Transnasal endoscopic removal of a retrochiasmatic cavernoma: A case report and review of literature
- Neurosurgery Unit, Department of Surgical Sciences, Fondazione IRCCS Policlinico San Matteo, Pavia, Italy
- Neurosurgery Unit, Department of Clinical-Surgical, Diagnostic and Pediatric Sciences, University of Pavia, viale Golgi 19, Pavia, Italy
Correspondence Address:
Sabino Luzzi
Neurosurgery Unit, Department of Surgical Sciences, Fondazione IRCCS Policlinico San Matteo, Pavia, Italy
Neurosurgery Unit, Department of Clinical-Surgical, Diagnostic and Pediatric Sciences, University of Pavia, viale Golgi 19, Pavia, Italy
DOI:10.25259/SNI-132-2019
Copyright: © 2019 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Cesare Zoia, Daniele Bongetta, Gianluigi Dorelli, Sabino Luzzi, Mattia Del Maestro, Renato J. Galzio. Transnasal endoscopic removal of a retrochiasmatic cavernoma: A case report and review of literature. 10-May-2019;10:76
How to cite this URL: Cesare Zoia, Daniele Bongetta, Gianluigi Dorelli, Sabino Luzzi, Mattia Del Maestro, Renato J. Galzio. Transnasal endoscopic removal of a retrochiasmatic cavernoma: A case report and review of literature. 10-May-2019;10:76. Available from: https://surgicalneurologyint.com/?post_type=surgicalint_articles&p=9317
Abstract
Background:Cavernomas arising in the region of the optic apparatus are extremely rare, accounting for
Case Description:We report the case of a 53-year-old woman with a hemorrhagic, cavernous malformation of the optic chiasm region and we discuss the technical nuances of the endoscopic transnasal approach we employed. We also review literature for patients with cavernomas treated with a transsphenoidal approach. In our case, the patient underwent the gross-total resection of the lesion through a fully endoscopic transnasal route and the visual disturbances improved immediately after the operation. The ventral access allowed an adequate exposure of the chiasm/anterior circulation complex, thus avoiding an excessive dissection and retraction of neurovascular structures. Moreover, with the aid of angled scopes, we were able to identify the frail vascular supply of the chiasm by employing the suprachiasmatic corridor as well as the infrachiasmatic one. We stress the need for a careful reconstruction of the skull base to avoid a postoperative cerebrospinal fluid leak.
Conclusions:With the evolution and spreading of the neuroendoscopic techniques, in selected cases, we advocate the use of the transnasal route for the removal of cavernomas arising in the region of the anterior optic pathways.
Keywords: Anterior optic pathways, cavernoma, endoscopic transnasal approach
INTRODUCTION
Cavernous malformations (CMs) are developmental vascular abnormalities consisting of sinusoid-like capillary vessels with very slow circulation which could arise throughout the whole nervous system. The walls of the cavernous vessels are fragile and these lesions usually become clinically evident during hemorrhagic events.[
CASE REPORT
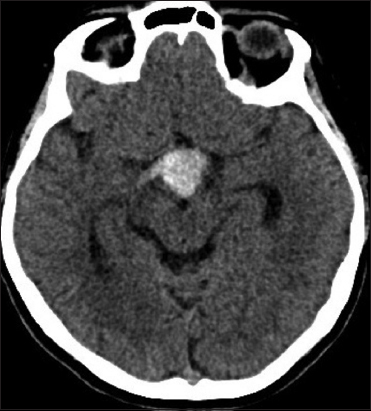
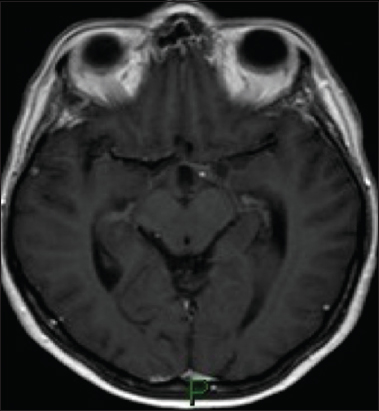
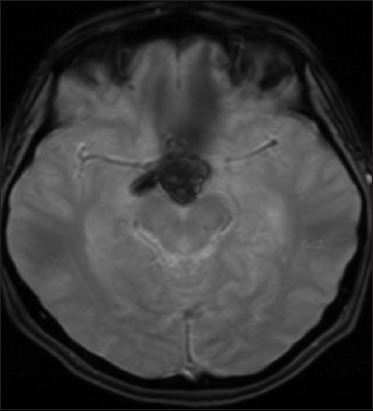
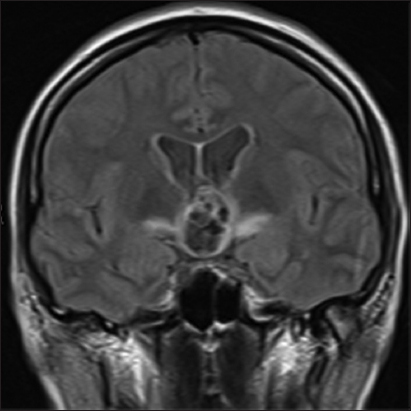
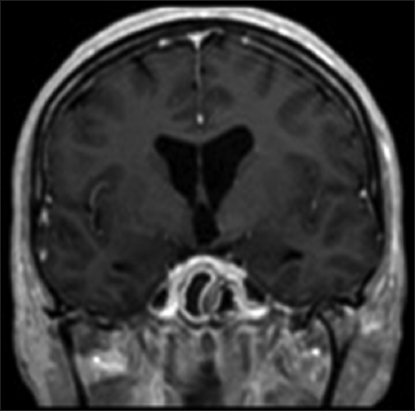
A 53-year-old woman was admitted to our institution complaining of a mild fever, severe headache, and progressive visual loss that had started 4 days before. On admission, the general condition of the patient and the results of the gross neurological examination were unremarkable. Goldmann perimetry testing showed bitemporal hemianopsia. Her visual acuity was 20/400 in the right and 20/200 in the left eye. A computed tomography scan showed a hyperdense, hemorrhagic mass measuring approximately 2.5 cm in diameter in the suprasellar region [
Methods
The PubMed search engine (
DISCUSSION
In literature, the first relevant article about OPH CMs was published in 1978. To date, there are approximately 70 cases of histologically confirmed OPH CM. Still, only three papers[
CONCLUSIONS
Symptomatic OPH CMs are extremely rare and both early decompression and complete resection are indicated to improve the visual dysfunction in cases of hemorrhage. The endoscopic endonasal, transsphenoidal route may be a valid choice to decompress and remove selected lesions. Further studies about the natural history of these specific lesions are needed to evaluate the role of surgery in cases of asymptomatic OPH CMs.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form the patient(s) has/have given his/her/their consent for his/her/their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Aiba T, Tanaka R, Koike T, Kameyama S, Takeda N, Komata T. Natural history of intracranial cavernous malformations. J Neurosurg. 1995. 83: 56-9
2. Al-Sharydah AM, Al-Suhibani SS, Al-Jubran SA, Al-Abdulwahhab AH, Al-Bar M, Al-Jehani HM. Endoscopic management of atypical sellar cavernous hemangioma:A case report and review of the literature. Int J Surg Case Rep. 2018. 42: 161-4
3. Curling OD, Kelly DL, Elster AD, Craven TE. An analysis of the natural history of cavernous angiomas. J Neurosurg. 1991. 75: 702-8
4. Deshmukh VR, Albuquerque FC, Zabramski JM, Spetzler RF. Surgical management of cavernous malformations involving the cranial nerves. Neurosurgery. 2003. 53: 352-7
5. Divitiis ED, Cavallo LM, Cappabianca P, Esposito F. Extended endoscopic endonasal transsphenoidale approach for the removal of suprasellar tumors. Neurosurgery. 2007. 60: 46-59
6. Goldstein HE, Solomon RA. Epidemiology of cavernous malformations. Handb Clin Neurol. 2017. 143: 241-7
7. Hempelmann RG, Mater E, Schröder F, Schön R. Complete resection of a cavernous haemangioma of the optic nerve, the chiasm, and the optic tract. Acta Neurochir. 2007. 149: 699-703
8. Kim DS, Park YG, Choi JU, Chung SS, Lee KC. An analysis of the natural history of cavernous malformations. Surg Neurol. 1997. 48: 9-17
9. Lehner M, Fellner FA, Wurm G. Cavernous haemangiomas of the anterior visual pathways. Short review on occasion of an exceptional case. Acta Neurochir. 2006. 148: 571-8
10. Liu JK, Lu Y, Raslan AM, Gultekin SH, Delashaw JB. Cavernous malformations of the optic pathway and hypothalamus:Analysis of 65 cases in the literature. Neurosurg Focus. 2010. 29: E17-
11. Meng X, Feng X, Wan J. Endoscopic endonasal transsphenoidal approach for the removal of optochiasmatic cavernoma:Case report and literature review. World Neurosurg. 2017. 106: 1053.e11-1.053E+17
12. Muta D, Nishi T, Koga K, Yamashiro S, Fujioka S, Kuratsu JI. Cavernous malformation of the optic chiasm:Case report. Br J Neurosurg. 2006. 20: 312-5
13. Ozer E, Kalemci O, Yücesoy K, Canda S. Optochiasmatic cavernous angioma:Unexpected diagnosis. Case report. Neurol Med Chir. 2007. 47: 128-31
14. Robinson JR, Awad IA, Little JR. Natural history of the cavernous angioma. J Neurosurg. 1991. 75: 709-14
15. Son DW, Lee SW, Choi CH. Cavernous malformation of the optic chiasm:Case report. J Korean Neurosurg Soc. 2008. 44: 88-
16. Sorenson TJ, Chen J, Burrows A, Lanzino G. Surgical resection of cavernous malformation of the optic nerve. Oper Neurosurg (Hagerstown). 2018. 14: 314-
17. Venkataramana N, Rao SV, Arun L, Krishna C. Cavernous malformation of the optic chiasm:Neuro-endoscopic removal. Asian J Neurosurg. 2016. 11: 71-