- Department of Neurosurgery, Saitama Medical Center, Saitama Medical University, 1981 Kamoda, Kawagoe, Saitama 350-8550, Japan
Correspondence Address:
Soichi Oya
Department of Neurosurgery, Saitama Medical Center, Saitama Medical University, 1981 Kamoda, Kawagoe, Saitama 350-8550, Japan
DOI:10.4103/2152-7806.157445
Copyright: © 2015 Oya S. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.How to cite this article: Oya S, Nejo T, Fujisawa N, Tsuchiya T, Indo M, Nakamura T, Matsui T. Usefulness of repetitive intraoperative indocyanine green-based videoangiography to confirm complete obliteration of micro-arteriovenous malformations. Surg Neurol Int 21-May-2015;6:85
How to cite this URL: Oya S, Nejo T, Fujisawa N, Tsuchiya T, Indo M, Nakamura T, Matsui T. Usefulness of repetitive intraoperative indocyanine green-based videoangiography to confirm complete obliteration of micro-arteriovenous malformations. Surg Neurol Int 21-May-2015;6:85. Available from: http://surgicalneurologyint.com/surgicalint_articles/usefulness-repetitive-intraoperative-indocyanine-green%e2%80%91based/
Abstract
Background:It is difficult to intraoperatively confirm the total disappearance of arteriovenous (AV) shunts during surgery for microarteriovenous malformations (micro-AVMs), especially when the nidus is extremely small or diffuse on preoperative angiography. Although intraoperative angiography is effective for evaluating residual shunts, procedure-related risks raise important concerns. The purpose of this study was to assess the usefulness of intraoperative indocyanine green-based videoangiography (ICG–VA) to determine complete disappearance of micro-AVMs during surgery.
Methods:We retrospectively analyzed eight patients with ruptured micro-AVMs who were treated using craniotomy with ICG–VA at our institution.
Results:Two patients underwent emergency partial evacuation of hematoma and external decompression before the diagnostic angiography. While three patients had a nidus smaller than 1 cm, five patients had only early draining veins without an appreciable nidus. The draining veins were superficial in six cases and deep in two cases. The average interval from onset to surgery was 33 days (range, 2–57). ICG–VA was repetitively conducted until disappearance of the AV shunt was confirmed. No residual AV shunt was observed on postoperative radiological examinations. In all cases, the diagnosis of AVM was confirmed from the results of postoperative pathological examination.
Conclusions:ICG–VA could detect early draining veins more clearly in situ than diagnostic angiography. Although it is not as effective for visualizing lesions with deep draining veins, repetitive ICG–VA was safe and effective for confirming the disappearance of AV shunts with superficial drainage.
Keywords: Arteriovenous malformation, hematoma, indocyanine green-based videoangiography, microarteriovenous malformation
INTRODUCTION
Since Margolis emphasized small cerebrovascular malformations as an important cause of intracerebral hemorrhage (ICH),[
METHODS
Patients, diagnosis, and treatment strategies
This study was approved by the Saitama Medical Center Institutional Review Board (No. 954). Between January 2011 and April 2014, 19 patients with AVM were treated at the Saitama Medical Center: There were 2 cases of unruptured AVM and 17 of ruptured AVM. In the present study, micro-AVM was angiographically defined as an AVM with a nidus smaller than 1 cm or as an early draining vein without an angiographically appreciable nidus.[
Our policy regarding the treatment of ICH in young patients or atypically located hypertensive ICH is to investigate the pathology of the hemorrhage using angiography as well as 3D-CT angiography and magnetic resonance (MR) imaging. When the patients’ condition is critical because of the mass effect of the hematoma, emergency partial evacuation of the hematoma and external decompression are conducted before the angiography. Because a small nidus may not be visible even on angiograms recorded in the acute phase of hemorrhage, we carefully examined digital subtraction angiography images, especially for cases of lobar hemorrhage in young patients with no history of hypertension. If the result of the initial angiography performed immediately after onset is negative, angiography is repeated 4–6 weeks after hematoma onset. To delineate the early draining vein clearly on angiography, we obtain images with a short serial duration (12 shots per second) and a double dose of contrast medium. Patients diagnosed with micro-AVMs were scheduled for craniotomy and resection of superficial micro-AVMs.
Surgical technique and intraoperative ICG–VA
The patient's head was fixed in a 3-pin Mayfield headholder. The Stealth Navigation system (Medtronic, Surgical Navigation Technologies, Louisville, CO) was set up to confirm the location of the small nidus in the operative field. After standard craniotomy was performed to create ample space for surgery, ICG–VA was routinely conducted, and early draining veins were visualized. To compare early draining veins and normally irrigating veins, it was important to adequately expose the cortex. ICG dye diluted to 2.5 mg/mL was injected intravenously as a bolus (0.1 mg/kg of ICG dye, followed by 20 mL of saline). Vessels were visualized using surgical microscopes integrated with a video camera (OPMI PENTERO with INFRARED 800 microscope [Carl Zeiss, Oberkochen, Germany] or M720 OH5 [Leica Microsystems, Heerbrugg, Switzerland]). ICG–VA was performed to confirm the disappearance of the early draining vein when complete shunt obliteration was suspected. If the targeted early draining vein was still visualized on angiography, exploration of the small nidus remaining on the hematoma walls was continued. The ICG–VA was repeated until all the veins around the hematoma could be visualized in the normal venous phase. Postoperative angiography or MR imaging was performed 1–2 weeks after the surgery to confirm total obliteration of AV shunts.
RESULTS
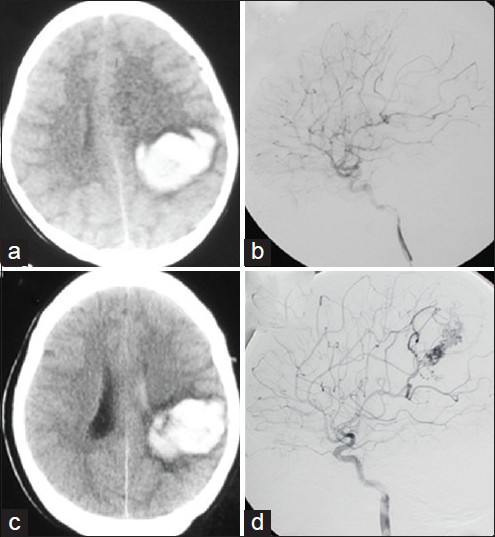
The patient characteristics and outcomes are summarized in [
Six patients showed complete recovery (modified Rankin Scale [mRS] 0), while two experienced moderate neurological deficits rated as mRS 2 and 3). The deficits were attributed to initial damage due to the hemorrhage and were not considered surgical complication. The average duration of follow-up was 5.2 months (range, 1.2–15.5). The disappearance of the AV shunt was postoperatively confirmed on angiography in seven cases and on MR images in one case.
Illustrative case
A 43-year-old male with no past medical history woke up one day with a severe headache, nausea, and subjective visual disturbance. On admission, neurological examination showed slight disturbance of consciousness and homonymous hemianopia on the left side. An emergency CT scan showed a 5.3 × 2.2 × 3.8 cm-sized ICH in the right parietal lobe [
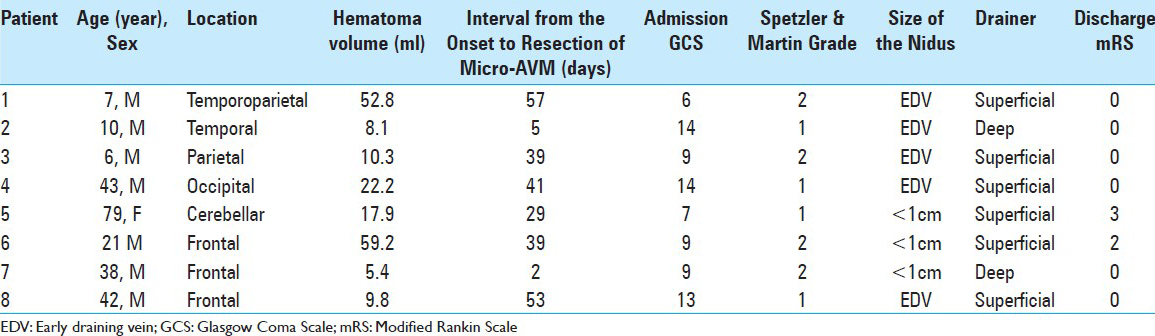
Figure 1
(a) CT scan at admission showing an intracerebral hemorrhage in the right parietal lobe. (b) A left internal carotid angiogram on admission showing no definitive vascular abnormality. (c) A time-of-flight MR image showing a small but abnormally dilated vein (arrow) near the hematoma. (d) A left internal carotid angiogram repeated in 2 weeks clearly showing the early draining vein (arrowhead) near the hematoma
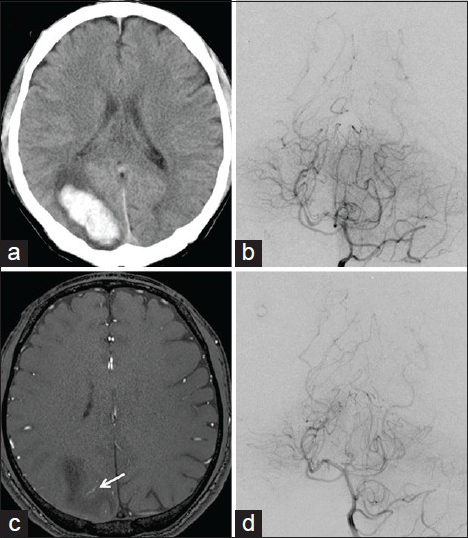
Figure 2
(a) A photograph showing cortical arteries and veins around the hematoma. (b) Initial ICG–VA showing an early draining vein (arrow) (c) ICG–VA in the late venous phase showing the hematoma cavity as a dark area (*). (d) A photograph showing that the evacuation was almost complete. (e) Second ICG–VA showing the disappearance of the early draining vein (arrowhead). However, another early draining vein (arrowhead) is visible near the hematoma cavity. (f) Final ICG–VA confirming the complete disappearance of all drainers. (g) Postoperative angiogram showing no residual nidus (arrowhead)
DISCUSSION
In the present study, we examined the usefulness and limitations of ICG–VA during surgical treatment for micro-AVMs. We highlighted some diagnostic and operative tips specific for micro-AVMs and not large AVMs. Our technique using ICG–VA seem especially useful for intraoperative confirmation of complete AV shunt obliteration.
Problems related to the preoperative diagnosis of micro-AVMs and the importance of in situ evaluation of early venous drainage using ICG–VA
Despite their small size, micro-AVM can cause large hematomas, leading to serious neurological impairments.[
Effectiveness of ICG–VA in confirming total obliteration of AV shunts for micro-AVMs or diffuse AVMs
Localization of the nidus and drainer is of paramount importance in micro-AVM surgery in order to ensure complete AV shunt obliteration,[
In most cases of compact AVMs with a medium-to-large nidus, surgeons are usually able to find the lesion easily and ensure complete obliteration of AV shunts by visual confirmation of changes in the color of the drainer. This empirical judgment can be rationally reinforced by intraoperative angiography, which may also help improve the obliteration rate.[
To our knowledge, the clinical significance of intraoperative angiography for micro-AVMs has not been discussed previously, but given the preoperative difficulties in lesion delineation, its effectiveness would probably be limited. Identification of residual AV shunt after surgical resection is critical because partial resection of AVMs may increase the risk of hemorrhage.[
Takagi et al. described the case of a 2-year-old patient with a diffuse AVM.[
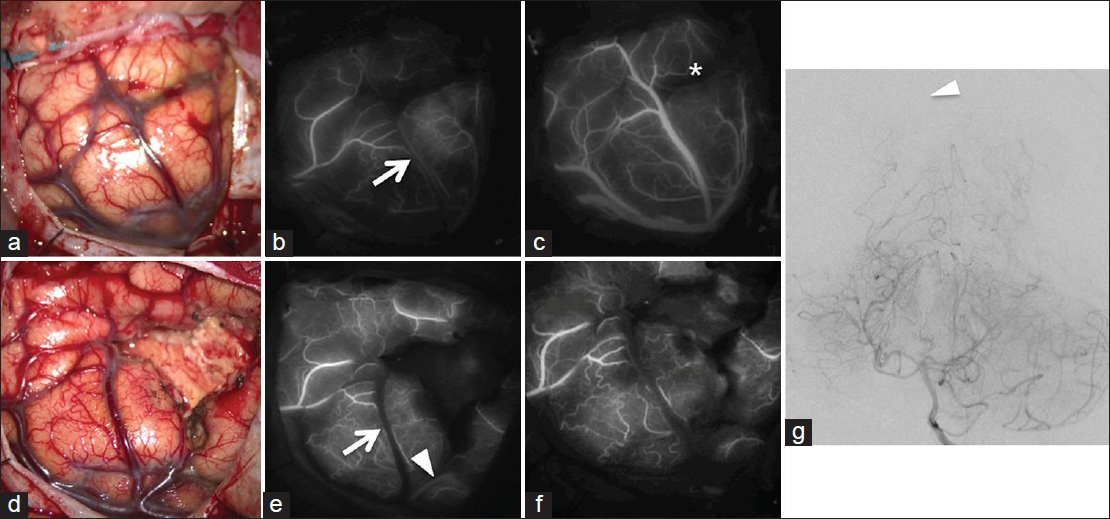
Figure 3
(a) CT scan at admission showing an intracerebral hematoma in the left frontoparietal region. (b) A left internal carotid angiogram at admission showing no abnormal findings that could explain the hemorrhage. (c) A CT scan captured 8 years after the initial episode showing hematoma recurrence at the same location as that when the patient first presented with disturbed consciousness. (d) A left internal carotid angiogram showing a diffuse AVM
If the early draining vein had been detected on ICG–VA in situ during the evacuation surgery, we could have followed the vein and rigorously searched for hidden AV shunts. Zaidi et al. recently reported that the significance of intraoperative ICG–VA alone did not improve the identification of residual nidus and clinical outcome based on the data of 56 patients with AVM treated using ICG.[
Limitations of ICG–VA
There are some limitations regarding the use of ICG–VA. Deep-seated lesions are not visualized on ICG–VA.[
CONCLUSIONS
ICG–VA is a powerful tool for in situ localization of AV shunts of micro-AVMs with superficial drainage. It is also extremely useful for confirming complete obliteration of AV shunts for such micro-AVMs. We believe that repetitive ICG–VA contributes to reducing the chances of residual AV shunts, especially micro-AVMs.
Video Available on : www.surgicalneurologyint.com
References
1. Alén JF, Lagares A, Paredes I, Campollo J, Navia P, Ramos A. Cerebral microarteriovenous malformations: A series of 28 cases. J Neurosurg. 2013. 119: 594-602
2. Andreou A, Ioannidis I, Lalloo S, Nickolaos N, Byrne JV. Endovascular treatment of intracranial microarteriovenous malformations. J Neurosurg. 2008. 109: 1091-7
3. Balamurugan S, Agrawal A, Kato Y, Sano H. Intra operative indocyanine green video-angiography in cerebrovascular surgery: An overview with review of literature. Asian J Neurosurg. 2011. 6: 88-93
4. Barrow DL, Boyer KL, Joseph GJ. Intraoperative angiography in the management of neurovascular disorders. Neurosurgery. 1992. 30: 153-9
5. Becker DH, Townsend JJ, Kramer RA, Newton TH. Occult cerebrovascular malformations. A series of 18 histologically verified cases with negative angiography. Brain. 1979. 102: 249-87
6. Cellerini M, Mangiafico S, Villa G, Nistri M, Pandolfo C, Noubari H. Cerebral microarteriovenous malformations: Diagnostic and therpeutic features in a series of patients. AJNR Am J Neuroradiol. 2002. 23: 945-52
7. Crawford JV, Russell DS. Cryptic arteriovenous and venous hamartomas of the brain. J Neurol Neurosurg Psychiatr. 1956. 19: 1-11
8. Dehdashti AR, Thines L, Da C, Leodante B, terBrugge KG, Willinsky RA. Intraoperative biplanar rotational angiography during neurovascular surgery. Technical note. J Neurosurg. 2009. 111: 188-92
9. Derdeyn CP, Moran CJ, Cross DT, Grubb RL, Dacey RG. Intraoperative digital subtraction angiography: A review of 112 consecutive examinations. AJNR Am J Neuroradiol. 1995. 16: 307-18
10. Elhammady MS, Heros RC. Microarteriovenous malformations. J Neurosurg. 2013. 119: 591-3
11. Elhammady MS, Baskaya MK, Heros RC. Early elective surgical exploration of spontaneous intracerebral hematomas of unknown origin. J Neurosurg. 2008. 109: 1005-11
12. Ellis MJ, Armstrong D, Vachhrajani S, Kulkarni AV, Dirks PB, Drake JM. Angioarchitectural features associated with hemorrhagic presentation in pediatric cerebral arteriovenous malformations. J Neurointerv Surg. 2012. 5: 191-5
13. Ellis MJ, Kulkarni AV, Drake JM, Rutka JT, Armstrong D, Dirks PB. Intraoperative angiography during microsurgical removal of arteriovenous malformations in children. J Neurosurg Pediatr. 2010. 6: 435-43
14. Faber F, Thon N, Fesl G, Rachinger W, Guckler R, Tonn JC. Enhanced analysis of intracerebral arterioveneous malformations by the intraoperative use of analytical indocyanine green videoangiography: Technical note. Acta Neurochir. 2011. 153: 2181-7
15. Ferroli P, Acerbi F, Broggi M, Broggi G. Arteriovenous micromalformation of the trigeminal root: Intraoperative diagnosis with indocyanine green videoangiography: Case report. Neurosurgery. 2010. 67: onsE309-10
16. Gaballah M, Storm PB, Rabinowitz D, Ichord RN, Hurst RW, Krishnamurthy G. Intraoperative cerebral angiography in arteriovenous malformation resection in children: A single institutional experience. J Neurosurg Pediatr. 2014. 13: 222-8
17. Ghosh S, Levy ML, Stanley P, Nelson M, Giannotta SL, McComb JG. Intraoperative angiography in the management of pediatric vascular disorders. Pediatr Neurosurg. 1999. 30: 16-22
18. Golden JB, Kramer RA. The angiographically occult cerebrovascular malformation. Report of three cases. J Neurosurg. 1978. 48: 292-6
19. Goren O, Monteith SJ, Hadani M, Bakon M, Harnof S. Modern intraoperative imaging modalities for the vascular neurosurgeon treating intracerebral hemorrhage. Neurosurg Focus. 2013. 34: E2-
20. Grossart KW, Turner JW. Operative angiography in cerebral vascular surgery. Br J Radiol. 1973. 46: 401-2
21. Häggi D, Etminan N, Steiger HJ. The impact of microscope-integrated intraoperative near-infrared indocyanine green videoangiography on surgery of arteriovenous malformations and dural arteriovenous fistulae. Neurosurgery. 2010. 67: 1094-103
22. Hino A, Fujimoto M, Yamaki T, Iwamoto Y, Katsumori T. Value of repeat angiography in patients with spontaneous subcortical hemorrhage. Stroke. 1998. 29: 2517-21
23. Hoh BL, Carter BS, Ogilvy CS. Incidence of residual intracranial AVMs after surgical resection and efficacy of immediate surgical re-exploration. Acta Neurochir (Wien). 2004. 146: 1-7
24. Kader A, Young WL, Pile-Spellman J, Mast H, Sciacca RR, Mohr JP. The influence of hemodynamic and anatomic factors on hemorrhage from cerebral arteriovenous malformations. Neurosurgery. 1994. 34: 801-7
25. Killory BD, Nakaji P, Gonzales LF, Ponce FA, Wait SD, Spetzler RF. Prospective evaluation of surgical microscope-integrated intraoperative near-infrared indocyanine green angiography during cerebral arteriovenous malformation surgery. Neurosurgery. 2009. 65: 456-62
26. Kiriş T, Sencer A, Sahinbaş M, Sencer S, Imer M, Izgi N. Surgical results in pediatric Spetzler-Martin grades I-III intracranial arteriovenous malformations. Childs Nerv Syst. 2005. 21: 69-74
27. Klimo P, Rao G, Brockmeyer D. Pediatric arteriovenous malformations: A 15-year experience with an emphasis on residual and recurrent lesions. Childs Nerv Syst. 2007. 23: 31-7
28. Kono K, Uka A, Mori M, Haga S, Hamada Y, Nagata S. Intra-arterial injection of indocyanine green in cerebral arteriovenous malformation surgery. Turk Neurosurg. 2013. 23: 676-9
29. Kotowski M, Sarrafzadeh A, Schatlo B, Boex C, Narata AP, Pereira VM. Intraoperative angiography reloaded: A new hybrid operating theater for combined endovascular and surgical treatment of cerebral arteriovenous malformations: A pilot study on 25 patients. Acta Neurochir. 2013. 155: 2071-8
30. Kramer RA, Wing SD. Computed tomography of angiographically occult cerebral vascular malformations. Radiology. 1977. 123: 649-52
31. Lane BC, Cohen-Gadol AA. A prospective study of microscope-integrated intraoperative fluorescein videoangiography during arteriovenous malformation surgery: Preliminary results. Neurosurg Focus. 2014. 36: E15-
32. Lang SS, Beslow LA, Bailey RL, Vossough A, Ekstrom J, Heuer GG. Follow-up imaging to detect recurrence of surgically treated pediatric arteriovenous malformations. J Neurosurg Pediatr. 2012. 9: 497-504
33. Lobato RD, Perez C, Rivas JJ, Cordobes F. Clinical, radiological, and pathological spectrum of angiographically occult intracranial vascular malformations. Analysis of 21 cases and review of the literature. J Neurosurg. 1988. 68: 518-31
34. Loop JW, Foltz EL. Applications of angiography during intracranial operation. Acta Radiol Diagn (Stockh). 1966. 5: 363-7
35. Margolis G, Odom GL, Woodhall B. Further experiences with small vascular malformations as a cause of massive intracerebral bleeding. J Neuropathol Exp Neurol. 1961. 20: 161-7
36. Margolis G, Odom GL, Woodhall B, Bloor BM. The role of small angiomatous malformations in the production of intracerebral hematomas. J Neurosurg. 1951. 8: 564-75
37. Miyamoto S, Hashimoto N, Nagata I, Nozaki K, Morimoto M, Taki W. Posttreatment sequelae of palliatively treated cerebral arteriovenous malformations. Neurosurgery. 2000. 46: 589-94
38. Moon HS, Joo SP, Seo BR, Jang JW, Kim JH, Kim TS. Value of indocyanine green videoangiography in deciding the completeness of cerebrovascular surgery. J Korean Neurosurg Soc. 2013. 53: 349-55
39. Munshi I, Macdonald RL, Weir BK. Intraoperative angiography of brain arteriovenous malformations. Neurosurgery. 1999. 45: 491-7
40. Ng YP, King NK, Wan KR, Wang E, Ng I. Uses and limitations of indocyanine green videoangiography for flow analysis in arteriovenous malformation surgery. J Clin Neurosci. 2013. 20: 224-32
41. Ogilvy CS, Heros RC, Ojemann RG, New PF. Angiographically occult arteriovenous malformations. J Neurosurg. 1988. 69: 350-5
42. Perrini P, Scollato A, Cellerini M, Mangiafico S, Ammannati F, Mennonna P. Results of surgical and endovascular treatment of intracranial micro-arteriovenous malformations with emphasis on superselective angiography. Acta Neurochir (Wien). 2004. 146: 755-66
43. Rahal JP, Malek AM. Benefit of cone-beam computed tomography angiography in acute management of angiographically undetectable ruptured arteriovenous malformations. J Neurosurg. 2013. 119: 1015-20
44. Stiver SI, Ogilvy CS. Micro-arteriovenous malformations: Significant hemorrhage from small arteriovenous shunts. Neurosurgery. 2000. 46: 811-8
45. Takagi Y, Kikuta K, Nozaki K, Sawamura K, Hashimoto N. Detection of a residual nidus by surgical microscope-integrated intraoperative near-infrared indocyanine green videoangiography in a child with a cerebral arteriovenous malformation. J Neurosurg. 2007. 107: 416-8
46. Takagi Y, Sawamura K, Hashimoto N, Miyamoto S. Evaluation of serial intraoperative surgical microscope-integrated intraoperative near-infrared indocyanine green videoangiography in patients with cerebral arteriovenous malformations. Neurosurgery. 2012. 70: 34-42
47. Tanaka Y, Furuse M, Iwasa H, Masuzawa T, Saito K, Sato F. Lobar intracerebral hemorrhage: Etiology and a long-term follow-up study of 32 patients. Stroke. 1986. 17: 51-7
48. Tung H, Giannotta SL, Chandrasoma PT, Zee CS. Recurrent intraparenchymal hemorrhages from angiographically occult vascular malformations. J Neurosurg. 1990. 73: 174-80
49. Vitaz TW, Gaskill-Shipley M, Tomsick T, Tew JM. Utility, safety, and accuracy of intraoperative angiography in the surgical treatment of aneurysms and arteriovenous malformations. AJNR Am J Neuroradiol. 1999. 20: 1457-61
50. Wakai S, Ueda Y, Inoh S, Nagai M. Angiographically occult angiomas: A report of thirteen cases with analysis of the cases documented in the literature. Neurosurgery. 1985. 17: 549-56
51. Willinsky R, Lasjaunias P, Comoy J, Pruvost P. Cerebral micro arteriovenous malformations (mAVMs). Review of 13 cases. Acta Neurochir (Wien). 1988. 91: 37-41
52. Willinsky R, Terbrugge K, Montanera W, Wallace C, Aggarwal S.editors. Micro-arteriovenous malformations of the brain: Superselective angiography in diagnosis and treatment. AJNR Am J Neuroradiol. 1992. 13: 325-30
53. Wong GK, Siu DY, Ahuja AT, King AD, Yu SC, Zhu XL. Comparisons of DSA and MR angiography with digital subtraction angiography in 151 patients with subacute spontaneous intracerebral hemorrhage. J Clin Neurosci. 2010. 17: 601-5
54. Yaşagil MG.editors. Microneurosurgery. Volume IIIA. AVM of the Brain, History, Embryology, Pathological Considerations, Hemodynamics, Diagnostic Studies, Microsurgical Anatomy. New York: Thieme; 1987. p.
55. Zaidi HA, Abla AA, Nakaji P, Chowdhry SA, Albuquerque FC, Spetzler RF. Indocyanine green angiography in the surgical management of cerebral arteriovenous malformations: Lessons learned in 130 consecutive cases. Neurosurgery. 2014. 10 Suppl 2: 246-51