- Department of Neurosurgery, Hospital das Clínicas da Faculdade de Medicina da Universidade de São Paulo, Brazil.
- Department of Neurology, University of São Paulo, Brazil.
- Department of Neurosurgery, University of São Paulo, São Paulo, Brazil.
Correspondence Address:
Iuri Santana Neville
Department of Neurosurgery, Hospital das Clínicas da Faculdade de Medicina da Universidade de São Paulo, Brazil.
DOI:10.25259/SNI_476_2020
Copyright: © 2020 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Joao Paulo Mota Telles1, Vitor Nagai Yamaki1, Renata Gobbato Yamashita1, Davi Jorge Fontoura Solla1, Wellingson Silva Paiva2, Manoel Jacobsen Teixeira3, Iuri Santana Neville1. The impact of urgent neurosurgery on the survival of cancer patients. 21-Aug-2020;11:258
How to cite this URL: Joao Paulo Mota Telles1, Vitor Nagai Yamaki1, Renata Gobbato Yamashita1, Davi Jorge Fontoura Solla1, Wellingson Silva Paiva2, Manoel Jacobsen Teixeira3, Iuri Santana Neville1. The impact of urgent neurosurgery on the survival of cancer patients. 21-Aug-2020;11:258. Available from: https://surgicalneurologyint.com/?post_type=surgicalint_articles&p=10218
Abstract
Background: Patients with cancer are subject to all neurosurgical procedures of the general population, even if they are not directly caused by the tumor or its metastases. We sought to evaluate the impact of urgent neurosurgery on the survival of patients with cancer.
Methods: We included patients submitted to neurosurgeries not directly related to their tumors in a cancer center from 2009 to 2018. Primary endpoints were mortality in index hospitalization and overall survival.
Results: We included 410 patients, 144 went through elective procedures, functional (26.4%) and debridement (73.6%) and 276 urgent neurosurgeries were performed: one hundred and sixty-three ventricular shunts (59%), and 113 intracranial hemorrhages (41%). Median age was 56 (IQR = 24), 142 (51.4%) of patients were metastatic, with 101 (36.6%) having brain metastasis. In 82 (33.7%) of the urgent surgeries, the patient died in the same admission. Urgent surgeries were associated with mortality in index hospitalization (OR 3.45, 95% CI 1.93–6.15), as well as non-primary brain tumors (OR 3.13, 95% CI 1.48–6.61). Median survival after urgent surgeries was 102 days, compared to 245 days in the control group (Log rank, P
Conclusion: Conditions that require urgent neurosurgery in patients with cancer have a very poor prognosis. We present concrete data on the magnitude of several factors that need to be taken into account when deciding whether or not to recommend surgery.
Keywords: Brain neoplasms, Neoplasm metastasis, Neurosurgery, Survival analysis
INTRODUCTION
Patients with cancer can be submitted to neurosurgeries for multiple reasons not directly related to their oncologic disease. Cerebrovascular diseases and hydrocephalus are the most common central nervous system (CNS) complications that require urgent neurosurgeries in patients with cancer.[
The patients in oncologic centers are vulnerable to hemorrhagic complications, such as intratumoral hemorrhage, spontaneous bleeding from coagulopathies, or leukostasis in hematologic neoplasias. Hydrocephalus is often related to obstructive mass effect, infectious complications, or carcinomatous meningitis. Nonetheless, any patient with a given tumor can present to the hospital with neurological urgencies (i.e., subdural hematoma), just as any patient who does not have cancer. Other neurosurgeries indicated for this group include debridement and functional procedures for pain, for example.
In the particular case of urgent procedures, decision-making based on prognosis prediction for those non-oncologic complications is challenging and must be done immediately in the emergency room.[
In light of this, we reviewed neurosurgeries performed for non-oncological purposes in patients with cancer over 10 years in an oncologic center. We aimed to evaluate and, if possible, to quantify outcomes of those interventions in patient survival.
MATERIALS AND METHODS
We retrospectively reviewed the medical records of patients that underwent urgent neurosurgeries for non-oncological complications from 2009 to 2018 at the Instituto do Cancer do Estado de São Paulo (ICESP), a reference tertiary cancer center. Brain tumor resections, spine surgeries, and early postoperative complications were excluded from the study. This study was approved by the local Ethics and Research Committee.
We analyzed the outcomes of those surgeries in search of prognostic factors. Each surgery was entered as an individual observation. For comparison purposes, we separated groups of patients with cancer who underwent elective neurosurgeries unrelated to the tumor, such as debridement or functional procedures, during the same period.
Information on age, gender, primary tumor site (primary of the CNS or other tumors), histopathologic diagnosis, and presence of metastasis were collected. Surgeries were categorized as ventricular shunts – external ventricular derivation and ventriculoperitoneal shunts – or intracranial hemorrhage evacuations. The follow-up was recorded based on days until death or the last outpatient appointment.
The primary endpoints were survival and mortality in index hospitalization. In-hospital mortality was studied using logistic regression, and survival was studied using Kaplan– Meier survival curves, log-rank tests, and Cox proportional hazards regressions. All analyses were performed both with and without the control group. Variables presenting a P = 0.05 in the univariate models were included as covariates in the final multivariable models. Results are presented as odds ratio (95% confidence interval) for logistic regressions, and Hazard Ratio (95% confidence interval) for Cox regressions.
Data are presented as mean (standard deviation) for normally distributed variables, median (interquartile range) for other continuous variables, and frequencies (%) for categorical variables. Linearity and proportional hazards assumptions were verified graphically through the Schoenfeld residuals. Statistical significance was considered as P < 0.05 in the multivariable models. Analyses were performed using Python 3.7.0 (Python Software Foundation, Beaverton, Oregon, USA) and GraphPad Prism version 8.3.0 for macOS (GraphPad, La Jolla, California, USA).
RESULTS
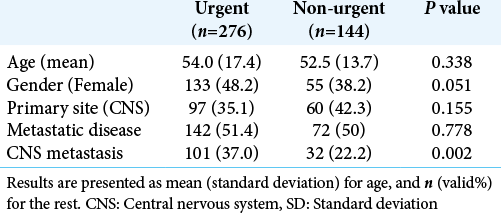
We included 276 non-oncologic urgent neurosurgeries performed over 10 years. Ventricular shunts accounted for 163 (59.1%), while intracranial hematoma (IC) evacuations accounted for 113 (40.9%). The median patient age was 56 years (interquartile range 24), and 51.8% were male. Overall, 142 patients had metastatic cancer (51.4%), while 101 (71.1%) of those were CNS metastasis. More than one procedure was performed in 24 patients, which accounted for 56 surgeries, or 20.3% of the total. The mean follow- up was 335.6 days (±491.5 days). Surgeries for non-urgent conditions included 38 functional procedures (26.4%) and 106 debridements (73.6%). [
Mortality in index hospitalization
In 82 (33.7%) of the cases, the patient undergoing urgent neurosurgery died in the course of the same hospitalization. [
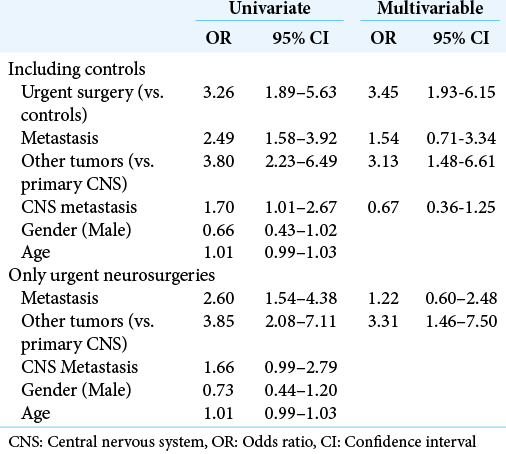
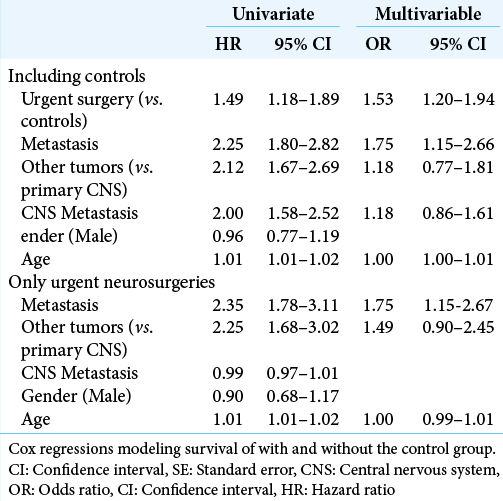
In univariate analyses including the non-urgent group, mortality in index hospitalization was associated with urgent surgeries (OR 3.26 [1.89–5.63]), presence of any metastasis (OR 2.49 [1.58–3.92]), other tumor sites compared to primary CNS (OR 3.80 [2.23–6.49]), and presence of CNS metastasis (OR 1.70 [1.01–2.67]). In the multivariable analysis, intra-hospital mortality was predicted by urgent surgeries (OR 3.45 [1.93–6.15]), and other tumors compared to primary CNS (OR 3.13 [1.48–6.61]).
When looking exclusively at urgent neurosurgeries, the predictors of intra-hospital mortality were the presence of metastasis (OR 2.60, 95% CI [1.54–4.38]), and other tumors compared to primary CNS (OR 3.85 [2.08–7.11]). In the multivariable model, only other tumors predicted mortality compared to primary CNS malignancies (OR 3.31 [1.46–7.50]).
Survival analysis
[
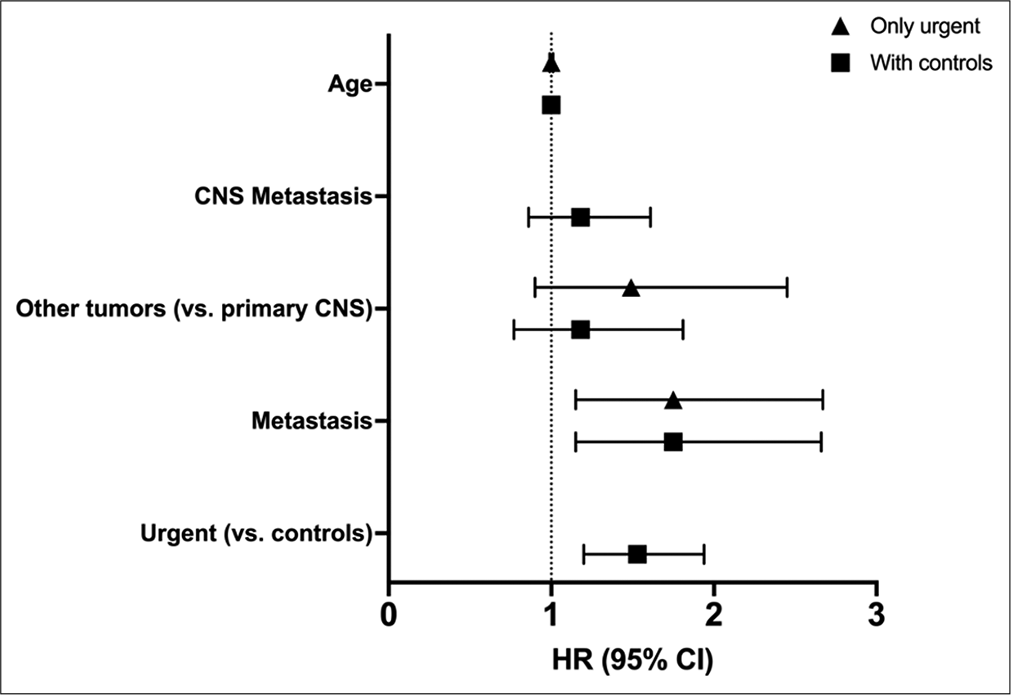
Figure 2:
Predictors of survival after non-oncologic neurosurgery for cancer patients Forest plot showing the results of the two multivariable Cox models - with and without control groups – for assessment of survival predictors in cancer patients submitted to neurosurgery with purposes not directly linked to the tumor. HR: Hazard ratio, CI: Confidence interval.
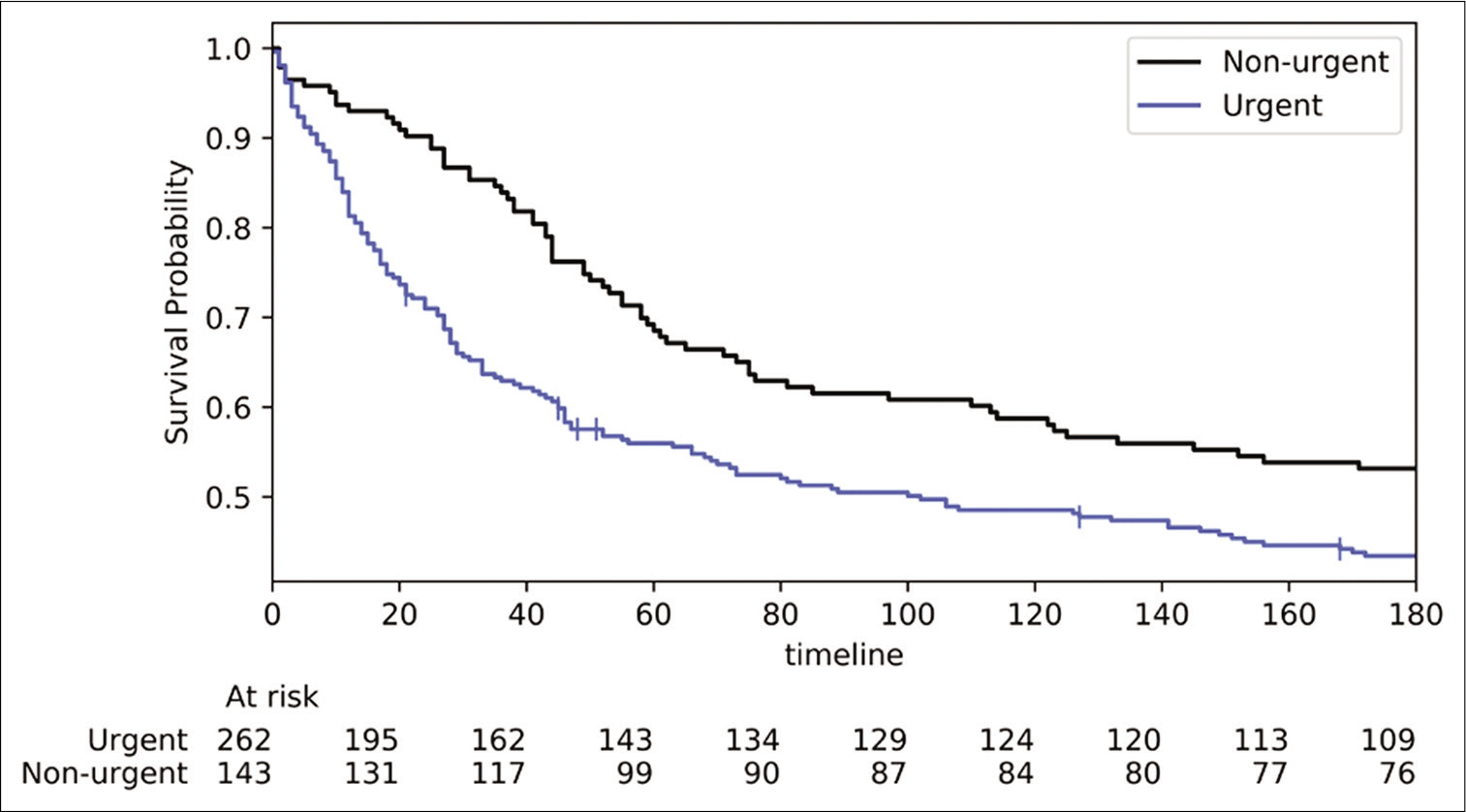
In the univariate Cox regressions including non-urgent, the factors associated with survival were urgent surgeries (HR 1.49 [1.18–1.89]), presence of any metastasis (HR 2.25 [1.80–2.82]), other tumors compared to primary CNS (HR 2.12 [1.67–2.69]), presence of CNS metastasis (HR 2.00 [1.58–2.52]), and each additional year of age (HR 1.01 [1.01–1.02]). The final multivariable model confirmed significant values only for urgent surgeries (HR 1.53 [1.20–1.94]), and presence of any metastasis (HR 1.75 [1.15–2.66]).
Within the urgent surgeries group, univariate models reached significance thresholds for the presence of any metastasis, HR 2.35 (1.78–3.11), other tumors compared to primary CNS, HR 2.25 (1.68–3.02), and each additio nal year of age, HR 1.01 (1.01–1.02). On the final multivariable regression, only metastasis was significant, HR 1.75 (1.15–2.67).
DISCUSSION
Neurosurgical procedures are performed in patients with cancer for reasons other than the tumor. In those cases, neurosurgical urgency often represents a catastrophic event. In the context of advanced metastatic disease, both families and caregivers can find robust, compelling evidence supporting the choice for palliative care instead of invasive, often pointless treatments. However, in acute complications such as intracranial hemorrhages, it is impossible to predict functionality after treatment, and most patients are submitted to urgent surgery despite the limited prognosis.[
Our analyses focused on patients with cancer who experienced “non-oncologic” CNS complications, and the group submitted to urgent surgical treatments received special attention. In some way, we used patients with cancer undergoing elective neurosurgical procedures as a control group. The rationale behind this was to control for inherent risks associated with neurosurgical procedures, isolating effects of the urgency itself on the outcomes.
Patients with cancer undergoing urgent neurosurgeries had a very high mortality rate. Their median survival was 102 days, and mortality in the index admission occurred after 33.7% of surgeries. A visual inspection of the Kaplan–Meier curves shows that this intra-hospital mortality accounts for a steep decrease in the probability of survival during the 1st day. Most of the in-hospital mortality occurred in the 1st postoperative day as a consequence of severe CNS complications. Bosscher et al., 2015, studied emergency consultations in general surgery for patients with cancer, reporting a 30-day mortality rate of only 13%.[
The analyses show that the presence of systemic metastases might be the most crucial aspect to predict mortality. In the model including the electives, who also had cancer and went through neurosurgical procedures, the hazard ratio associated with urgent surgeries was 1.53 (1.20–1.94), while the one associated with presence of any metastasis was 1.75 (1.15–2.66). Effectively, to decide to operate, the patient’s oncological status might be more important than the severity of the urgency.
The high prevalence of metastatic cancer in our analysis could, therefore, explain the high mortality encountered. The metastatic disease represents a systemic condition with widespread inflammation and metabolic disturbance. Therefore, those patients are sometimes too fragile to support invasive procedures. Besides, coagulopathy is not rare in patients with metastasis, advanced stage tumors, or hematological malignancies. In a previous series with more than 200 patients with cancer and intracranial hemorrhage, 46% of patients presented spontaneous bleeding secondary to coagulopathy.[
Primary CNS tumors presented better prognosis compared to other solid tumors in our analysis, which is consistent with literature findings for intracranial hemorrhage.[
Our study presents several limitations, for it is a retrospective view of a rather heterogeneous group of patients with multiple different clinical pictures. Regarding the elective group, there were fewer patients with CNS metastasis, which might have certainly influenced the results. Randomized controlled studies are required for better determination of the prognosis of each specific condition. However, our analysis presents unique data on the prognosis of patients with cancer submitted to neurosurgical procedures. Hopefully, our results will aid in weighting the clinical variables to be considered in decision-making in such delicate, life-threatening scenarios.
CONCLUSION
Patients with cancer can be submitted to any kind of neurosurgery. Conditions that require urgent neurosurgery in patients with cancer have a very poor prognosis. We present concrete data on the magnitude of several factors that need to be taken into account when deciding whether or not to recommend surgery. Primary CNS tumors present better prognosis when compared to other primary sites and metastatic disease.
Declaration of patient consent
Institutional Review Board (IRB) permission obtained for the study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Achrol AS, Rennert RC, Anders C, Soffietti R, Ahluwalia MS, Nayak L. Brain metastases. Nat Rev Dis Primers. 2019. 5: 5
2. Aguilar MI, Freeman WD. Spontaneous intracerebral hemorrhage. Semin Neurol. 2010. 30: 555-64
3. Barbera L, Paszat L, Chartier C. Indicators of poor quality end-of-life cancer care in Ontario. J Palliat Care. 2006. 22: 12-7
4. Barbera L, Taylor C, Dudgeon D. Why do patients with cancer visit the emergency department near the end of life?. Can Med Assoc J. 2010. 182: 563-8
5. Bitoh S, Hasegawa H, Ohtsuki H, Obashi J, Fujiwara M, Sakurai M. Cerebral neoplasms initially presenting with massive intracerebral hemorrhage. Surg Neurol. 1984. 22: 57-62
6. Bosscher MR, Van Leeuwen BL, Hoekstra HJ. Current management of surgical oncologic emergencies. PLoS One. 2015. 10: e0124641
7. Caine GJ, Stonelake PS, Lip GY, Kehoe ST. The hypercoagulable state of malignancy: Pathogenesis and current debate. Neoplasia. 2002. 4: 465-73
8. Fernández-García MÁ, Cantarín-Extremera V, Andión-Catalán M, Duat-Rodríguez A, Jiménez-Echevarría S, Bermejo-Arnedo I. Secondary intracranial hypertension in pediatric patients with leukemia. Pediatr Neurol. 2017. 77: 48-53
9. Hemphill JC, Greenberg SM, Anderson CS, Becker K, Bendok BR, Cushman M. Guidelines for the management of spontaneous intracerebral hemorrhage: A guideline for healthcare professionals from the American heart association/ American stroke association. Stroke. 2015. 46: 2032-60
10. Kase CS. Intracerebral hemorrhage: Non-hypertensive causes. Stroke. 1986. 17: 590-5
11. Khorchid YM, Malkoff M.editors. Intracranial hemorrhage focused on cancer and hemato-oncologic patients. Oncologic Critical Care. Cham: Springer International Publishing; 2019. p. 1-14
12. Kondziolka D, Bernstein M, Resch L, Tator CH, Fleming JF, Vanderlinden RG. Significance of hemorrhage into brain tumors: Clinicopathological study. J Neurosurg. 1987. 67: 852-7
13. Kyrnetskiy EE, Kun LE, Boop FA, Sanford RA, Khan RB. Types, causes, and outcome of intracranial hemorrhage in children with cancer. J Neurosurg. 2005. 102: 31-5
14. Licata B, Turazzi S. Bleeding cerebral neoplasms with symptomatic hematoma. J Neurosurg Sci. 2003. 47: 201-10
15. Little JR, Dial B, Bélanger G, Carpenter S. Brain hemorrhage from intracranial tumor. Stroke. 1979. 10: 283-8
16. Mazerand E, Gallet C, Pallud J, Menei P, Bernard F. Acute intracranial hypertension management in metastatic brain tumor: A French national survey. Neurochirurgie. 2019. 65: 348-56
17. McArdle CS, Hole DJ. Emergency presentation of colorectal cancer is associated with poor 5-year survival. Br J Surg. 2004. 91: 605-9
18. Navi BB, Reichman JS, Berlin D, Reiner AS, Panageas KS, Segal AZ. Intracerebral and subarachnoid hemorrhage in patients with cancer. Neurology. 2010. 74: 494-501
19. Porta M, Fernandez E, Belloc J, Malats N, Gallén M, Alonso J. Emergency admission for cancer: A matter of survival?. Br J Cancer. 1998. 77: 477-84
20. Rogers LR. Cerebrovascular complications in cancer patients. Neurol Clin. 2003. 21: 167-92
21. Schrader B, Barth H, Lang EW, Buhl R, Hugo HH, Biederer J. Spontaneous intracranial haematomas caused by neoplasms. Acta Neurochir (Wien). 2000. 142: 979-85
22. Schwarzbach CJ, Schaefer A, Ebert A, Held V, Bolognese M, Kablau M. Stroke and cancer: The importance of cancer-associated hypercoagulation as a possible stroke etiology. Stroke. 2012. 43: 3029-34
23. Van Asch CJ, Luitse MJ, Rinkel GJ, Van Der Tweel I, Algra A, Klijn CJ. Incidence, case fatality, and functional outcome of intracerebral haemorrhage over time, according to age, sex, and ethnic origin: A systematic review and meta-analysis. Lancet Neurol. 2010. 9: 167-76
24. Vandyk AD, Harrison MB, Macartney G, Ross-White A, Stacey D. Emergency department visits for symptoms experienced by oncology patients: A systematic review. Support Care Cancer. 2012. 20: 1589-99