- Department of Neurosurgery, Prof. Dr. N. Oblu Clinical Emergency Hospital, Iasi, Romania,
- Department of Neurosurgery, Schwarzwald-Baar Klinikum, Villingen-Schwenningen, Germany.
Correspondence Address:
Bogdan Florin Iliescu
Department of Neurosurgery, Prof. Dr. N. Oblu Clinical Emergency Hospital, Iasi, Romania,
DOI:10.25259/SNI_274_2020
Copyright: © 2020 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Bogdan Florin Iliescu1, Pavel Gutu2, Marius Gabriel Dabija1. Traumatic subaxial cervical spine injury – Improving initial evaluation through correlation of diffusion tensor imaging and subaxial cervical spine injury classification SLIC score. 05-Jan-2021;12:10
How to cite this URL: Bogdan Florin Iliescu1, Pavel Gutu2, Marius Gabriel Dabija1. Traumatic subaxial cervical spine injury – Improving initial evaluation through correlation of diffusion tensor imaging and subaxial cervical spine injury classification SLIC score. 05-Jan-2021;12:10. Available from: https://surgicalneurologyint.com/surgicalint-articles/10505/
Abstract
Background: Traumatic injury to spine and spinal cord represents a devastating condition, with a huge risk for permanent severe disabilities. Predicting the long-term outcome in this type of trauma is a very difficult task being under the influence of a wide spectrum of biomechanical and pathophysiological factors. The advent of magnetic resonance imaging (MRI) structural evaluation of the spinal cord brought critical supplementary data in the initial evaluation of these cases. Although edema and hemorrhage proved to be valuable in predicting the outcome, there is a well-documented discrepancy between MRI findings and clinical status.
Methods: We performed diffusion tensor imaging (DTI) MR in 22 symptomatic patients with traumatic cervical spine injuries (mean age 49.6 ± 16, range from 17 to 74 years, 20 males and 2 females). DTI parameters were computed in 15 patients. Regional apparent diffusion coefficient, fractional anisotropy (FA), and fiber length (FL) were calculated in the region of interest defined as the region of maximum structural MR alterations and in the normal cord (above or below the level of the injury). The values for normal and pathological cord were compared. The clinical deficit was assessed with ASIA and subaxial cervical spine injury classification (SLIC) scores. We looked at the correlation between the DTI measures and clinical scores.
Results: There is a highly significant difference between normal and pathological spinal cord for all DTI properties measured. There is also a strong correlation between DTI measures and SLIC clinical score, especially for FA. Significant results were obtained for CDA and FL as well although with lesser statistical power.
Conclusion: Our results suggest that DTI measures, especially FA, represent a strong indicator of the severity of the traumatic cervical cord injury. It correlates very well with SLCI score and can be used as an additional confirmation of the real degree of level lesioning and as a prognostic factor for the neurological outcome regardless of the choice of treatment.
Keywords: Cervical, Diffusion tensor imaging, Magnetic resonance imaging, Spine, Subaxial cervical spine injury classification, Trauma
INTRODUCTION
Traumatic injuries of the cervical spine carry a huge risk for serious neurological deficit and other debilitating conditions. Neurological damage in these cases results in devastating medical, social, emotional, and financial consequences. Assessing the degree of structural destruction within the spinal cord and making a reliable and accurate prediction of the neurological outcome continues to be a very difficult task. Conventional magnetic resonance imaging (MRI) has become a standard in the routine exploration of the soft-tissue and spinal cord damages in spinal trauma.[
MATERIALS AND METHODS
Our institutional ethics review Board approved this study
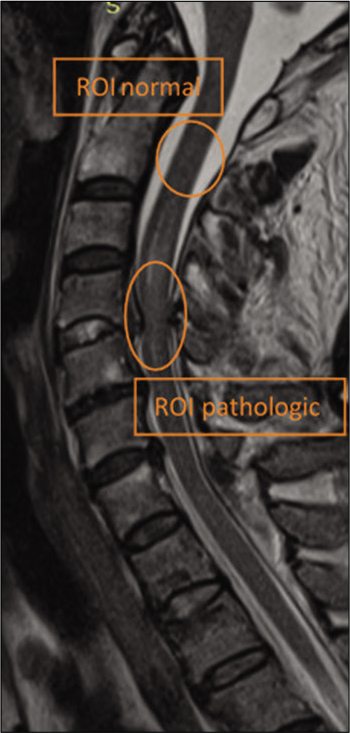
Twenty-eight patients with traumatic cervical spine injuries (mean, 50.1 ± 19.7 years of age; 3 women and 25 men) have been prospectively included in a MRI protocol that included conventional MRI and DTI. MRI studies were performed 10 h–6 days (mean, 36 ± 24 h) following the traumatic incident. Indications for MRI included neurologic deficit on clinical examination localized to the cervical spine (21 cases), spine fracture demonstrated on the X-ray or CT at admission – for the evaluation of the extent of ligamentous injury (3 cases), and cervical pain unexplained by the cervical spine radiographs or CT at admission (4 cases). Mechanisms of injury included motor vehicle accidents (6 cases), animal-powered vehicle accidents (7 cases), motorcycle accidents (3 cases), fall (8 cases), sports accidents (2 cases), and assault (2 cases). DTI data from 13 patients were not used for further analysis because of their poor quality and/or present artifacts. The 15 remaining patients formed the study group.
MRI technique
All MRI studies were performed on a 1.5T Siemens Avanto scanner (Siemens, Erlangen, Germany) with parallel imaging capability. Conventional MRI included axial and sagittal T1, sagittal and axial T2, fluid-attenuated inversion recovery (TE/ TR/echo train), and axial T2* images. DTIs were obtained using an echo-planar imaging (EPI) sequence at a resolution of 128/128 over a 20 cm FOV. Sections had a 3 mm thickness and on average 80 (between 76 and 84) of them were produced to image the whole spine from cervical-medullar junction to the cervical-thoracic junction. A 12-channel head-neck array coil was used on all patients. Parallel imaging was carried on with the phase encoding in the anterior-to-posterior direction. For anatomic reference, we used sagittal T2-weighted images and b = 0 s/mm2 images from the DTIs.
Image analysis
In echo-planar-based diffusion-weighted imaging (DWI) and DTI, the evaluation of diffusion parameters such as ADCs and anisotropy indices is affected by image distortions that arise from residual eddy currents produced by the diffusion-sensitizing gradients. We used FMRIB’s Diffusion Toolbox from FMRIB Software Library v5.0 (
Clinical evaluation
Each patient underwent a full neurological examination at admission. The SLIC score as described by Vaccaro et al.[
RESULTS
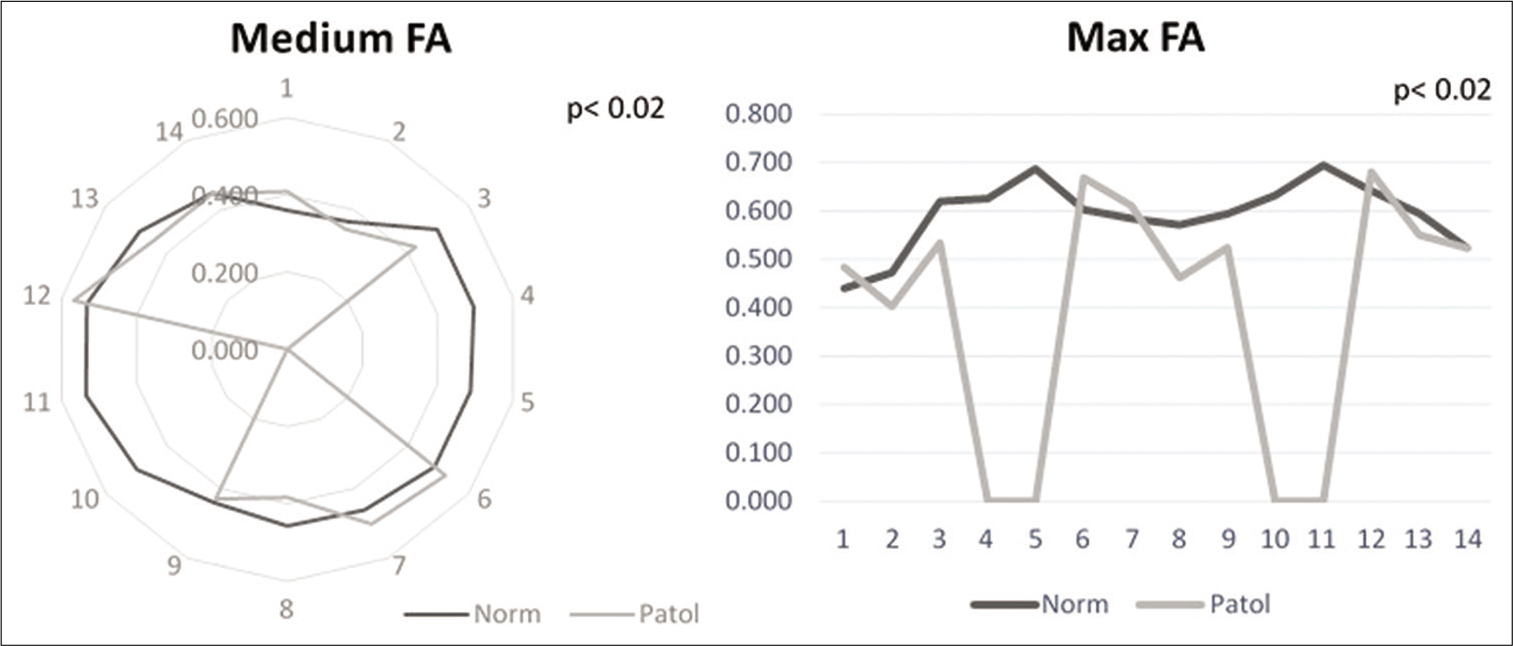
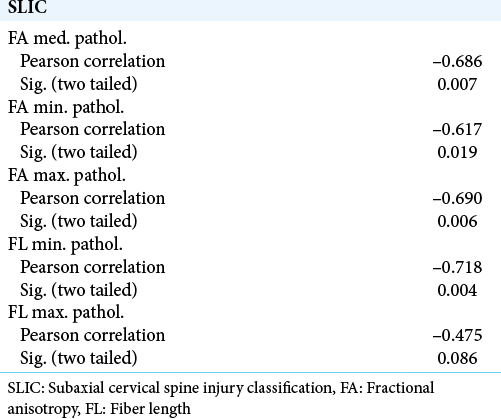
Analyzing the differences in FA between the level of maximal injury and the distant, apparently intact spinal cord we found that both the maximum and the medium values were significantly different between the two ROIs (P < 0.02, Student’s t-test). The minimum did not reach the significance level (P > 0.05). The two significant measures were used in the clinical correspondence test [
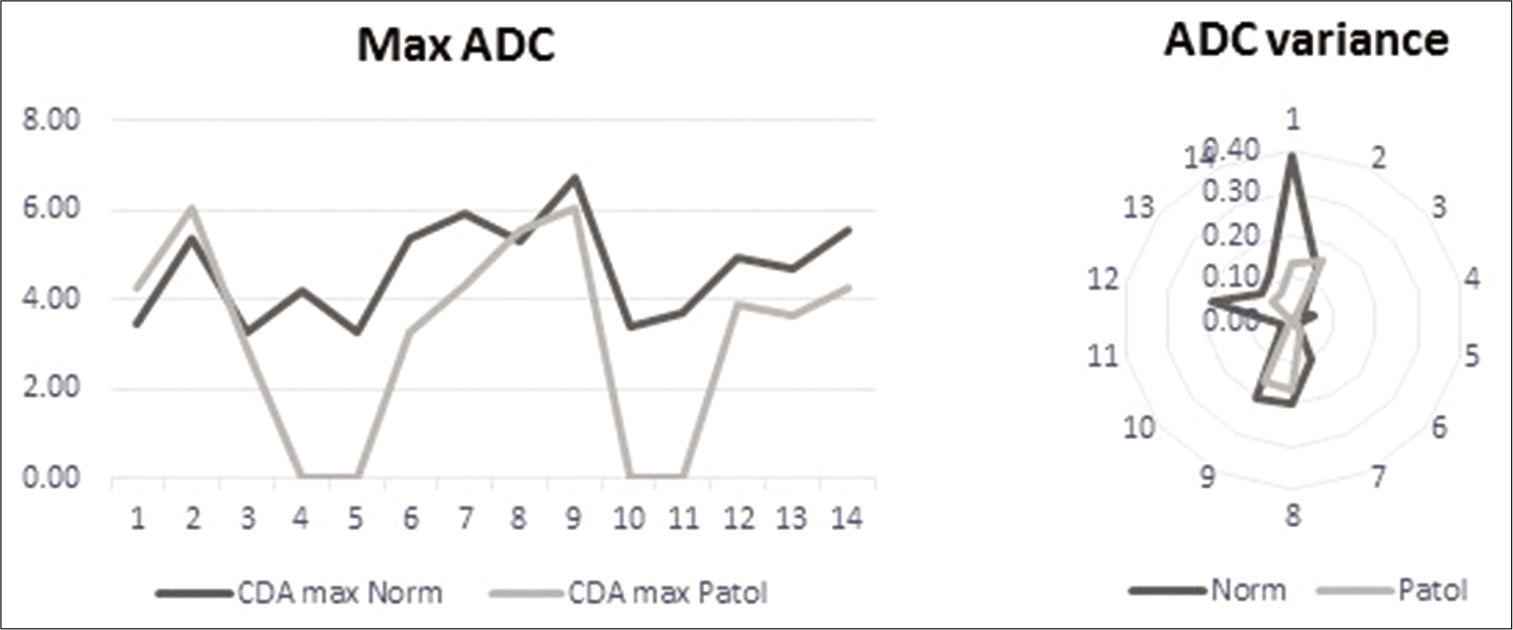
For ADC, we computed, besides the maximal, medial, and minimal values, the variance. When we tested the values between the two ROIs, the minimal and medial values did not reach the level of significance (P > 0.05). The maximal value and the variance, however, showed a highly significant difference (P < 0.003 and P < 0.006) [
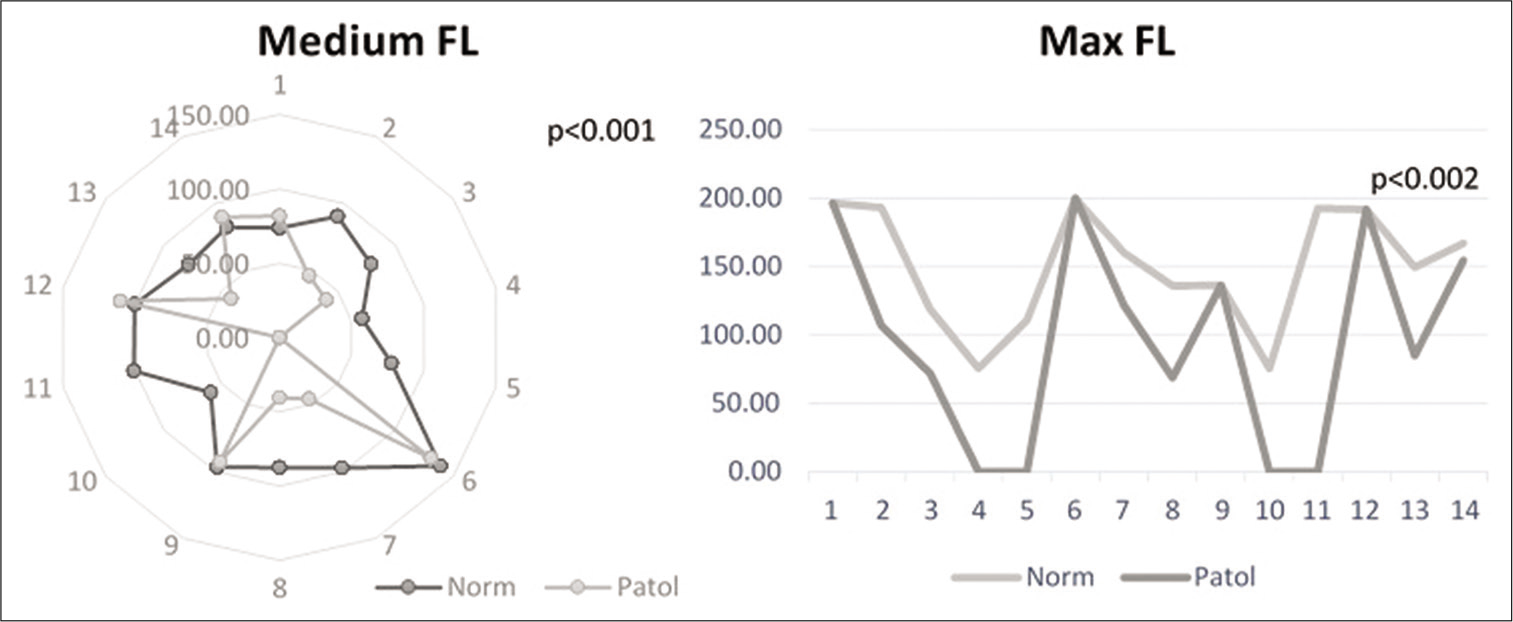
Finally, for FL, the same for values were computed (maximal, minimal, medial, and variance). Again, the minimal values and the variance did not reach the level of significance (P > 0.05). For the maximal and medial values, however, the differences between the two regions were highly significant and were used further in the study [
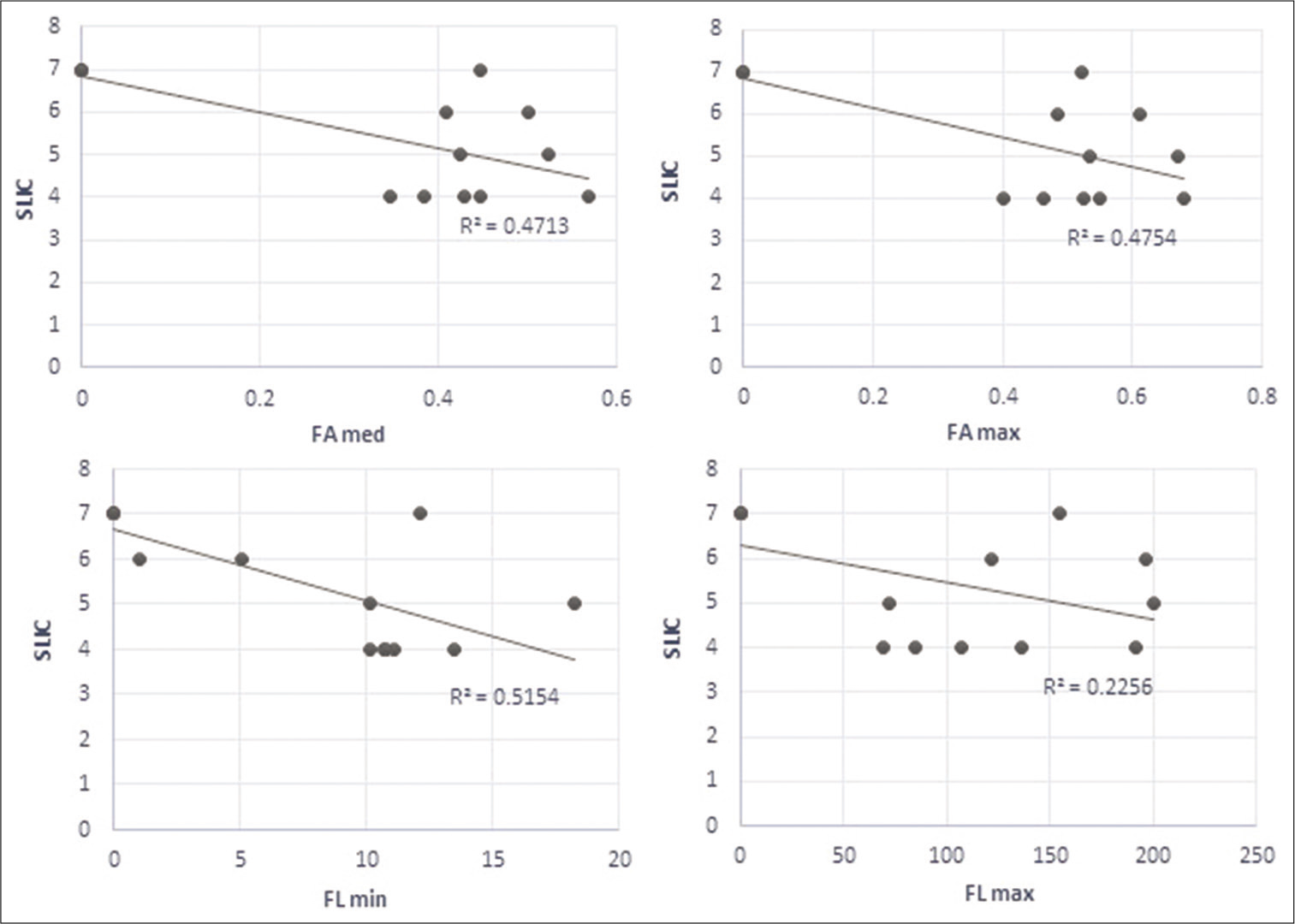
We found that for measures that we decided to use in the context of clinical data, the measured values of DTI parameters and the SLIC score had a strong linear regression, except for the maximal FL [
DISCUSSION
Traumatic cervical spine injuries may lead to significant morbidity and generate a number of disastrous consequences culminating in severe neurological deficit and death. Two major issues are commanded by these patients. First is the question of the best treatment for each particular case. Second is communicating to the patient and his family an accurate evaluation of the present status and of the prognostic based as much as possible on real data. A multitude of evaluation and classification systems have been introduced overtime. However, none of these showed a real practical validation for a reliable and suggestive subaxial cervical trauma assessment due to a number of factors (vague terminology, retrospective injury mechanism devise, failure in taking into account other essential variables related to spine stability, and insufficient data about spinal cord structural and functional integrity).
Imaging of the spinal cord through conventional MRI is routinely used to document the location of cord injury and the amount of cord compression from extramedullary pathologic factors. Extending the investigation with DTI proved to be of benefit in further defining the morphology changes in white matter tracts within the spinal cord. The measurable parameters of DWI showed a high sensitivity in discovering changes in patients with apparently normal conventional MR images.[
DTI parameters offer valuable information that evaluate axonal injuries across different axonal tracts, therefore, the diffusion technique is one of the most widely used in evaluating the effects of different pathologies on the nervous tissue. Decreased values of FA were found in studies regarding brain tumor (low- vs. high-grade glioma, glioblastoma, and white matter tracts around glioblastoma), epidermoid cyst, or brain edema.[
Progress has also been made in the early detection of neurodegenerative diseases such as multiple sclerosis or amyotrophic lateral sclerosis. In his study, Gatto et al.[
The single direction of the cord white matter tracts (craniocaudal) and their symmetry within the cord makes them potential very good candidates for diffusion measurements.[
Our results support the hypothesis that DTI parameters, including ADC values for cervical spine contusions, are significantly lower than comparative values in the apparently intact spinal cord. These data suggest that the three DTI parameters we measured (FA, ADC, and FL) represent sensitive markers of spinal cord injury. If we look at the maximal values for all parameters, they are the most sensitive markers of cord injury, especially the ADC and FL. They showed a similar regression with a strong r2 when run against clinical status scale and also showed strong correlations with the clinical status. The apparently normal cord failed to produce the same strong correlations. Our findings are in agreement with previously reported results.
Furthermore, our data suggest that the abnormalities in the DTI parameters in areas of the spinal cord that appears normal on conventional MRI could be used to document the true extent of injury and correlate with the neurologic deficit with a high degree of statistical significance. Our results also indicate that DTI parameters used in conjunction with the clinical evaluation could be an independent predictor of the severity of spinal cord injury.
However, we could not produce histological evidence of the injured cord in the studied patient group. Future studies that can correlate imagistic findings with histologic data are due to confirm these findings. There are also certain limitations inherent in this study that needs further addressing. In addition to its small sample size, the study did not correlate DTI parameters with histologic observations, as mentioned above. Future studies with a large sample size are necessary to further validate our results. Last but not least, further investigation of the dynamics of correlation from the supra-acute to chronic phases of cervical trauma (based on the findings from the above-mentioned studies looking at different pathologies) would be of interest and further added value.
CONCLUSION
Our study comes to strengthen the value of DTI parameters as an indicator of the extent of spinal cord injury and they are apparent even in the absence of changes on the conventional MR images. The DTI changes show a strong correlation with the clinical status and could be used as a supplementary, more in-depth predictor of the neurological outcome in these cases.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Agarwal V, Kumar M, Singh JK, Rathore RK, Misra R, Gupta RK. Diffusion tensor anisotropy magnetic resonance imaging: A new tool to assess synovial inflammation. Rheumatology (Oxford). 2009. 48: 378-82
2. Biton IE, Duncan ID, Cohen Y. High b-value q-space diffusion MRI in myelin-deficient rat spinal cords. Magn Reson Imaging. 2006. 24: 161-6
3. Cortez-Conradis D, Rios C, Moreno-Jimenez S, RoldanValadez E. Partial correlation analyses of global diffusion tensor imaging-derived metrics in glioblastoma multiforme: Pilot study. World J Radiol. 2015. 7: 405-14
4. Demir A, Ries M, Moonen CT, Vital JM, Dehais J, Arne P. Diffusion-weighted MR imaging with apparent diffusion coefficient and apparent diffusion tensor maps in cervical spondylotic myelopathy. Neuroradiology. 2003. 229: 37-43
5. El-Serougy L, Razek AA, Ezzat A, Eldawoody H, El-Morsy A. Assessment of diffusion tensor imaging metrics in differentiating low-grade from high-grade gliomas. Neuroradiol J. 2016. 29: 400-7
6. Flanders AE, Schaefer DM, Doan HT, Mishkin MM, Gonzalez CF, Northrup BE. Acute cervical spine trauma: Correlation of MR imaging findings with degree of neurologic deficit. Radiology. 1990. 177: 25-33
7. Flanders AE, Spettell CM, Tartaglino LM, Friedman DP, Herbison GJ. Forecasting motor recovery after cervical spinal cord injury: Value of MR imaging. Radiology. 1996. 201: 649-55
8. Gatto RG, Weissmann C, Amin M, Finkielsztein A, Sumagin R, Mareci TH. Assessing neuraxial microstructural changes in a transgenic mouse model of early stage amyotrophic lateral sclerosis by ultra-high field MRI and diffusion tensor metrics. Animal Model Exp Med. 2020. 3: 117-29
9. Gupta RK, Srivastava S, Saksena S, Rathore RK, Awasthi R, Prasad KN. Correlation of DTI metrics in the wall and cavity of brain abscess with histology and immunohistochemistry. NMR Biomed. 2010. 23: 262-9
10. Jolapara M, Kesavadas C, Radhakrishnan VV, Saini J, Patro SN, Gupta AK. Diffusion tensor mode in imaging of intracranial epidermoid cysts: One step ahead of fractional anisotropy. Neuroradiology. 2009. 51: 123-9
11. Katzberg RW, Benedetti PT, Drake CM, Ivanovic M, Levine RA, Beatty CS. Acute cervical spine injuries: Prospective MR imaging assessment at a level 1 trauma center. Radiology. 1999. 213: 203-12
12. Kulkarni MV, McArdle CB, Kopanicky D, Miner M, Cotler HB, Lee KF. Acute spinal cord injury: MR imaging at 1.5 T. Radiology. 1987. 164: 837-43
13. Mamata H, Jolesz FA, Maier SE. Apparent diffusion coefficient and fractional anisotropy in spinal cord: Age and cervical spondylosis-related changes. J Magn Reson Imaging. 2005. 22: 38-43
14. Shanmuganathan K, Gullapalli RP, Zhuo J, Mirvis SE. Diffusion tensor MR imaging in cervical spine trauma. AJNR Am J Neuroradiol. 2008. 29: 655-9
15. Silberstein M, Tress BM, Hennessy O. Prediction of neurologic outcome in acute spinal cord injury: The role of CT and MR. AJNR Am J Neuroradiol. 1992. 13: 1597-608
16. Vaccaro AR, Hulbert RJ, Patel AA, Fisher C, Dvorak M, Lehman RA. The subaxial cervical spine injury classification system: A novel approach to recognize the importance of morphology, neurology, and integrity of the disco-ligamentous complex. Spine (Phila Pa 1976). 2007. 32: 2365-74
17. Wolanczyk M, Bladowska J, Kołtowska A, PokryszkoDragan A, Podgórski P, Budrewicz S. Diffusion tensor imaging of normal-appearing cervical spinal cords in patients with multiple sclerosis: Correlations with clinical evaluation and cerebral diffusion tensor imaging changes, Preliminary experience. Adv Clin Exp Med. 2020. 29: 441-8