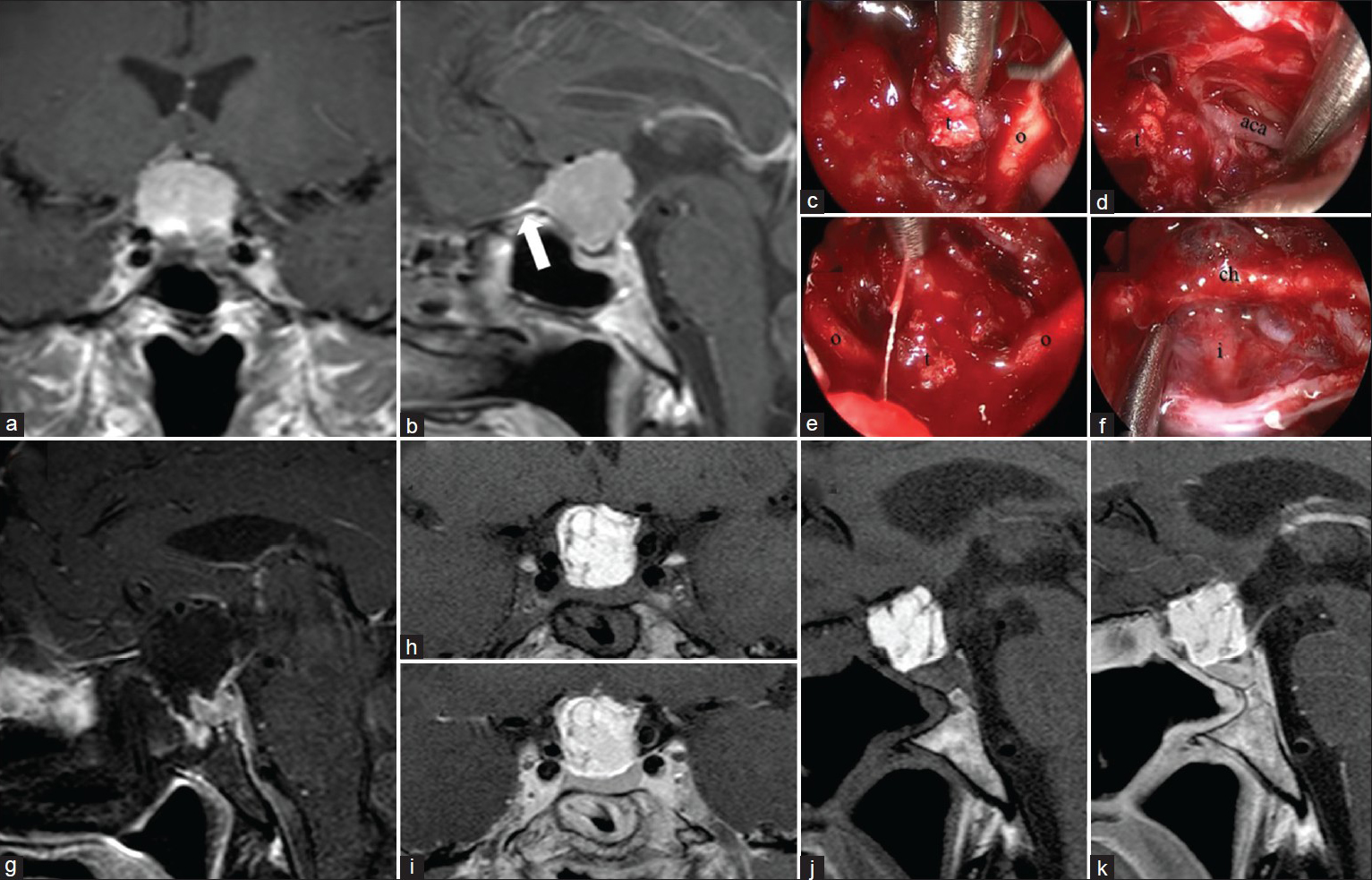
Figure 3
A 65-year-old female with gradual visual deterioration. The preoperative T1 MRI with gadolinium (a and b) demonstrated an extra-axial mass arising from the tuberculum sella dura with dural tail (white arrow). The patient underwent an endonasal endoscopic transplanum, transsellar skull base approach for resection of the mass. Pathology was consistent with a benign meningioma. Intraoperative views (c,d,e,f) of tumor (t) removal with separation away from the optic nerves (o), chiasm (ch), anterior cerebral artery (aca), and infundibulum (i). The immediate postoperative T1 MRI with fat suppression and gadolinium (g) demonstrated a GTR of the tumor. The 3-month postoperative T1 MRI without gadolinium (h and j) and with gadolinium (i and k) demonstrate fat graft in the resection cavity without evidence of tumor recurrence
Skull base chordomas
Chordomas are low grade malignant neoplasms that arise from notochordal remnants[
For midline approaches, the EEA offers the benefit of better illumination and visualization of tumor in deeper seated areas as well as tumor within the lateral extremes via angled endoscopes over traditional midline microscopic approaches. [
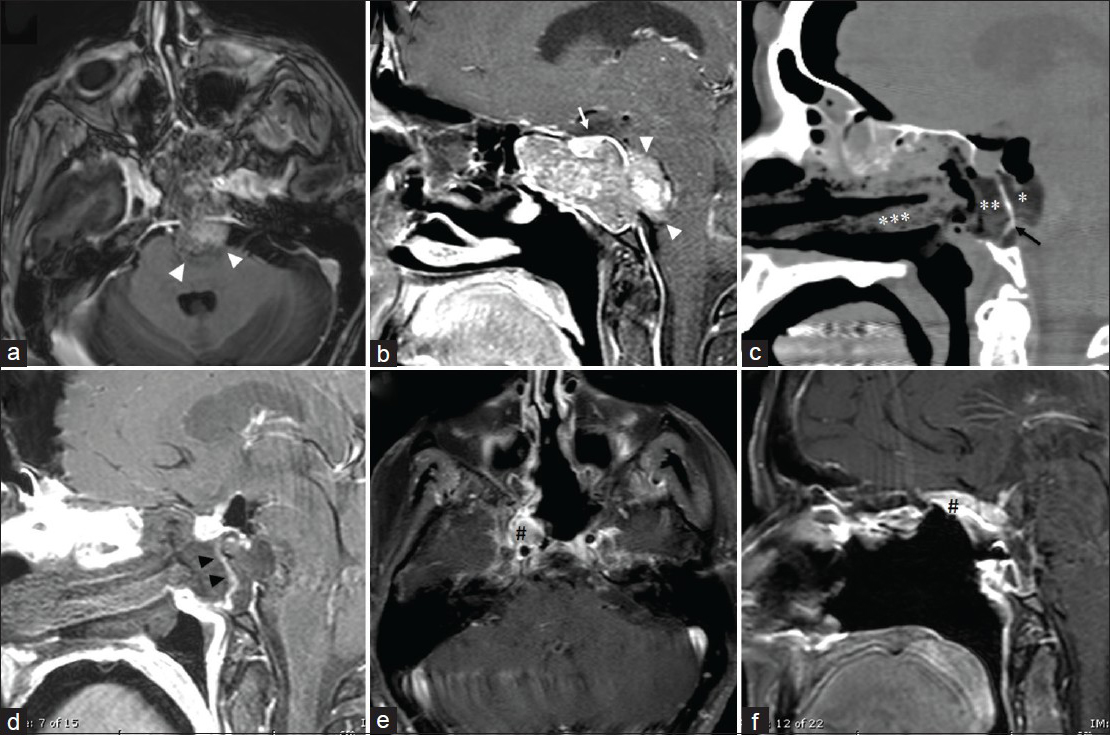
Figure 4
A 61-year-old female who presented with headaches. Her MRI with gadolinium (a and b) revealed a sphenoclival mass with preservation of sellar contents (white arrow) and intradural invasion and compression of the brain stem (white arrowheads). The patient underwent a transsphenoidal, transclival EEA for resection of the tumor. The pathology was consistent with chordoma. A postoperative CT (c) was completed immediately after surgery to assure no dislodgment of her multilayered skull base reconstruction with intradural fat graft (*), bone (black arrow), extradural fat graft (**), and right nasoseptal flap, which were buttressed with Merocel packs (***). Fat suppression T1 MRI with gadolinium was completed on postoperative day 1 (d) with clear enhancement of the nasoseptal flap (black arrowheads). Her fat suppression MRI with gadolinium 3 months after surgery (e and f) demonstrate no definitive tumor enhancement. The enhancement along the right posterior and superior nasal cavity (#) is consistent with the right nasoseptal flap enhancement
In the second largest reported series of chordomas managed with the EEA, Chibbaro et al. reported a similarly high rate of overall GTR of 65% in 54 patients, with a GTR rate of 88% in the subset of 32 patients with in newly diagnosed tumors.[
Recently, Amit et al. completed a meta-analysis to assess the efficacy of different surgical approaches for patients harboring chordomas.[
Based on these studies, the EEA provides an efficacious surgical management for patients with skull base chordomas with high rates of GTR in experienced hands. Given the relatively modern application of this approach, studies with long-term assessment of disease-free survival and overall survival will be needed to properly compare them with more traditional open surgical approaches. Nonetheless, in cases of large tumors and tumors with significant lateral extension, both an EEA and lateral approach may be needed to properly manage these tumors and achieve a GTR.
FUTURE ENDEAVORS
Three-dimensional neuroendoscopy
A notable limitation of current neuroendoscopy has been the loss of three-dimensional (3D) visuospatial orientation and depth perception with standard endoscopes. Although this can be recreated with dynamic movement of standard endoscopes, it may in part account for the significant learning curve when transitioning from a microscopic to a fully endoscopic technique. With laparoscopic surgery, earlier 3D endoscopes were noted to help with improved depth perception but were also associated with more vision strain and headaches over conventional endoscopes.[
Malleable endoscopes
A common problem experienced by many surgeons is the collision of the endoscope with surgical instrumentation resulting in limited surgical freedom. Although with experience this problem is surmountable, frustration and inadvertent collision with the endoscope during a critical surgical moment has potential for resulting in a significant complication. Sources for collision of the surgical instrument with the endoscope are extranasaly at the site of the camera head and light cord, within the nasal cavity along the shaft of the endoscope, and at the tip of the endoscope. A further source of limited surgical freedom occurs when advancement of endoscope for visualization of deeper structures results in the camera head, lighting cord, and hand of endoscope driver obstructing the movement of lead surgeon's hands. Malleable endoscopes offer the potential advantage of reducing surgical collision by adjusting the camera head out of the way from entrance of the nasal cavity. Elhadi et al. assessed surgical freedom of the VisionSense 3D malleable endoscope with the VisionSense 3D rigid endoscope utilizing a cadaver model.[
CONCLUSION
Significant advances over the past two decades in imaging technology, surgical instrumentation, skull base anatomical knowledge, and reconstructive techniques have resulted in the extended EEA becoming an integral part of the modern skull base surgeon's armamentarium. With growing use and greater experience, surgical outcomes continue to incrementally improve across many skull base pathologies, both benign and malignant. However, the importance of the learning curve in endoscopic surgery and use of a multi-disciplinary approach cannot be over-emphasized. As with any new surgical tool and technique, a firm foundation built on basic surgical technique and anatomical knowledge should be obtained prior to advancing to more complex surgical approaches and pathology. Additionally, realizing the limits of the endonasal approach and the reasonable use of transcranial approaches is critical. Finally, understanding the indications for radiosurgery, stereotactic radiotherapy, and medical treatment options within the treatment armamentarium for skull base pathology is also essential to optimize outcomes and reduce risk of surgical complications.
References
1. Al-Mefty O, Borba LA. Skull base chordomas: A management challenge. J Neurosurg. 1997. 86: 182-9
2. Alahmadi H, Cusimano MD, Woo K, Mohammed AA, Goguen J, Smyth HS. Impact of technique on cushing disease outcome using strict remission criteria. Can J Neurol Sci. 2013. 40: 334-41
3. Amit M, Na’ara S, Binenbaum Y, Billan S, Sviri G, Cohen JT. Treatment and Outcome of Patients with Skull Base Chordoma: A Meta-analysis. J Neurol Surg B Skull Base. 2014. 75: 383-90
4. Apuzzo ML, Heifetz MD, Weiss MH, Kurze T. Neurosurgical endoscopy using the side-viewing telescope. J Neurosurg. 1977. 46: 398-400
5. Barkhoudarian G, Del Carmen Becerra Romero A, Laws ER. Evaluation of the 3-dimensional endoscope in transsphenoidal surgery. Neurosurgery. 2013. 73: ons74-8
6. Battaglia P, Turri-Zanoni M, Dallan I, Gallo S, Sica E, Padoan G. Endoscopic endonasal transpterygoid transmaxillary approach to the infratemporal and upper parapharyngeal tumors. Otolaryngol Head Neck Surg. 2014. 150: 696-702
7. Bouropoulou V, Bosse A, Roessner A, Vollmer E, Edel G, Wuisman P. Immunohistochemical investigation of chordomas: Histogenetic and differential diagnostic aspects. Curr Top Pathol. 1989. 80: 183-203
8. Bramerson A, Nordin S, Bende M. Clinical experience with patients with olfactory complaints, and their quality of life. Acta Otolaryngol. 2007. 127: 167-74
9. Bresson D, McLaughlin N, Ditzel Filho LF, Griffiths CF, Carrau RL, Kelly DF. Endoscopic endonasal approach for the treatment of schwannomas of the pterygopalatine fossa: Case report and review of the literature. Neurochirurgie. 2014. 60: 174-9
10. Bushe KA, Halves E. Modified technique in transsphenoidal operations of pituitary adenomas. Technical note (author's transl). Acta Neurochir. 1978. 41: 163-75
11. Cavallo LM, Frank G, Cappabianca P, Solari D, Mazzatenta D, Villa A. The endoscopic endonasal approach for the management of craniopharyngiomas: A series of 103 patients. J Neurosurg. 2014. 121: 100-13
12. Chakrabarti I, Amar AP, Couldwell W, Weiss MH. Long-term neurological, visual, and endocrine outcomes following transnasal resection of craniopharyngioma. J Neurosurg. 2005. 102: 650-7
13. Chan AC, Chung SC, Yim AP, Lau JY, Ng EK, Li AK. Comparison of two-dimensional vs three-dimensional camera systems in laparoscopic surgery. Surg Endosc. 1997. 11: 438-40
14. Chibbaro S, Cornelius JF, Froelich S, Tigan L, Kehrli P, Debry C. Endoscopic endonasal approach in the management of skull base chordomas--clinical experience on a large series, technique, outcome, and pitfalls. Neurosurg Rev. 2014. 37: 217-24
15. Clark AJ, Jahangiri A, Garcia RM, George JR, Sughrue ME, McDermott MW. Endoscopic surgery for tuberculum sellae meningiomas: A systematic review and meta-analysis. Neurosurg Rev. 2013. 36: 349-59
16. Colli B, Al-Mefty O. Chordomas of the craniocervical junction: follow-up review and prognostic factors. J Neurosurg. 2001. 95: 933-43
17. Colli BO, Al-Mefty O. Chordomas of the skull base: Follow-up review and prognostic factors. Neurosurg Focus. 2001. 10: E1-
18. Conger AR, Lucas J, Zada G, Schwartz TH, Cohen-Gadol AA. Endoscopic extended transsphenoidal resection of craniopharyngiomas: Nuances of neurosurgical technique. Neurosurg Focus. 2014. 37: E10-
19. Couldwell WT, Weiss MH, Rabb C, Liu JK, Apfelbaum RI, Fukushima T. Variations on the standard transsphenoidal approach to the sellar region, with emphasis on the extended approaches and parasellar approaches: Surgical experience in 105 cases. Neurosurgery. 2004. 55: 539-50
20. Dallapiazza R, Bond AE, Grober Y, Louis RG, Payne SC, Oldfield EH. Retrospective analysis of a concurrent series of microscopic versus endoscopic transsphenoidal surgeries for Knosp Grades 0-2 nonfunctioning pituitary macroadenomas at a single institution. J Neurosurg. 2014. 121: 511-7
21. Dallapiazza RF, Grober Y, Starke RM, Laws ER, Jane JA. Long-term Results of Endonasal Endoscopic Transsphenoidal Resection of Nonfunctioning Pituitary Macroadenomas. Neurosurgery. 2015. 76: 42-52
22. Deraniyagala RL, Yeung D, Mendenhall WM, Li Z, Morris CG, Mendenhall NP. Proton therapy for skull base chordomas: An outcome study from the university of Florida proton therapy institute. J Neurol Surg B Skull Base. 2014. 75: 53-7
23. Drazin D, Zhuang L, Schievink WI, Mamelak AN. Expanded endonasal approach for the clipping of a ruptured basilar aneurysm and feeding artery to a cerebellar arteriovenous malformation. J Clin Neurosci. 2012. 19: 144-8
24. Dusick JR, Esposito F, Malkasian D, Kelly DF. Avoidance of carotid artery injuries in transsphenoidal surgery with the Doppler probe and micro-hook blades. Neurosurgery. 2007. 60: 322-8
25. Elhadi AM, Zaidi HA, Hardesty DA, Williamson R, Cavallo C, Preul MC. Malleable endoscope increases surgical freedom compared with a rigid endoscope in endoscopic endonasal approaches to the parasellar region. Neurosurgery. 2014. 10 Suppl 3: 393-9
26. Elliott RE, Hsieh K, Hochm T, Belitskaya-Levy I, Wisoff J, Wisoff JH. Efficacy and safety of radical resection of primary and recurrent craniopharyngiomas in 86 children. J Neurosurg Pediatr. 2010. 5: 30-48
27. Elliott RE, Jane JA, Wisoff JH. Surgical management of craniopharyngiomas in children: Meta-analysis and comparison of transcranial and transsphenoidal approaches. Neurosurgery. 2011. 69: 630-43
28. Elliott RE, Wisoff JH. Surgical management of giant pediatric craniopharyngiomas. J Neurosurg Pediatr. 2010. 6: 403-16
29. Ensenat J, Alobid I, de Notaris M, Sanchez M, Valero R, Prats-Galino A. Endoscopic endonasal clipping of a ruptured vertebral-posterior inferior cerebellar artery aneurysm: Technical case report. Neurosurgery. 2011. 69: onsE121-7
30. Fatemi N, Dusick JR, De Paiva Neto MA, Malkasian D, Kelly DF. Endonasal versus supraorbital keyhole removal of craniopharyngiomas and tuberculum sellae meningiomas. Neurosurgery. 2009. 64: S269-86
31. Fatemi N, Dusick JR, de Paiva Neto MA, Malkasian D, Kelly DF. Endonasal versus supraorbital keyhole removal of craniopharyngiomas and tuberculum sellae meningiomas. Neurosurgery. 2009. 64: S269-84
32. Fernandez-Miranda JC, Morera VA, Snyderman CH, Gardner P. Endoscopic endonasal transclival approach to the jugular tubercle. Neurosurgery. 2012. 71: 146-58
33. Gamea A, Fathi M, el-Guindy A. The use of the rigid endoscope in trans-sphenoidal pituitary surgery. J Laryngol Otol. 1994. 108: 19-22
34. Gandhi CD, Christiano LD, Eloy JA, Prestigiacomo CJ, Post KD. The historical evolution of transsphenoidal surgery: Facilitation by technological advances. Neurosurg Focus. 2009. 27: E8-
35. Gardner PA, Kassam AB, Thomas A, Snyderman CH, Carrau RL, Mintz AH. Endoscopic endonasal resection of anterior cranial base meningiomas. Neurosurgery. 2008. 63: 36-54
36. Gardner PA, Prevedello DM, Kassam AB, Snyderman CH, Carrau RL, Mintz AH. The evolution of the endonasal approach for craniopharyngiomas. J Neurosurg. 2008. 108: 1043-7
37. Gazzeri R, Nishiyama Y, Teo C. Endoscopic supraorbital eyebrow approach for the surgical treatment of extraaxialand intraaxial tumors. Neurosurg Focus. 2014. 37: E20-
38. Gerganov V, Metwali H, Samii A, Fahlbusch R, Samii M. Microsurgical resection of extensive craniopharyngiomas using a frontolateral approach: Operative technique and outcome. J Neurosurg. 2014. 120: 559-70
39. Griffiths CF, Cutler AR, Duong HT, Bardo G, Karimi K, Barkhoudarian G. Avoidance of postoperative epistaxis and anosmia in endonasal endoscopic skull base surgery: A technical note. Acta Neurochir. 2014. 156: 1393-401
40. Hadad G, Bassagasteguy L, Carrau RL, Mataza JC, Kassam A, Snyderman CH. A novel reconstructive technique after endoscopic expanded endonasal approaches: Vascular pedicle nasoseptal flap. Laryngoscope. 2006. 116: 1882-6
41. Hadad G, Rivera-Serrano CM, Bassagaisteguy LH, Carrau RL, Fernandez-Miranda J, Prevedello DM. Anterior pedicle lateral nasal wall flap: A novel technique for the reconstruction of anterior skull base defects. Laryngoscope. 2011. 121: 1606-10
42. Hafez MA, ElMekkawy S, AbdelBadie H, Mohy M, Omar M. Pediatric craniopharyngioma--rationale for multimodal management: The Egyptian experience. J Pediatr Endocrinol Metab. 2006. 19 Suppl 1: 371-80
43. Hanna GB, Shimi SM, Cuschieri A. Randomised study of influence of two-dimensional versus three-dimensional imaging on performance of laparoscopic cholecystectomy. Lancet. 1998. 351: 248-51
44. Helal MZ. Combined micro-endoscopic trans-sphenoid excisions of pituitary macroadenomas. Eur Arch Oto-rhinolaryngol. 1995. 252: 186-9
45. Hoffman HJ. Surgical management of craniopharyngioma. Pediatr Neurosurg. 1994. 21 Suppl 1: S44-9
46. Jho HD, Carrau RL. Endoscopic endonasal transsphenoidal surgery: Experience with 50 patients. J Neurosurg. 1997. 87: 44-51
47. Jho HD, Ha HG. Endoscopic endonasal skull base surgery: Part 1-the midline anterior fossa skull base. Minim Invasive Neurosurg. 2004. 47: 1-8
48. Jho HD, Ha HG. Endoscopic endonasal skull base surgery: Part 3-the clivus and posterior fossa. Minim Invasive Neurosurg. 2004. 47: 16-23
49. Jho HD, Carrau RL. Endoscopy assisted transsphenoidal surgery for pituitary adenoma. Technical note. Acta Neurochir. 1996. 138: 1416-25
50. Kaptain GJ, Vincent DA, Sheehan JP, Laws ER. Transsphenoidal approaches for the extracapsular resection of midline suprasellar and anterior cranial base lesions. Neurosurgery. 2001. 49: 94-101
51. Kari E, Oyesiku NM, Dadashev V, Wise SK. Comparison of traditional 2-dimensional endoscopic pituitary surgery with new 3-dimensional endoscopic technology: Intraoperative and early postoperative factors. Int Forum Allergy Rhinol. 2012. 2: 2-8
52. Kassam A, Gardner P, Snyderman C, Carrau R, Zimmer L, Hirsch B. Endoscopic, Expanded Endonasal Approach to the Jugular Foramen. Oper Tech Neurosurg. 2005. 8: 35-41
53. Kassam A, Snyderman C, Gardner P, Carrau R, Spiro R. The expanded endonasal approach: A fully endoscopic transnasal approach and resection of the odontoid process: Technical case report. Neurosurgery. 2005. 57: E213-
54. Kassam A, Snyderman CH, Mintz A, Gardner P, Carrau RL. Expanded endonasal approach: The rostrocaudal axis. Part I. Crista galli to the sella turcica. Neurosurg Focus. 2005. 19: E3-
55. Kassam A, Snyderman CH, Mintz A, Gardner P, Carrau RL. Expanded endonasal approach: The rostrocaudal axis. Part II. Posterior clinoids to the foramen magnum. Neurosurg Focus. 2005. 19: E4-
56. Kassam A, Carrau RL, Snyderman CH, Gardner P, Mintz A. Evolution of reconstructive techniques following endoscopic expanded endonasal approaches. Neurosurg Focus. 2005. 19: E8-
57. Kassam AB, Gardner P, Snyderman C, Mintz A, Carrau R. Expanded endonasal approach: Fully endoscopic, completely transnasal approach to the middle third of the clivus, petrous bone, middle cranial fossa, and infratemporal fossa. Neurosurg Focus. 2005. 19: E6-
58. Kassam AB, Prevedello DM, Carrau RL, Snyderman CH, Gardner P, Osawa S. The front door to meckel's cave: An anteromedial corridor via expanded endoscopic endonasal approach-technical considerations and clinical series. Neurosurgery. 2009. 64: S71-83
59. Komatsu F, Komatsu M, Di leva A, Tschabitscher M. Endoscopic approaches to the trigeminal nerve and clinical consideration for trigeminal schwannomas: A cadaveric study. J Neurosurgery. 2012. 117: 690-6
60. Komotar RJ, Starke RM, Raper DM, Anand VK, Schwartz TH. Endoscopic endonasal compared with microscopic transsphenoidal and open transcranial resection of craniopharyngiomas. World Neurosurg. 2012. 77: 329-41
61. Komotar RJ, Starke RM, Raper DM, Anand VK, Schwartz TH. Endoscopic endonasal compared with microscopic transsphenoidal and open transcranial resection of giant piuitary adenomas. Pituitary. 2012. 15: 150-9
62. Komotar RJ, Starke RM, Raper DM, Anand VK, Schwartz TH. Endoscopic endonasal versus open transcranial resection of anterior midline skull base meningiomas. World Neurosurg. 2012. 77: 713-24
63. Kouri JG, Chen MY, Watson JC, Oldfield EH. Resection of suprasellar tumors by using a modified transsphenoidal approach. Report of four cases. J Neurosurg. 2000. 92: 1028-35
64. Koutourousiou M, Fernandez-Miranda JC, Stefko ST, Wang EW, Snyderman CH, Gardner PA. Endoscopic endonasal surgery for suprasellar meningiomas: Experience with 75 patients. J Neurosurg. 2014. 120: 1326-39
65. Koutourousiou M, Fernandez-Miranda JC, Wang EW, Snyderman CH, Gardner PA. Endoscopic endonasal surgery for olfactory groove meningiomas: Outcomes and limitations in 50 patients. Neurosurg Focus. 2014. 37: E8-
66. Koutourousiou M, Filho FV, Costacou T, Fernandez-Miranda JC, Wang EW, Snyderman CH. Pontine encephalocele and abnormalities of the posterior fossa following transclival endoscopic endonasal surgery. J Neurosurg. 2014. 121: 359-66
67. Koutourousiou M, Gardner PA, Fernandez-Miranda JC, Tyler-Kabara EC, Wang EW, Snyderman CH. Endoscopic endonasal surgery for craniopharyngiomas: Surgical outcome in 64 patients. J Neurosurg. 2013. 119: 1194-207
68. Koutourousiou M, Gardner PA, Tormenti MJ, Henry SL, Stefko ST, Kassam AB. Endoscopic endonasal approach for resection of cranial base chordomas: Outcomes and learning curve. Neurosurgery. 2012. 71: 614-24
69. Laws ER. Surgical outcome in 90 patients with craniopharyngiomas: An evaluation of the transsphenoidal approach. World Neurosurg. 2010. 74: 254-5
70. Liu JK, Schmidt RF, Choudhry OJ, Shukla PA, Eloy JA. Surgical nuances for nasoseptal flap reconstruction of cranial base defects with high-flow cerebrospinal fluid leaks after endoscopic skull base surgery. Neurosurg Focus. 2012. 32: E7-
71. Maira G, Anile C, Rossi GF, Colosimo C. Surgical treatment of craniopharyngiomas: An evaluation of the transsphenoidal and pterional approaches. Neurosurgery. 1995. 36: 715-24
72. Marcus HJ, Hughes-Hallett A, Cundy TP, Di Marco A, Pratt P, Nandi D. Comparative effectiveness of 3-dimensional vs 2-dimensional and high-definition vs standard-definition neuroendoscopy: A preclinical randomized crossover study. Neurosurgery. 2014. 74: 375-80
73. Mascarenhas L, Moshel YA, Bayad F, Szentirmai O, Salek AA, Leng LZ. The transplanum transtuberculum approaches for suprasellar and sellar-suprasellar lesions: Avoidance of cerebrospinal fluid leak and lessons learned. World Neurosurg. 2014. 82: 186-95
74. Mazzatenta D, Zoli M, Mascari C, Pasquini E, Frank G. Endoscopic endonasal odontoidectomy: Clinical series. Spine. 2014. 39: 846-53
75. McLaughlin N, Carrau RL, Kassam AB, Kelly DF. Neuronavigation in endonasal pituitary and skull base surgery using an autoregistration mask without head fixation: An assessment of accuracy and practicality. J Neurol Surg A Cent Eur Neurosurg. 2012. 73: 351-7
76. McLaughlin N, Eisenberg AA, Cohan P, Chaloner CB, Kelly DF. Value of endoscopy for maximizing tumor removal in endonasal transsphenoidal pituitary adenoma surgery. J Neurosurg. 2013. 118: 613-20
77. McLaughlin N, Laws ER, Oyesiku NM, Katznelson L, Kelly DF. Pituitary centers of excellence. Neurosurgery. 2012. 71: 916-24
78. Morera VA, Fernandez-Miranda JC, Prevedello DM, Madhok R, Barges-Coll J, Gardner P. “Far-medial” expanded endonasal approach to the inferior third of the clivus: The transcondylar and transjugular tubercle approaches. Neurosurgery. 2010. 66: S211-20
79. Ostrom QT, Gittleman H, Liao P, Rouse C, Chen Y, Dowling J. CBTRUS statistical report: Primary brain and central nervous system tumors diagnosed in the United States in 2007-2011. Neuro Oncol. 2014. 16 Suppl 4: iv1-63
80. Ottenhausen M, Banu MA, Placantonakis DG, Tsiouris AJ, Khan OH, Anand VK. Endoscopic endonasal resection of suprasellar meningiomas: The importance of case selection and experience in determining extent of resection, visual improvement, and complications. World Neurosurg. 2014. 82: 442-9
81. Patel MR, Taylor RJ, Hackman TG, Germanwala AV, Sasaki-Adams D, Ewend MG. Beyond the nasoseptal flap: Outcomes and pearls with secondary flaps in endoscopic endonasal skull base reconstruction. Laryngoscope. 2014. 124: 846-52
82. Prevedello DM, Barges-Coll J, Fernandez-Miranda JC, Morera V, Jacobson D, Madhok R. Middle turbinate flap for skull base reconstruction: Cadaveric feasibility study. Laryngoscope. 2009. 119: 2094-8
83. Puget S, Garnett M, Wray A, Grill J, Habrand JL, Bodaert N. Pediatric craniopharyngiomas: Classification and treatment according to the degree of hypothalamic involvement. J Neurosurg. 2007. 106: 3-12
84. Rivera-Serrano CM, Bassagaisteguy LH, Hadad G, Carrau RL, Kelly D, Prevedello DM. Posterior pedicle lateral nasal wall flap: New reconstructive technique for large defects of the skull base. Am J Rhinol Allergy. 2011. 25: e212-6
85. Sanjari R, Mortazavi SA, Amiri RS, Ardestani SH, Amirjamshidi A. Intrasphenoidal Meningo-encephalocele: Report of two rare cases and review of literature. Surg Neurol Int. 2013. 4: 5-
86. Sen C, Triana AI, Berglind N, Godbold J, Shrivastava RK. Clival chordomas: Clinical management, results, and complications in 71 patients. J Neurosurg. 2010. 113: 1059-71
87. Somma AD, Bronzoni C, Guadagno E, Solari D, Dell’aversana GO, De Caro BS. The “extended” endoscopic endonasal approach for the removal of a mixed intrasuprasellar germinoma: Technical case report. Surg Neurol Int. 2014. 5: 14-
88. Starke RM, Raper DM, Payne SC, Vance ML, Oldfield EH, Jane JA. Endoscopic vs microsurgical transsphenoidal surgery for acromegaly: Outcomes in a concurrent series of patients using modern criteria for remission. J Clin Endocrinol Metab. 2013. 98: 3190-8
89. Uhl M, Mattke M, Welzel T, Roeder F, Oelmann J, Habl G. Highly effective treatment of skull base chordoma with carbon ion irradiation using a raster scan technique in 155 patients: First long-term results. Cancer. 2014. 120: 3410-7
90. Van Gompel JJ, Alikhani P, Tabor MH, van Loveren HR, Agazzi S, Froelich S. Anterior inferior petrosectomy: Defining the role of endonasal endoscopic techniques for petrous apex approaches. J Neurosurg. 2014. 120: 1321-5
91. Wilson DA, Duong H, Teo C, Kelly DF. The supraorbital endoscopic approach for tumors. World Neurosurg. 2014. 82: e243-56
92. Zanation AM, Snyderman CH, Carrau RL, Kassam AB, Gardner PA, Prevedello DM. Minimally invasive endoscopic pericranial flap: A new method for endonasal skull base reconstruction. Laryngoscope. 2009. 119: 13-8
93. Zygourakis CC, Kaur G, Kunwar S, McDermott MW, Madden M, Oh T. Modern treatment of 84 newly diagnosed craniopharyngiomas. J Clin Neurosci. 2014. 21: 1558-66