- Department of Ear Nose and Throat/Head and Neck Surgery, Maastricht University Medical Center, Maastricht, The Netherlands
- Department of Neurology, Maastricht University Medical Center, Maastricht, The Netherlands
- Department of Neurosurgery, Maastricht University Medical Center, Maastricht, The Netherlands
- Department of Neurology, Academic Medical Center, Amsterdam, The Netherlands
- Department of Neurosurgery, Academic Medical Center, Amsterdam, The Netherlands
- Department of Neurology, Haga Teaching Hospital, The Hague, The Netherlands
Correspondence Address:
Jasper V. Smit
Department of Ear Nose and Throat/Head and Neck Surgery, Maastricht University Medical Center, Maastricht, The Netherlands
DOI:10.4103/2152-7806.194156
Copyright: © 2016 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Jasper V. Smit, Marcus L. F. Janssen, Malou Engelhard, Rob M. A. de Bie, P. Richard Schuurman, Maria F. Contarino, Arne Mosch, Yasin Temel, Robert J. Stokroos. The impact of deep brain stimulation on tinnitus. 14-Nov-2016;7:
How to cite this URL: Jasper V. Smit, Marcus L. F. Janssen, Malou Engelhard, Rob M. A. de Bie, P. Richard Schuurman, Maria F. Contarino, Arne Mosch, Yasin Temel, Robert J. Stokroos. The impact of deep brain stimulation on tinnitus. 14-Nov-2016;7:. Available from: http://surgicalneurologyint.com/surgicalint_articles/the-impact-of-deep-brain-stimulation-on-tinnitus/
Abstract
Background:Tinnitus is a disorder of the nervous system that cannot be adequately treated with current therapies. The effect of neuromodulation induced by deep brain stimulation (DBS) on tinnitus has not been studied well. This study investigated the effect of DBS on tinnitus by use of a multicenter questionnaire study.
Methods:Tinnitus was retrospectively assessed prior to DBS and at the current situation (with DBS). From the 685 questionnaires, 443 were returned. A control group was one-to-one matched to DBS patients who had tinnitus before DBS (n = 61). Tinnitus was assessed by the tinnitus handicap inventory (THI) and visual analog scales (VAS) of loudness and burden.
Results:The THI decreased significantly during DBS compared to the situation prior to surgery (from 18.9 to 15.1, P P = 0.50) and other DBS targets did not change. The VAS loudness increased in the control group (5.4 to 6.0 P
Conclusion:DBS might have a modulatory effect on tinnitus. Our study suggests that DBS of the STN may have a beneficial effect on tinnitus, but most likely other nuclei linked to the tinnitus circuitry might be even more effective.
Keywords: Deep brain stimulation, neuromodulation, survey study, tinnitus
INTRODUCTION
Tinnitus, also known as ringing in the ears, is defined as a perception of sound when no actual external sound is present. The prevalence of frequent and burdensome tinnitus is 10–15% and has increased over the last decennia.[
Deep brain stimulation (DBS) can be applied within neuronal networks to reduce the pathological neuronal activity.[
Our goal was to assess the severity of tinnitus before and during DBS in patients who were treated with DBS in a broad spectrum of targets and for various indications. For this purpose, we conducted a multicenter survey study in a large DBS patient cohort. We carefully matched this group with a control cohort that contained tinnitus patients without DBS.
MATERIALS AND METHODS
Patient characteristics
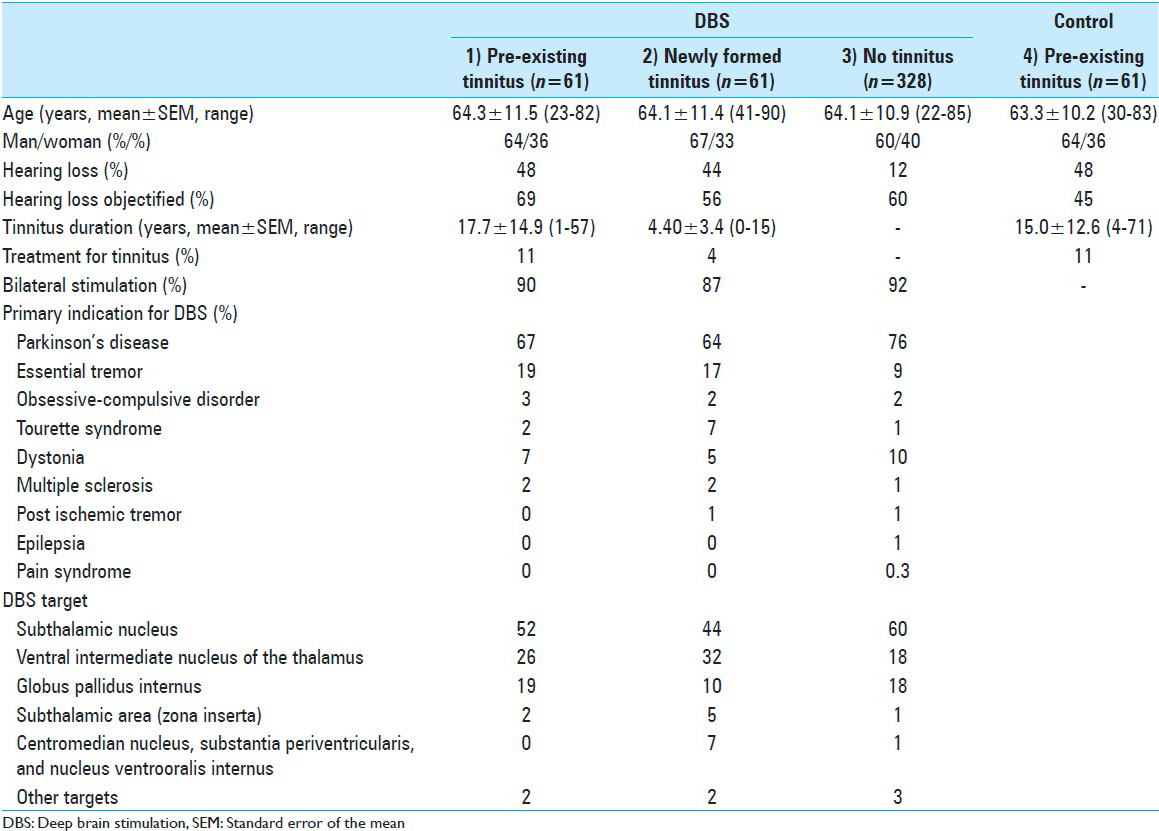
All patients who underwent DBS surgery in the Maastricht University Medical Center, Amsterdam Medical Center or the Haga Teaching Hospital (The Hague) were included in this study and received a questionnaire by mail. The first part of the questionnaire consisted of questions regarding the situation before DBS surgery (t = 0) and the second part consisted of questions regarding the current situation with DBS (t = 1).
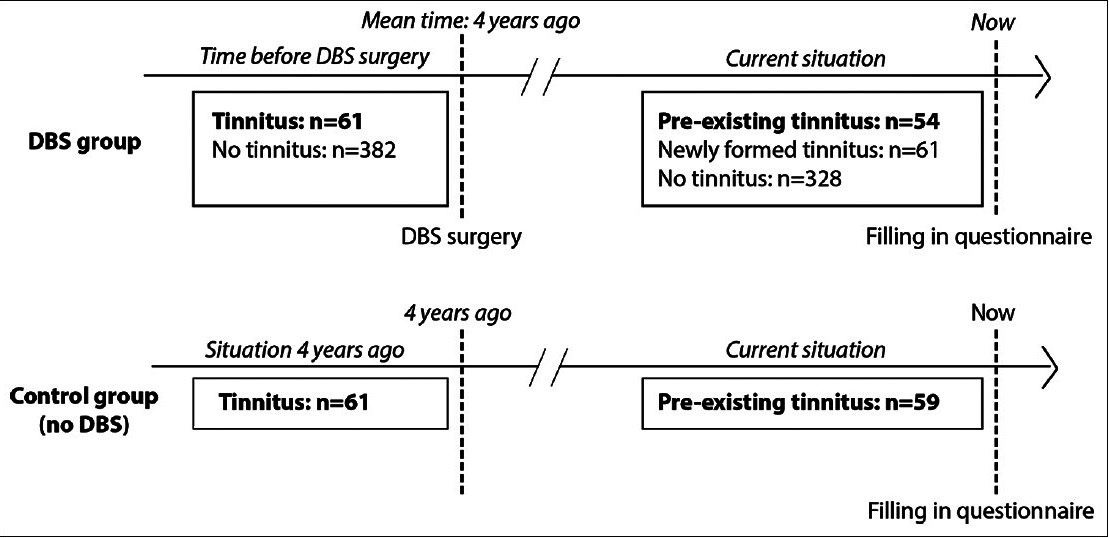
After analyzing the patient characteristics of the DBS patients, we gathered a control group by advertisements among patients known to have tinnitus to retrospectively examine the natural course of tinnitus over time. These tinnitus patients were asked to fill out the same questionnaire online, which consisted of questions regarding the situation 4 years ago (t = 0) and the current situation (t = 1). The 4-year interval was based on the mean time between DBS surgery and completion of the questionnaire in the DBS group, which was 3.9 ± 2.9 years. Control patients were matched with the 61 DBS patients who declared to have had tinnitus before surgery, in a matched subject design for the dichotomous variables gender, tinnitus treatment (e.g., medication or cognitive therapy) and self-reported hearing loss (“yes, measured by audiometry,” “yes, noticed by my relatives or myself,” “no.”). From the available matched controls, the control subjects that matched age the best were chosen. Informed consents were obtained from all patients. No ethical approval was required for this type of questionnaire study according to local regulations.
Study design
Both parts of the questionnaire included general questions about tinnitus (duration, received treatment, etc.), hearing loss (objective or subjective), and three tinnitus severity measures. The main tinnitus outcome measure was the tinnitus handicap inventory (THI), which consists of 25 questions and reflects the impact of tinnitus on daily living. For each item or situation, subjects were asked to fill in one of the following responses: “Yes” (4 points), “sometimes” (2 points), or “no” (0 points), resulting in a maximum burden score of 100. Furthermore, the questionnaire included a visual analog scale (VAS) of tinnitus loudness (0 is no loudness and 10 is being the most loud tinnitus) and a VAS of burden (0 is no burden and 10 is the most burdening tinnitus).
For all measures, the absolute mean at t = 0 and t = 1 was calculated for the within-group comparisons. Because of the baseline differences between the DBS and control group, between-group comparison was based on the mean of the relative change in percentage from each subject, expressed as 100.
In the group of patients that only experienced tinnitus after DBS surgery, the 5-year incidence was calculated and compared with the literature.
Statistics
We used a Student's t-test for normally distributed data and the Wilcoxon signed-rank test for nonparametric data. The within-subjects comparisons were analyzed in a paired manner. P values less than 0.05 were considered statistically significant. All data were analyzed by using the Statistical Package for the Social Sciences (Version 20, IBM, Somers, NY, USA).
RESULTS
Patient characteristics
Questionnaires were sent to 685 DBS patients. Six patients or their relatives wrote that they were not able to fill out the form because of mental or physical problems. A return rate of filled-out questionnaires of 65% (n = 443) was achieved.
From the responders, 61 (14%) had tinnitus before DBS (group 1), 61 (14%) had newly formed tinnitus following DBS surgery (group 2), and 328 DBS patients (72%) did not experience tinnitus at all (group 3) [
Figure 1
Scheme of the study design. Deep brain stimulation (DBS) patients were asked to fill in a questionnaire about the situation before DBS (mean duration: 3.9 years ago) and about the current situation. In a matched-subject design, the control group was matched to those patients of the DBS group who had pre-existing tinnitus. Patients from the control group were first asked to fill in a tinnitus questionnaire regarding their situation 4 years ago (similar to the situation of the DBS group). Second, the patients were asked to fill in the questionnaire for the current situation. Note that 7 patients who had tinnitus prior to DBS surgery did not have tinnitus during DBS. In the control group, 2 patients only experienced tinnitus 4 years ago
Table 1
Characteristics of patients with pre-existing tinnitus before deep brain stimulation (DBS) (1), newly formed tinnitus during DBS (2), no tinnitus (3) and a control group with pre-existing tinnitus (4), that was matched to group 1. Matching variables were age, gender, hearing loss, and treatment. Other targets (last row) include combinations of subthalamic nucleus and ventral intermediate nucleus of the thalamus, nucleus accumbens, and posterior hypothalamus
Tinnitus outcome
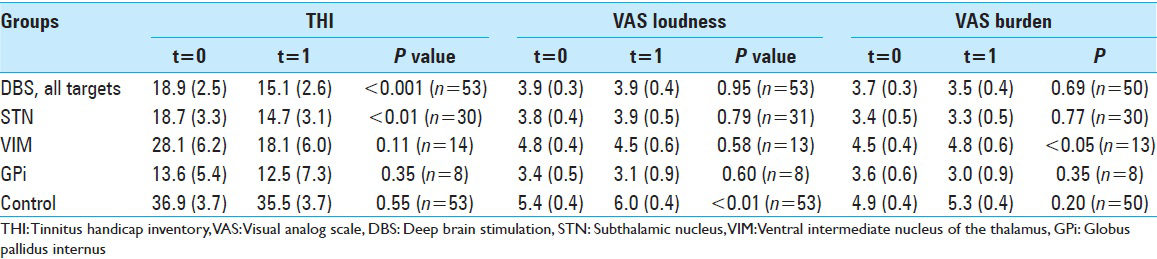
From the 61 patients who experienced tinnitus before DBS surgery, 7 were completely tinnitus-free at the time of filling the questionnaire. The THI decreased significantly compared to the situation before DBS surgery (from 18.9 to 15.1) whereas the THI in the control group did not significantly change (36.9 to 35.5). With respect to the THI in the DBS group, 53% improved, 36% did not change, and 11% worsened. In the control group 54% improved, 7% did not change, and 39% worsened.
For the patients with DBS, the reported loudness and burden on a VAS scale did not change significantly (3.9 to 3.9 and 3.7 to 3.5, respectively). The control group showed a significant increase in VAS loudness (5.4 to 6.0) and a non-significant increase in VAS burden (4.9 to 5.3). Detailed results with P values are shown in
Table 2
The THI, VAS loudness, and VAS burden of tinnitus before (t=0) and during DBS (t=1). Measurements were performed for all DBS patients and in subgroup analyses that consisted of only patients with electrodes implanted in the STN, VIM or GPi. Note that the control group did not receive DBS. The mean is shown, with the standard error of the mean between brackets
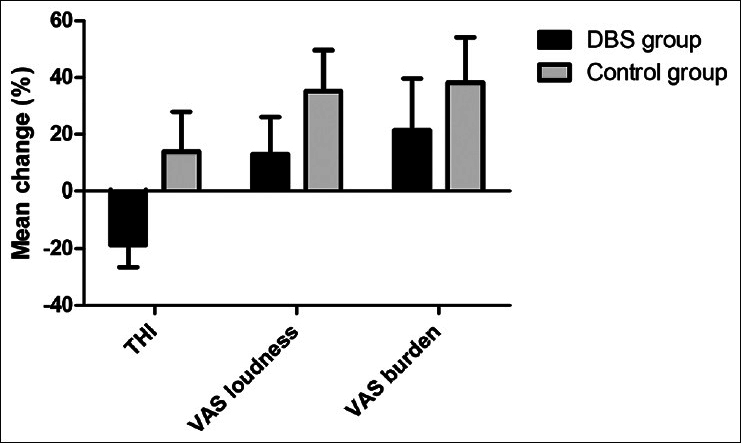
Relative measures were used to compare the DBS group to the control group. The relative mean difference of the THI was −19% for the DBS group and +14% for the control group. In the DBS and control group, VAS loudness increased with 13% and 35% and VAS burden increased with 21% and 38%, respectively. Relative measures of the three outcome measures are visualized in
Figure 2
The mean of the relative change (in percentage) is calculated for the tinnitus handicap inventory, visual analog scale (VAS) loudness, and VAS burden. A positive number indicates an increase and a negative number a decrease in the outcome. Error bars indicate the absolute standard error of the mean
Subgroup analyses
For the DBS group with existing tinnitus, the mean values of the THI were calculated for the most common targets: STN (n = 31), the VIM (n = 17), and the GPi (n = 11). The STN was the only target that showed a statistically significant decrease in THI (18.7 to 14.7), see
The primary indication for DBS, which is not independent from the target subgroup analysis, was subdivided in PD, ET, and others. When analyzing the subgroups for primary indication, only in the group with PD, a significant difference in mean THI difference was seen (18.6 to 14.8, P < .01, n = 34).
On comparing males and females, only males showed a significant difference in THI (20.1 to 15.4, P <.001, n = 34). Females did not show a significant difference (16.7 to 14.6, P = 0.26, n = 19).
Both subjects who received treatment for tinnitus (e.g., medication or cognitive therapy), as subjects who did not receive treatment for tinnitus showed a significant decrease of THI (17.6 to 14.2, P < .01, n = 40 and 35.4 to 27.4, P < .05, n = 7, respectively).
The only subgroup analysis that showed a significant difference in the VAS scale was VIM as a DBS target; VAS loudness increased from 4.5 to 4.7 (P < .05, n = 13).
Newly formed tinnitus
The 5-year incidence of tinnitus is based on the percentage of subjects who developed tinnitus after DBS. From the 200 patients with 5-year follow-up, 21 developed tinnitus after DBS surgery, all with STN as the target. The 5-year incidence is 10.5%. From all subjects who developed tinnitus following DBS, 6 patients did not fill in the onset date of tinnitus and were excluded from analysis.
DISCUSSION
The present study showed that the THI reduced significantly in patients with DBS, whereas it did not significantly change in the control group that represented the natural course of tinnitus over time. In the patients who received DBS, the relative change of the THI was a decrease of 19%, whereas the relative change in the control group was an increase of 14%. During DBS, there was no change on the VAS loudness and burden, whereas the VAS loudness increased significantly in the control group.
The results from the subgroup analysis showed that the STN, which is the most common DBS target for PD, is the only target that reached statistical significance in the within-subject comparison of the THI. The VIM showed a large decrease of the THI (28.1 to 18.1), which might not have reached significance due to the small sample size (n = 14). The heterogeneity of the study population could lead to an underestimation of the effectiveness of DBS on tinnitus.
It was not our purpose in this study to assess the clinical relevance of performing DBS for tinnitus. In this study, tinnitus was assessed in patients who did not seek for treatment of their tinnitus by way of DBS. Therefore, the baseline THI is relatively low and therefore a clinical relevant decrease of 7 points on the THI[
To the best of our knowledge, the effect of STN stimulation on tinnitus has not been assessed before. The influence on tinnitus by VIM stimulation has been reported in another study.[
As far as we know, no other study retrospectively or prospectively assessed the natural course of tinnitus over time with absolute values of tinnitus severity. Some studies only presented the presence of tinnitus[
DBS has good therapeutic effects in PD and ET, both on motor outcome and the quality of life. The effect of DBS on tinnitus could be explained by interference with the tinnitus-related neuronal network. In the present study, the STN showed the best outcome in tinnitus reduction. The STN, which is not directly linked to auditory nuclei, is subdivided in a motor, associative, and limbic part. The STN, however, is connected to the nucleus accumbens, which has been implicated to play a role in tinnitus.[
Although only one case study reported tinnitus as a side effect of DBS (in the VIM),[
One might conclude that the STN is a potential target to treat tinnitus by DBS. However, this conclusion needs to be tempered. The targets investigated by this study are clinically used as a target for other primary indications than tinnitus. Therefore, it seems reasonable to think that stimulation of brain areas that are more directly involved in the neural network involved in tinnitus will be more effective. One of the promising targets is the dorsal cochlear nucleus. Stimulation of this target has already been shown to attenuate tinnitus in an animal study.[
The main limitation of this is study is the retrospective design of the questionnaire which could be prone to “recall bias.” It could be difficult to remember tinnitus characteristics from the past and patients may tend to have a better recall on past exposures than controls.[
Furthermore, to strengthen the design of the study, we used a control group. To match the DBS patients as much as possible with the controls, we used a one-to-one matching strategy with multiple matching variables. The control group consisted of subjects from the general population who suffered from tinnitus. Despite the fact that some studies reported an abnormal hearing in PD and ET patients,[
This study is one of the first steps in exploring the feasibility and target specificity of DBS as a future treatment for tinnitus. The present study indicates that DBS might reduce the handicap that is caused by tinnitus. Furthermore, while the tinnitus loudness remained the same in the DBS group, it increased in the control group. Stimulation of the STN resulted in the most beneficial effect on tinnitus, however, stimulation of other nuclei that are directly linked to the tinnitus circuitry might be even more effective.
Financial support and sponsorship
Heinsius Houbolt Foundation.
Conflicts of interest
There are no conflicts of interest.
Acknowledgements
We would like to thank Miranda Janssen, statistician, for her statistical support.
References
1. Andersson G, Vretblad P, Larsen HC, Lyttkens L. Longitudinal follow-up of tinnitus complaints. Arch Otolaryngol Head Neck Surg. 2001. 127: 175-9
2. Cheung SW, Larson PS. Tinnitus modulation by deep brain stimulation in locus of caudate neurons (area LC). Neuroscience. 2010. 169: 1768-78
3. Chiappa KH, Chiappa KH.editors. Short-latency somatosensory evoked potentials: Interpretation. Evoked Potentials in Clinical Medicine. New York: Lippincott-Raven; 1997. p. 199-250
4. Coughlin SS. Recall bias in epidemiologic studies. J Clin Epidemiol. 1990. 43: 87-91
5. Davis A, ElRafaie A, Tyler RS.editors. Epidemiology of tinnitus. Tinnitus Handbook. San Diego: Singular; 2000. p. 1-23
6. Eggermont JJ. Central tinnitus. Auris Nasus Larynx. 2003. 30: S7-12
7. Fradis M, Samet A, Ben-David J, Podoshin L, Sharf B, Wajsbort J. Brainstem auditory evoked potentials to different stimulus rates in parkinsonian patients. Eur Neurol. 1988. 28: 181-6
8. Kaltenbach JA. Tinnitus: Models and mechanisms. Hear Res. 2011. 276: 52-60
9. Kloostra FJ, Arnold R, Hofman R, Van Dijk P. Changes in tinnitus after cochlear implantation and its relation with psychological functioning. Audiol Neurootol. 2015. 20: 81-9
10. Landgrebe M, Azevedo A, Baguley D, Bauer C, Cacace A, Coelho C. Methodological aspects of clinical trials in tinnitus: A proposal for an international standard. J Psychosom Res. 2012. 73: 112-21
11. Larson PS, Cheung SW. Deep brain stimulation in area LC controllably triggers auditory phantom percepts. Neurosurgery. 2012. 70: 398-405
12. Larson PS, Cheung SW. A stroke of silence: Tinnitus suppression following placement of a deep brain stimulation electrode with infarction in area LC. J Neurosurg. 2013. 118: 192-4
13. Luo H, Zhang X, Nation J, Pace E, Lepczyk L, Zhang J. Tinnitus suppression by electrical stimulation of the rat dorsal cochlear nucleus. Neurosci Lett. 2012. 522: 16-20
14. Martin WH, Shi Y-B, Buciel KJ, Anderson VC. Deep brain stimulation effects on hearing function and tinnitus. Sixth International Tinnitus Seminar. 1999. p.
15. McIntyre CC, Hahn PJ. Network perspectives on the mechanisms of deep brain stimulation. Neurobiol Dis. 2010. 38: 329-37
16. Nondahl DM, Cruickshanks KJ, Huang GH, Klein BE, Klein R, Tweed TS. Generational differences in the reporting of tinnitus. Ear Hearing. 2012. 33: 640-4
17. Nondahl DM, Cruickshanks KJ, Wiley TL, Klein R, Klein BE, Tweed TS. Prevalence and 5-year incidence of tinnitus among older adults: The epidemiology of hearing loss study. J Am Acad Audiol. 2002. 13: 323-31
18. Norena AJ. An integrative model of tinnitus based on a central gain controlling neural sensitivity. Neurosci Biobehav Rev. 2011. 35: 1089-109
19. Ondo W, Dat Vuong K, Almaguer M, Jankovic J, Simpson RK. Thalamic deep brain stimulation: Effects on the nontarget limbs. Mov Disord. 2001. 16: 1137-42
20. Ondo WG, Sutton L, Dat Vuong K, Lai D, Jankovic J. Hearing impairment in essential tremor. Neurology. 2003. 61: 1093-7
21. Prasher D, Bannister R. Brain stem auditory evoked potentials in patients with multiple system atrophy with progressive autonomic failure (Shy-Drager syndrome). J Neurol Neurosurg Psychiatry. 1986. 49: 278-89
22. Rosenhall U, Karlsson AK. Tinnitus in old age. Scand Audiol. 1991. 20: 165-71
23. Rubenstein B, Ö T, Rosenhall U. Longitudinal fluctuations in tinnitus as reported by an elderly population. J Audiol Med. 1992. 1: 149-55
24. Schwartz CE, Bode R, Repucci N, Becker J, Sprangers MA, Fayers PM. The clinical significance of adaptation to changing health: A meta-analysis of response shift. Qual Life Res. 2006. 15: 1533-50
25. Shi Y, Burchiel KJ, Anderson VC, Martin WH. Deep brain stimulation effects in patients with tinnitus. Otolaryngol Head Neck Surg. 2009. 141: 285-7
26. Smit JV, Janssen ML, Schulze H, Jahanshahi A, Van Overbeeke JJ, Temel Y. Deep brain stimulation in tinnitus: Current and future perspectives. Brain Res. 2015. 1608: 51-65
27. Stouffer JL, Tyler RS. Characterization of tinnitus by tinnitus patients. J Speech Hearing Disord. 1990. 55: 439-53
28. Sullivan MD, Katon W, Dobie R, Sakai C, Russo J, Harrop-Griffiths J. Disabling tinnitus. Association with affective disorder. Gen Hosp Psychiatry. 1988. 10: 285-91
29. van Zwieten G, Smit JV, Jahanshahi A, Temel Y, Stokroos RJ. Tinnitus: Is there a place for brain stimulation?. Surg Neurol Int. 2016. 10: S125-9
30. Vitale C, Marcelli V, Allocca R, Santangelo G, Riccardi P, Erro R. Hearing impairment in Parkinson's disease: Expanding the nonmotor phenotype. Mov Disord. 2012. 27: 1530-5
31. Yardley L, Dibb B. Assessing subjective change in chronic illness: An examination of response shift in health-related and goal-oriented subjective status. Psychol Health. 2007. 22: 813-28
32. Yylmaz S, Karaly E, Tokmak A, Guclu E, Kocer A, Ozturk O. Auditory evaluation in Parkinsonian patients. Eur Arch Otorhinolaryngol. 2009. 266: 669-71
33. Zeman F, Koller M, Figueiredo R, Aazevedo A, Rates M, Coelho C. Tinnitus handicap inventory for evaluating treatment effects: Which changes are clinically relevant?. Otolaryngol Head Neck Surg. 2011. 145: 282-7