- Department of Neurological Surgery, Juntendo University Urayasu Hospital, Urayasu, Japan.
- Division of Radiological Technology, Medical Satellite Yaesu Clinic, Tokyo, Japan.
Correspondence Address:
Satoshi Tsutsumi, Department of Neurological Surgery, Juntendo University Urayasu Hospital, Urayasu, Japan.
DOI:10.25259/SNI_1161_2021
Copyright: © 2022 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Satoshi Tsutsumi1, Senshu Nonaka1, Hideo Ono2, Hisato Ishii1. Vertebral artery compression of the medulla oblongata: A benign radiological finding?. 04-Feb-2022;13:36
How to cite this URL: Satoshi Tsutsumi1, Senshu Nonaka1, Hideo Ono2, Hisato Ishii1. Vertebral artery compression of the medulla oblongata: A benign radiological finding?. 04-Feb-2022;13:36. Available from: https://surgicalneurologyint.com/?post_type=surgicalint_articles&p=11374
Abstract
Background: To the best of our knowledge, no study has documented the natural history of rostral medullary compression of the vertebral artery (RMCVA) as radiological finding. The aim of this study was to explore it.
Methods: A total of 57 patients with RMCVA and not presenting symptoms of medullary compression syndrome were enrolled. These participants underwent cerebral magnetic resonance imaging with contrast, and 19 of them who were followed for 5.7 ± 1.9 years (range: 3.0–10.3 years) were analyzed in detail. For comparison, clinical courses of two other patients with vertebrobasilar dolichoectasia (VBDE) were presented.
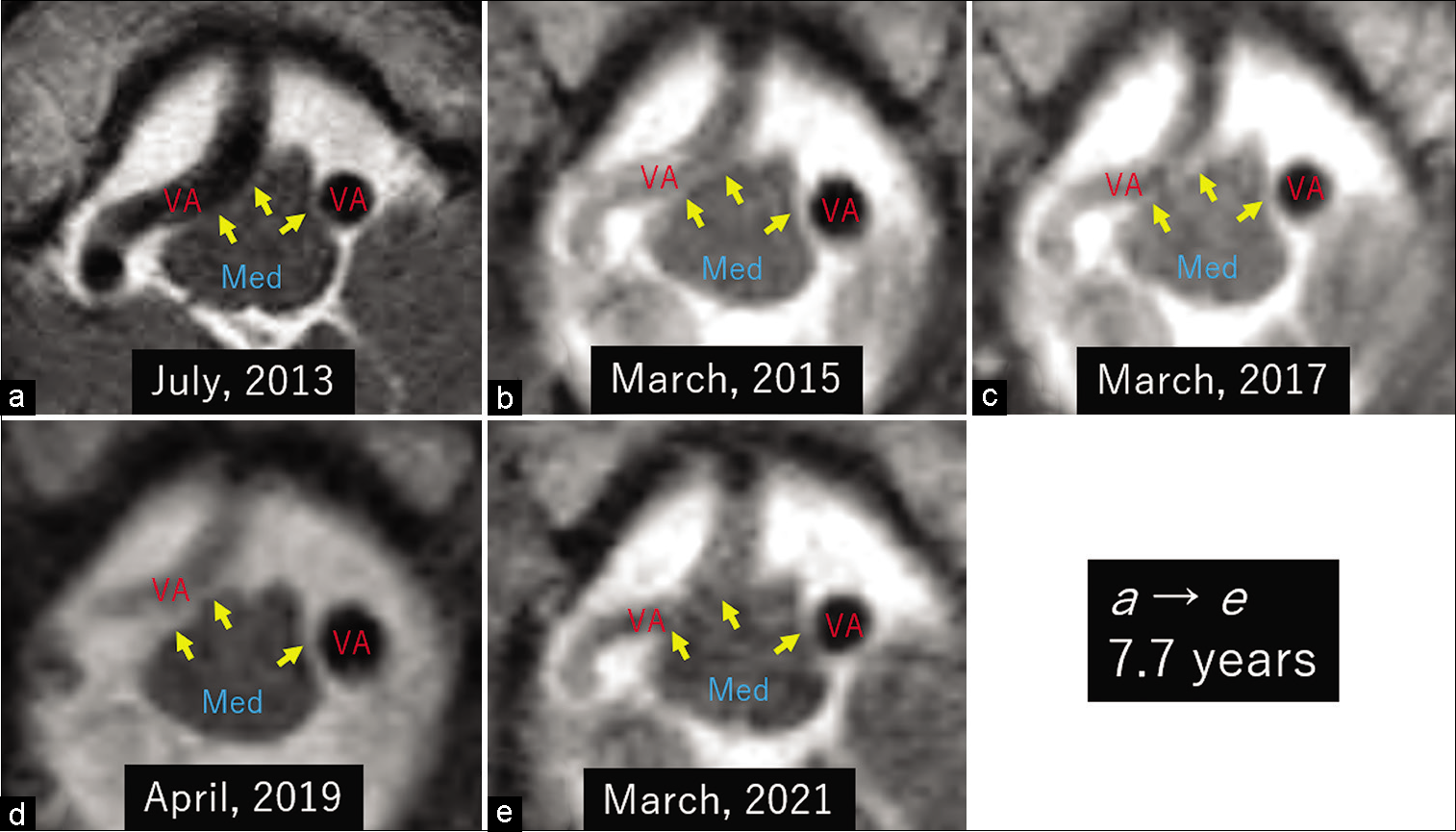
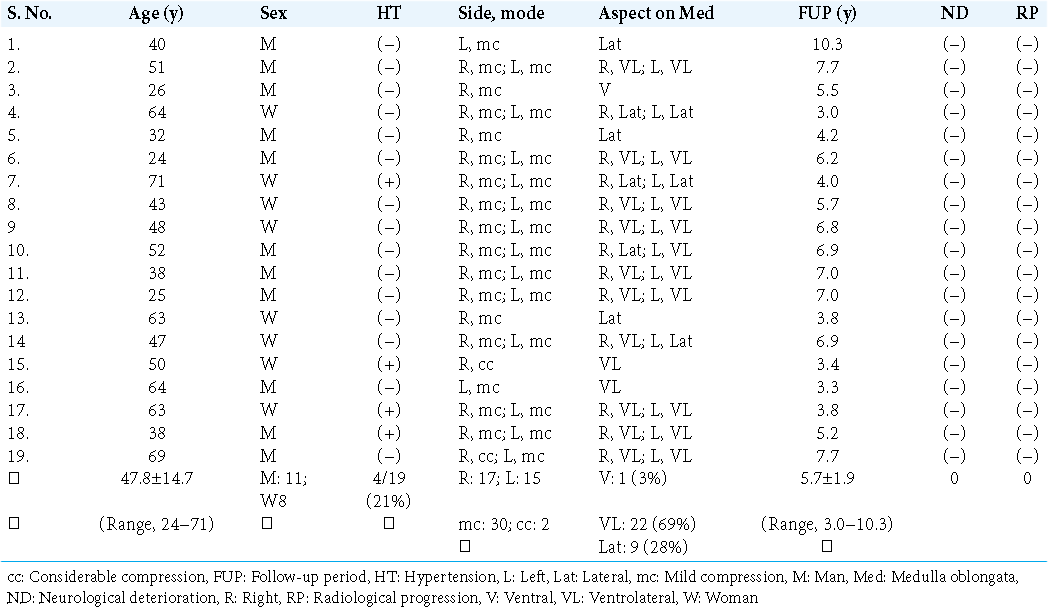
Results: RMCVA was well delineated in all 57 patients. In the 19 patients analyzed, RMCVA was found in 17 sides on the right and 15 on the left. Moreover, the ventrolateral medulla was the most frequent compression site, and it was found in 69% of cases, with 84.2% presenting as mild compression and 15.8% as considerable compression. During the follow-up period, no patients showed neurological deterioration or radiological progression. In contrast, the two VBDE patients demonstrated both neurological and radiological progressions during the follow-up period.
Conclusion: Unlike VBDE, RMCVA seems to be a benign condition without progression, even when with a considerable compression. Degree of the compression in RMCVA may not be relevant to the patient’s neurological status.
Keywords: Magnetic resonance imaging, Medullary compression, Natural history, Vertebral artery
INTRODUCTION
The medulla oblongata forms the lower part of the brainstem. The upper limit of it is adjacent to the bulbous pons, while the lower limit is estimated to lie approximately 3.4 mm caudal to the olivary inferior margin and 6.4 mm caudal to the obex or 10–12 mm above the foramen magnum plane.[
Dolichoectasia is an arterial disease causing abnormal dilation and tortuosity in affected vessels, which more frequently affects the vertebrobasilar system than the anterior circulation.[
Thus, the aim of the present study was to explore asymptomatic RMCVA in comparison with VBDE.
MATERIALS AND METHODS
The present retrospective study initially involved 89 outpatients and two VBDE patients. Patients in the former group, who presented with headaches, focal seizures, transient dizziness, tinnitus, hearing disturbance, and hemisensory disturbances, visited our hospital outpatient department from September 2010 to September 2013. Before MRI examination, detailed interviews and neurological evaluations confirmed that none of the 89 patients had a history of the previous cerebral infarction, intracerebral hemorrhage, meningitis, neurodegenerative disease, traumatic brain injury, hydrocephalus, or brain tumor. They were excluded using conventional MRI examination with axial T1- and T2-weighted imaging, T2 gradient echo, fluid-attenuated inversion recovery, diffusion-weighted sequences. Then, the patients underwent contrast studies through intravenous gadolinium infusion (0.1 mmol/kg) on T1-weighted sequences of the axial, coronal, and sagittal sections. Consecutively, 57 of the 89 patients (64.0%) were determined to have radiological RMCVA. Patients suspected of having MCS were not found. Following exclusion, the patient population finally comprised 30 men and 27 women, with a mean age of 50.0 ± 17.1 years (range: 18–77 years) and a mean body mass index (BMI) of 22.5 ± 3.8 (range: 14.1–32.7). Medically controlled hypertension, diabetes mellitus, and dyslipidemia and smoking history were found in 4 (7.0%), 1 (1.8%), 2 (3.5%), and 1 (1.8%) patient, respectively [
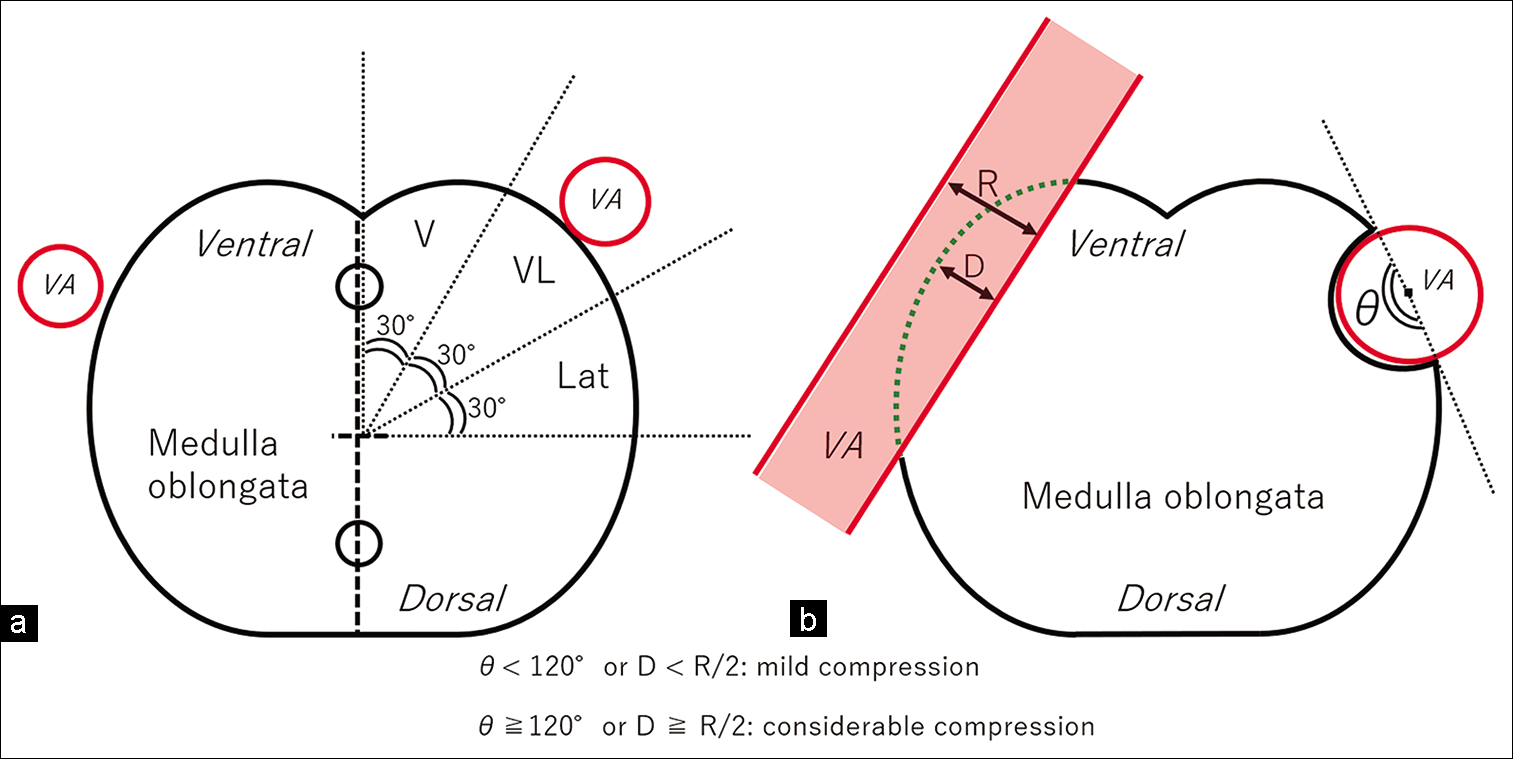
Figure 1:
Schematic drawings explaining the methodology for localizing the site of vertebral artery compression on the rostral medulla oblongata, carried out on the anterolateral 1/4 area of the axial plane divided into three parts (a) and that for defining the degree of medullary compression in the longitudinal or transverse dimension (b). D: maximal dimension of the hypothetically unaffected, intact medulla oblongata at the vertebral artery compression site, Lat: Lateral, R: Maximal vertebral artery dimension at the medullary compression site, V: Ventral, VA: Vertebral artery, VL: Ventrolateral; dotted line: hypothetically intact contour of the medulla oblongata.
Two patients in the latter group visited our hospital, were diagnosed with VBDE, and were followed from June 2010 to July 2013 and from September 2015 to March 2021.
All imaging sequences were performed using a 3.0 T MR scanner (Achieva R2.6; Philips Medical Systems, Best, The Netherlands). Imaging data were then transferred to a workstation (Virtual Place Lexus 64, 64th edition; AZE, Tokyo, Japan) and independently analyzed by three of the authors (S.T., S.N., and H.I.). Afterward, the Wilcoxon signed-rank test was used for statistical analyses.
The present study was performed in accordance with the guidelines of our institution for human research, and written informed consent was obtained from all the patients before the start of the study.
RESULTS
On axial T2- and postcontrast T1-weighted axial and coronal MRI images, RMCVA was well delineated in all 57 patients [
Figure 3:
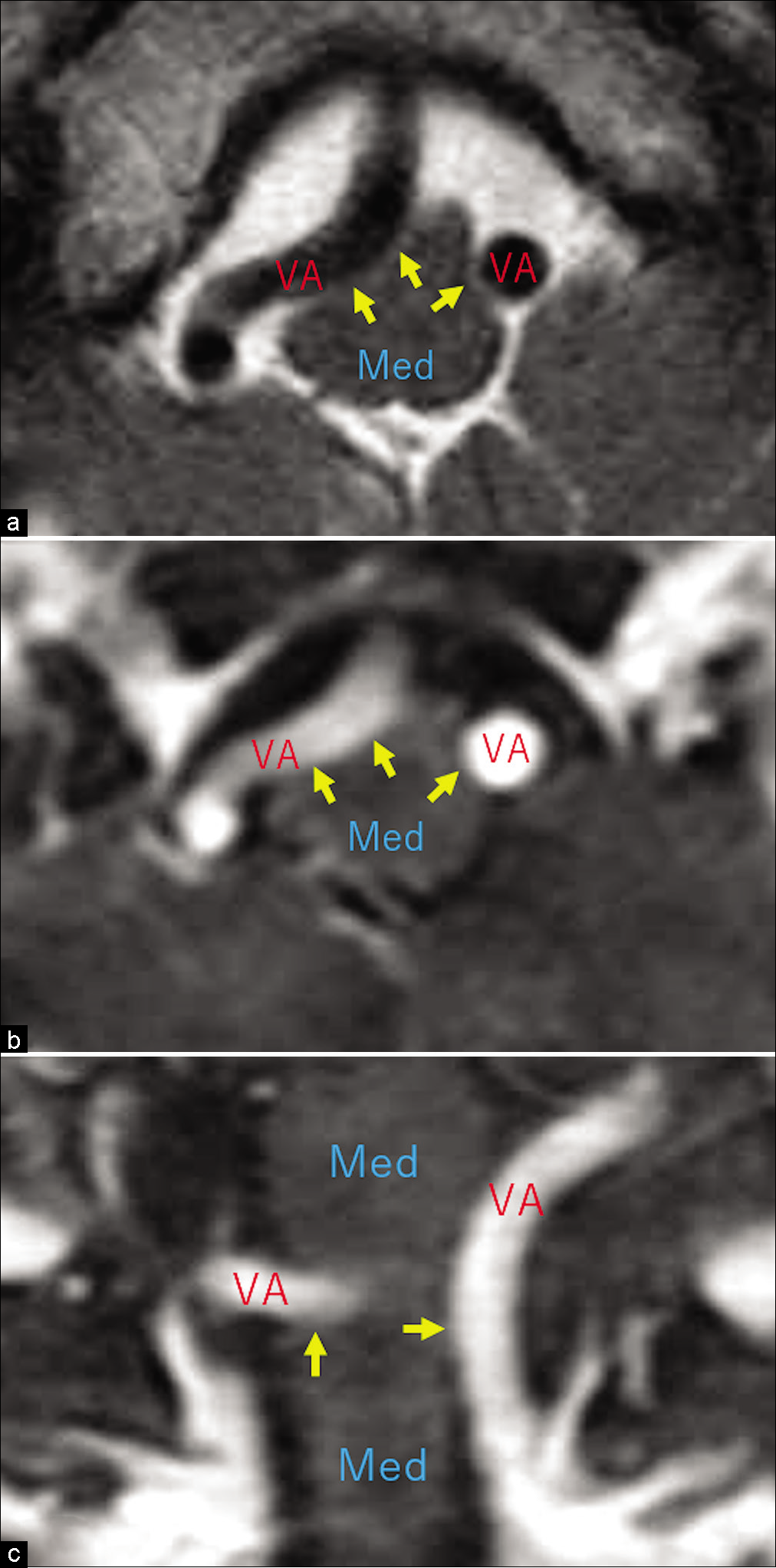
Postcontrast axial T1-weighted magnetic resonance images of different patients showing mild vertebral artery compression of the rostral medulla oblongata (a-f, arrows) in the ventral (a and b), ventrolateral (c and d), and lateral (e and f) aspects. CeT: Cerebellar tonsil, Med: Medulla oblongata, VA: Vertebral artery.
Figure 4:
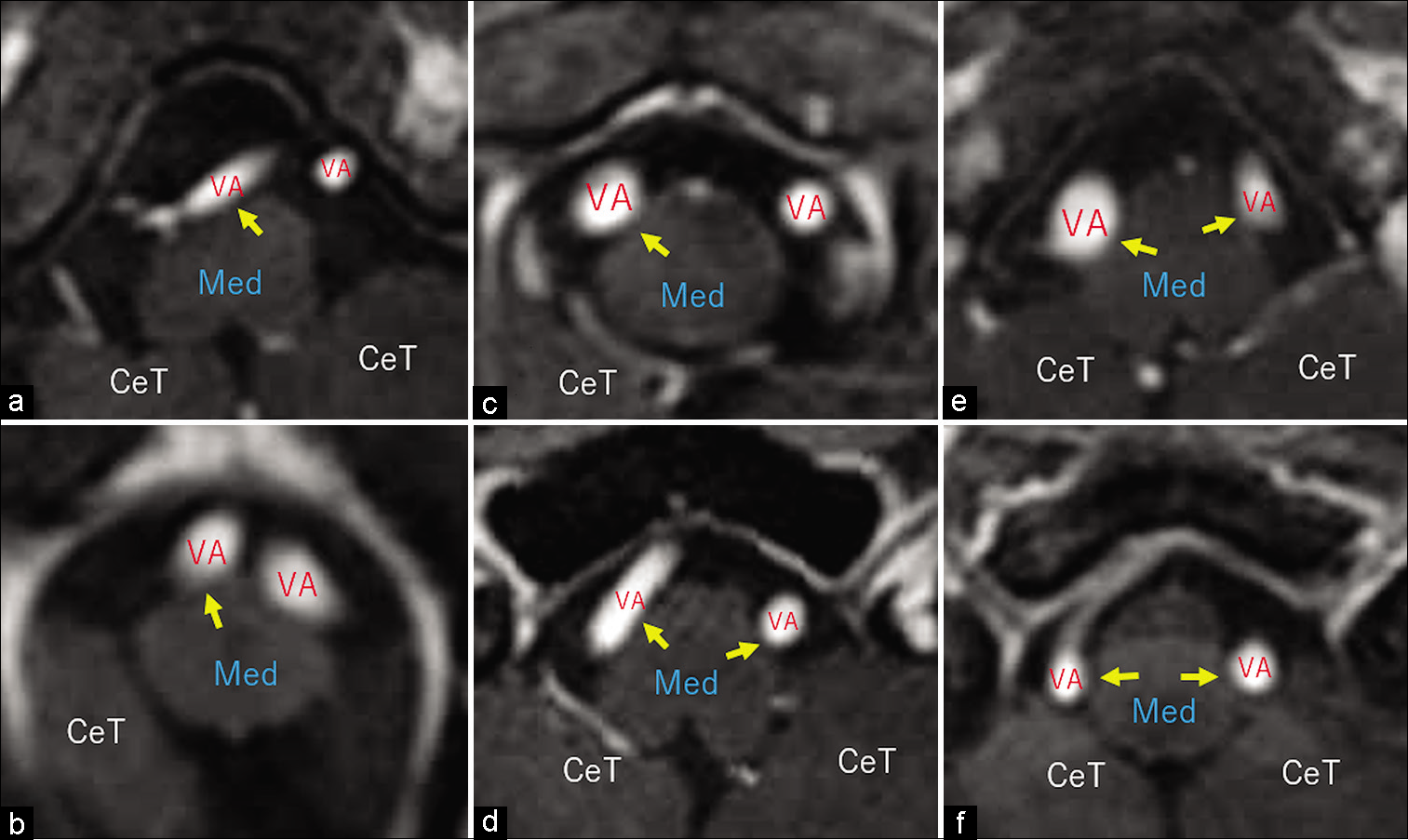
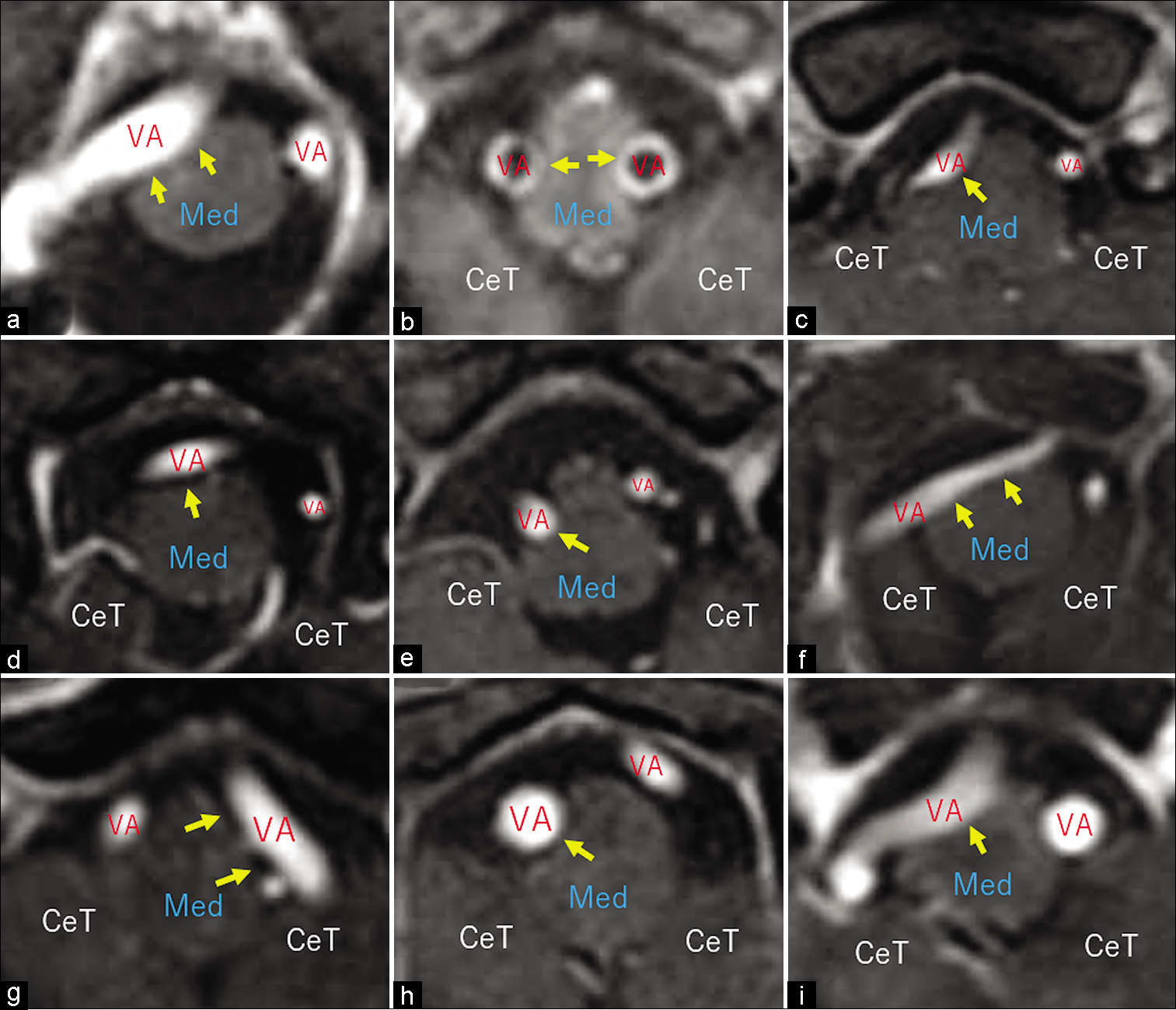
Postcontrast axial T1-weighted magnetic resonance images of different patients showing the vertebral arteries with variable diameters considerably compressing the rostral medulla oblongata (a-i, arrows) on the right (a, c-f, i), left (g), and both sides (b). CeT: Cerebellar tonsil, Med: Medulla oblongata, VA: Vertebral artery.
VBDE cases
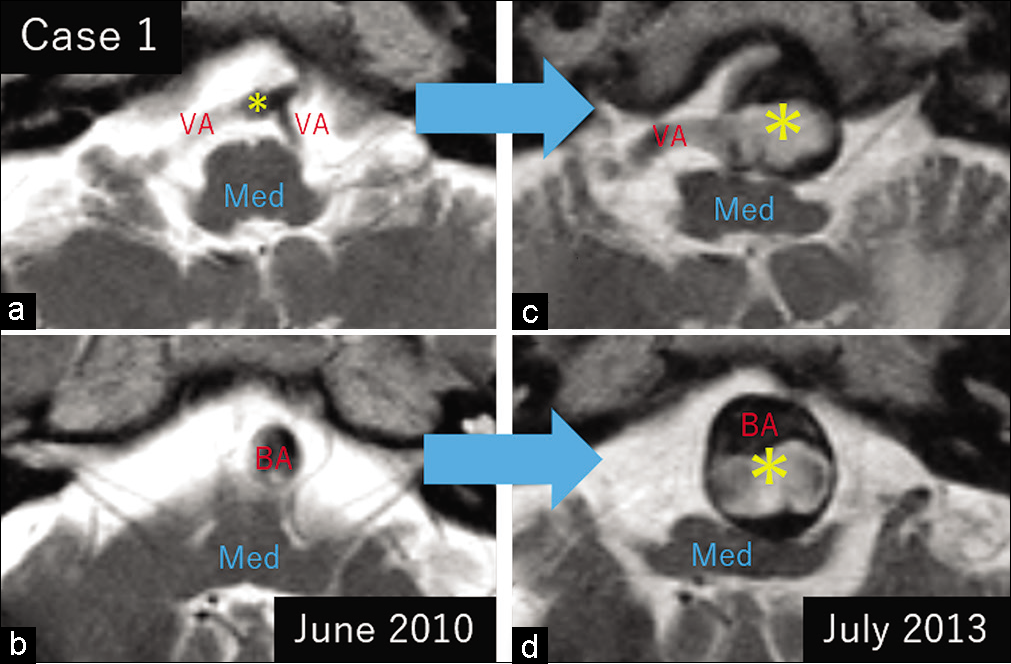
Case 1
A 70-year-old hypertensive man suffering from cerebral infarction presented with motor paresis in the left upper extremity and dysarthria. Cerebral MRI showed a VBDE mildly compressing the rostroventral medulla [
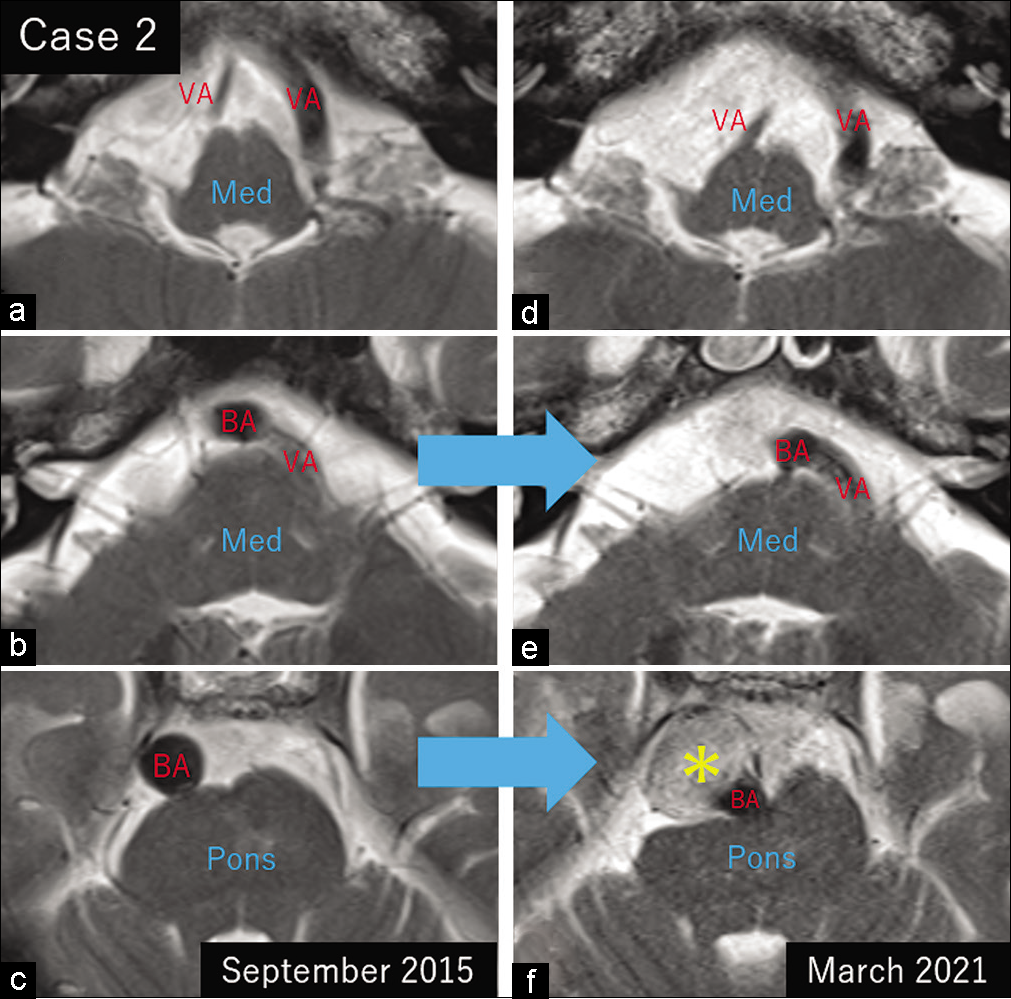
Case 2
A 75-year-old asymptomatic man underwent cerebral MRI for physical check-up and was diagnosed with VBDE and mild pontine compression [
DISCUSSION
In the present study, there were no patients with RMCVA presenting with neurological deterioration or radiological progression, even when with considerable degrees of compression. In contrast, as with the previous reports, patients with VBDE showed both neurological and radiological progressions.[
This study had several limitations. First, it was performed retrospectively in small populations, and the participants were not randomly assigned. In addition, the study population consisted of patients with inhomogeneous sex and age distributions. Finally, there were few patients with hypertension, diabetes mellitus, dyslipidemia, and smoking history in the RMCVA population, which made it difficult to evaluate them using a statistical approach. Even with these limitations, we believe that the findings of this study can help to better understand the characteristics and natural history of RMCVA.
CONCLUSION
Unlike VBDE, RMCVA seems to be a benign condition without progression even when with a considerably tortuous appearance. Degree of the compression may not be relevant to the patient’s neurological status. RMCVA conditions may be formed in association with aging and obesity.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Akaishi T, Kiyomoto H, Abe M, Okuda H, Ishikawa K, Endo T. A 29-year-old woman with recurrent pregnancy-induced hypertension based on vascular compression of the medulla oblongata. Intern Med. 2019. 58: 2257-61
2. Carlson EJ, Tominaga Y, Ivancic PC, Panjabi MM. Dynamic vertebral artery elongation during frontal and side impacts. Spine J. 2007. 7: 222-8
3. Cravo SL, Possas OS, Ferreira-Neto ML. Rostral ventrolateral medulla: An integrative site for muscle vasodilation during defense-altering reactions. Cell Mol Neurobiol. 2003. 23: 579-95
4. Deistung A, Schäfer A, Schweser F, Biedermann U, Güllmar D, Trampel R, Turner R. High-resolution MR imaging of the human brainstem in vivo at 7 Tesla. Front Hum Neurosci. 2013. 7: 710
5. DelRosso L, Gonzalez-Toledo E, Chesson AL, Hoque R. Positional central apnea and vascular medullar compression. Neurology. 2012. 79: 2156-7
6. Ding S, Yan X, Guo H, Yin F, Sun X, Yang A. Morphological characteristics of the vertebrobasilar artery system in patients with hemifacial spasm and measurement of bending length for evaluation of tortuosity. Clin Neurol Neurosurg. 2020. 198: 106144
7. Fontes MA, Silva LC, Campagnole-Santos MJ, Khosla MC, Guertzenstein PG, Santos RA. Evidence that angiotensin-(1-7) plays a role in the central control of blood pressure at the ventro-lateral medulla acting through specific receptors. Brain Res. 1994. 665: 175-80
8. Gutierrez J, Sacco RL, Wright CB. Dolichoectasia-an evolving arterial disease. Nat Rev Neurol. 2011. 7: 41-50
9. Ikeda K, Nakamura Y, Hirayama T, Sekine T, Nagata R, Kano O. Cardiovascular risk and neuroradiological profiles in asymptomatic vertebrobasilar dolichoectasia. Cerebrovasc Dis. 2010. 30: 23-8
10. Ivancic PC, Ito S, Tominaga Y, Carlson EJ, Rubin W, Panjabi MM. Effect of rotated head posture on dynamic vertebral artery elongation during simulated rear impact. Clin Biomech (Bristol Avon). 2006. 21: 213-20
11. Kutty RK, Yamada Y, Takizawa K, Kato Y. Medullary compression due to ectatic vertebral artery-case report and review of literature. J Stroke Cerebrovasc Dis. 2020. 29: 104460
12. Li Q, Xie P, Yang WS, Yan B, Davis S, Caplan LR. Vertebral artery compression syndrome. Front Neurol. 2019. 10: 1075
13. Manava P, Naraghi R, Schmieder R, Fahlbusch R, Doerfler A, Lell MM. 3D-visualization of neurovascular compression at the ventrolateral medulla in patients with arterial hypertension. Clin Neuroradiol. 2021. 31: 335-45
14. Middlebrooks EH, Yagmurlu K, Bennett JA, Bidari S. Normal relationship of the cervicomedullary junction with the obex and olivary bodies: A comparison of cadaveric dissection and in vivo diffusion tensor imaging. Surg Radiol Anat. 2015. 37: 493-7
15. Miyazaki M, Hashimoto T, Sakurama N, Yoshimoto T, Tayama M, Kuroda Y. Central sleep apnea and arterial compression of the medulla. Ann Neurol. 1991. 29: 564-5
16. Naraghi R, Gaab MR, Walter GF, Kleinberg B. Arterial hypertension and neurovascular compression at the ventrolateral medulla. A comparative microanatomical and pathological study. J Neurosurg. 1992. 77: 103-12
17. Nezu T, Hosomi N, Kuzume D, Naito H, Aoki S, Morimoto Y. Effects of vascular compression on the rostral ventrolateral medulla for blood pressure variability in stroke patients. J Hypertens. 2020. 38: 2443-50
18. Nibu N, Cholewicki J, Panjabi MM, Babat LB, Grauer JN, Kotha R. Dynamic elongation of the vertebral artery during an in vivo whiplash simulation. Eur Spine J. 1997. 6: 286-9
19. Nicholas JS, D’Agostino SJ, Patel SJ. Arterial compression of the retro-olivary sulcus of the ventrolateral medulla in essential hypertension and diabetes. Hypertension. 2005. 46: 982-5
20. Nishitaka M, Hirashima Y, Tomita T, Futatsuya R, Horie Y, Endo S. Measurement of basilar artery bending and elongation by magnetic resonance cerebral angiography: Relationship to age, sex and vertebral artery dominance. Arch Gerontol Geriatr. 2004. 38: 251-9
21. Passero S, Rossi S, Giannini F, Nuti D. Brain-stem compression in vertebrobasilar dolichoectasia. A multimodal electrophysiological study. Clin Neurophysiol. 2001. 112: 1531-9
22. Quisling RG, Quisling SG, Mickle JP. Obex/nucleus gracilis position: Its role as a marker for the cervicomedullary junction. Pediatr Neurosurg. 1993. 19: 143-50
23. Sadashiva N, Shukla D, Bhat DI, Devi BI. Vertebral artery dolichoectasia with brainstem compression: Role of microvascular decompression in relieving pyramidal weakness. Acta Neurochir (Wien). 2016. 158: 797-801
24. Salvi F, Mascalchi M, Bortolotti C, Meletti S, Plasmati R, Rubboli G. Hypertension, hyperekplexia, and pyramidal paresis due to vascular compression of the medulla. Neurology. 2000. 55: 1381-4
25. Savitz SI, Ronthal M, Caplan LR. Vertebral artery compression of the medulla. Arch Neurol. 2006. 63: 234-41
26. Schulz R, Fegbeutel C, Althoff A, Traupe H, Grimminger F, Seeger W. Central sleep apnoea and unilateral diaphragmatic paralysis associated with vertebral artery compression of the medulla oblongata. J Neurol. 2003. 250: 503-5
27. Sendeski MM, Consolim-Colombo FM, Leite CC, Rubira MC, Lessa P, Krieger EM. Increased sympathetic nerve activity correlates with neurovascular compression at the rostral ventrolateral medulla. Hypertension. 2006. 47: 988-95
28. Tomasello F, Alafaci C, Salpietro FM, Longo M. Bulbar compression by an ectatic vertebral artery: A novel neurovascular construct relieved by microsurgical decompression. Neurosurgery. 2005. 56: 117-24
29. Watters MR, Burton BS, Turner GE, Cannard KR. MR screening for brain stem compression in hypertension. AJNR Am J Neuroradiol. 1996. 17: 217-21
30. Zizka J, Ceral J, Elias P, Tintera J, Klzo L, Solar M. Vascular compression of rostral medulla oblongata: Prospective MR imaging study in hypertensive and normotensive subjects. Radiology. 2004. 230: 65-9