- Department of Neurosurgery, Harvard Medical School, Brigham and Women's Hospital, Boston, Massachusetts, USA
Correspondence Address:
Parviz Dolati
Department of Neurosurgery, Harvard Medical School, Brigham and Women's Hospital, Boston, Massachusetts, USA
DOI:10.4103/2152-7806.170023
Copyright: © 2015 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Dolati P, Gokoglu A, Eichberg D, Zamani A, Golby A, Al-Mefty O. Multimodal navigated skull base tumor resection using image-based vascular and cranial nerve segmentation: A prospective pilot study. Surg Neurol Int 19-Nov-2015;6:172
How to cite this URL: Dolati P, Gokoglu A, Eichberg D, Zamani A, Golby A, Al-Mefty O. Multimodal navigated skull base tumor resection using image-based vascular and cranial nerve segmentation: A prospective pilot study. Surg Neurol Int 19-Nov-2015;6:172. Available from: http://surgicalneurologyint.com/surgicalint_articles/multimodal-navigated-skull-base-tumor-resection-using/
Abstract
Background:Skull base tumors frequently encase or invade adjacent normal neurovascular structures. For this reason, optimal tumor resection with incomplete knowledge of patient anatomy remains a challenge.
Methods:To determine the accuracy and utility of image-based preoperative segmentation in skull base tumor resections, we performed a prospective study. Ten patients with skull base tumors underwent preoperative 3T magnetic resonance imaging, which included thin section three-dimensional (3D) space T2, 3D time of flight, and magnetization-prepared rapid acquisition gradient echo sequences. Imaging sequences were loaded in the neuronavigation system for segmentation and preoperative planning. Five different neurovascular landmarks were identified in each case and measured for accuracy using the neuronavigation system. Each segmented neurovascular element was validated by manual placement of the navigation probe, and errors of localization were measured.
Results:Strong correspondence between image-based segmentation and microscopic view was found at the surface of the tumor and tumor-normal brain interfaces in all cases. The accuracy of the measurements was 0.45 ± 0.21 mm (mean ± standard deviation). This information reassured the surgeon and prevented vascular injury intraoperatively. Preoperative segmentation of the related cranial nerves was possible in 80% of cases and helped the surgeon localize involved cranial nerves in all cases.
Conclusion:Image-based preoperative vascular and neural element segmentation with 3D reconstruction is highly informative preoperatively and could increase the vigilance of neurosurgeons for preventing neurovascular injury during skull base surgeries. Additionally, the accuracy found in this study is superior to previously reported measurements. This novel preliminary study is encouraging for future validation with larger numbers of patients.
Keywords: Blood vessels, cranial nerves, image segmentation, magnetic resonance imaging, neuronavigation, skull base neoplasms
BACKGROUND
Tumors of the skull base region represent some of the most challenging lesions to resect. Because skull base tumors are critically and deeply located, access and total removal have always been the main challenges in the surgical management of such cases. Additionally, invasion of bone and critical neurovascular structures often impedes complete resection of skull base neoplasms. For these reasons, it is unsurprising that skull base procedures entail a risk of new permanent cranial nerve deficits and may necessitate a subtotal resection, which may lead to the regrowth of the residual tumor.[
Interactive intraoperative image guidance is rapidly becoming an indispensable neurosurgical technique.[
Although current image guidance is primarily dependent on preoperative magnetic resonance images (MRIs) and does not account for brain shift during surgery,[
Here, we provide a prospective pilot study in which ten patients with skull base tumors underwent image-based preoperative vascular and neural element segmentation with three-dimensional (3D) reconstruction for intraoperative guidance, as well as intraoperative validation of each segmented neural or vascular element. We aimed to determine the accuracy and utility of image-based preoperative segmentation in skull base tumor resections. We hypothesized that this preoperative segmentation can improve resection rate, minimize surgical morbidities, and increase the accuracy of localization compared to previously reported accuracies.
PROCEDURE
Study size and setting
After approval by the Hospital Institutional Review Board, a total of 10 patients with skull base tumors underwent image-based preoperative vascular neural elements and tumor segmentation with 3D reconstruction. These procedures took place at a single institution from December 2013 to May 2014, and data were prospectively collected and retrospectively reviewed.
Participants
Patient selection for the use of image-guided techniques was based solely on surgeon preference; there were no qualifying or disqualifying factors. The authors have no personal interest in the system used. Adult patients with skull base tumors of any type were included.
Image acquisition
The patients underwent preoperative 3T MRI, which included thin section 3D space T2, 3D time of flight, magnetization-prepared rapid acquisition gradient echo, and dynamic computed tomography (CT) angiography. An expert neuroradiologist reviewed the images before segmentation. Subsequently, imaging sequences were loaded in either BrainLAB iPlan Net (Brainlab AG, Munich, Germany) for 8 patients, or Stryker (Stryker, Kalamazoo, Michigan, USA) for 2 patients, for fusion, segmentation, and preoperative planning. After fixing patients’ heads with a Mayfield 3-point fixation system, careful patient registration to the intraoperative neuronavigation system, and surgical exposure, each segmented neural or vascular element was validated by manual placement of the navigation probe over each target in different stages of tumor resection, and screenshots were captured. Errors of localization were measured in mm.
Study design and data sources
This pilot study was a prospective validation study. Primary endpoints were defined as correspondence between image-based segmentation and microscopic view at the surface of the tumor and tumor-brain interfaces, major arteries, veins, and venous sinuses or cranial nerves with measurements of errors in mm [
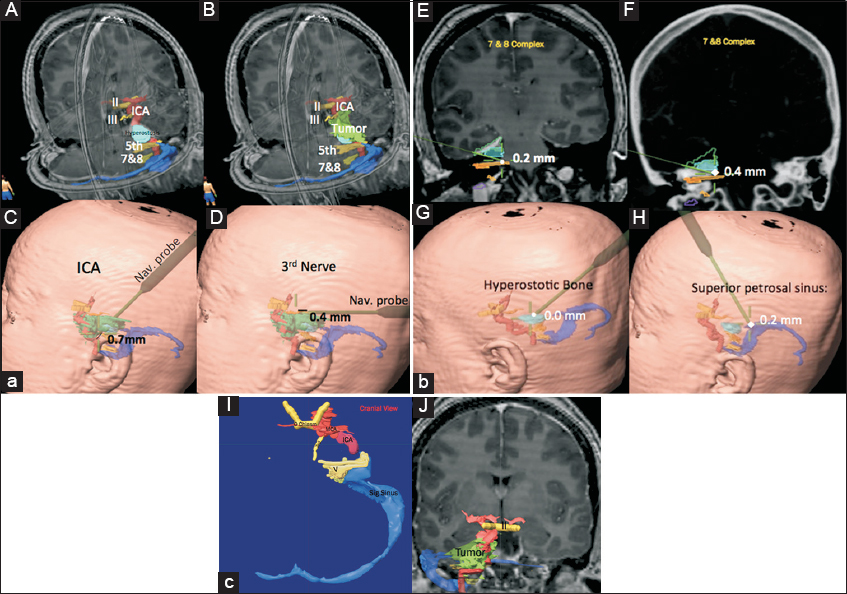
Figure 1
(a) Preoperative segmentation of the right petrous apex meningioma; Panel A, showing only the hyperostosis and Panel B, showing tumor at the top of the hyperostosis along with segmentation of the cranial nerves, II, III, V and VII and VIII complex. Panels C and D are intraoperative localization of the internal carotid artery and cranial nerve III, which showing also navigation errors from the targets (0.7 mm and 0.4 mm, respectively). (b) Panels E and F show cranial nerves 7 and 8 complex in magnetic resonance imaging and computed tomography scan, respectively. Panel G, shows navigation of the hyperostotic bone after resection of the soft tissue and finally Panel H shows localization of the superior petrosal sinus. The numbers on the panels are the accuracy errors from the target. (c) Panels J and I show object creation after segmentation of the involved regions; Panel I shows cranial view with tumor removed and Panel J shows frontal view with tumor in place
Statistical methods
Patients’ clinical and tumor characteristics (age, sex, tumor pathology, and, completeness of resection) were evaluated using the mean and range for continuous variables and the frequency count for categorical factors. The Sørensen–Dice index was used to compare the correspondence between image-based segmentation and microscopic view at the surface of the tumor and tumor-normal brain interfaces, as well as the major arteries, veins, and venous sinuses. The average accuracy of localization was evaluated using the mean and standard deviation (SD).
RESULTS
Descriptive data
Participants
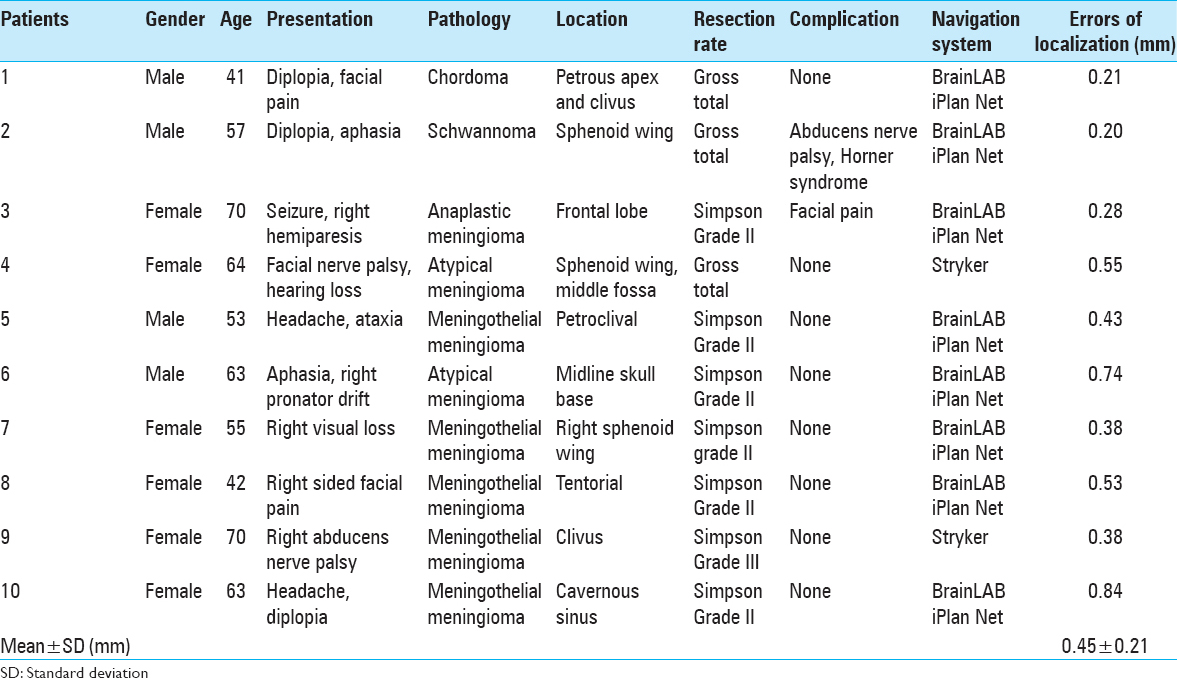
The mean age was 57.8 years, and there were 4 male and 6 female patients [
Tumor pathology
Five patients (50%) had meningothelial meningioma WHO Grade I, 2 patients (20%) had atypical meningioma WHO Grade II, 1 patient (10%) had chordoma, 1 patient (10%) had left Meckel's cave Schwannoma, and 1 patient (10%) had anaplastic meningioma WHO Grade III.
Operative data
Reasonable correspondence between image-based segmentation and the microscopic view was found at the surface of the tumor and tumor-normal brain interfaces in all cases (Dice-coefficient = 1). The accuracy of the measurements was 0.45 ± 0.21 mm (mean ± SD). This information reassured the surgeon and prevented vascular injury during different stages of tumor resection. Preoperative segmentation of the related cranial nerves was technically possible in only 80% of cases. However, image-guidance helped the surgeon localize the involved cranial nerves in all of these segmented cases reliably.
Resection rate
Eight patients (90%) had either gross total resection or Simpson Grade II meningioma resection, and 1 patient (10%) had Simpson Grade III meningioma resection.
Complications
The postoperative complication was noticed in only 1 patient who suffered transient partial abducens nerve palsy and Horner syndrome.
Review of literature
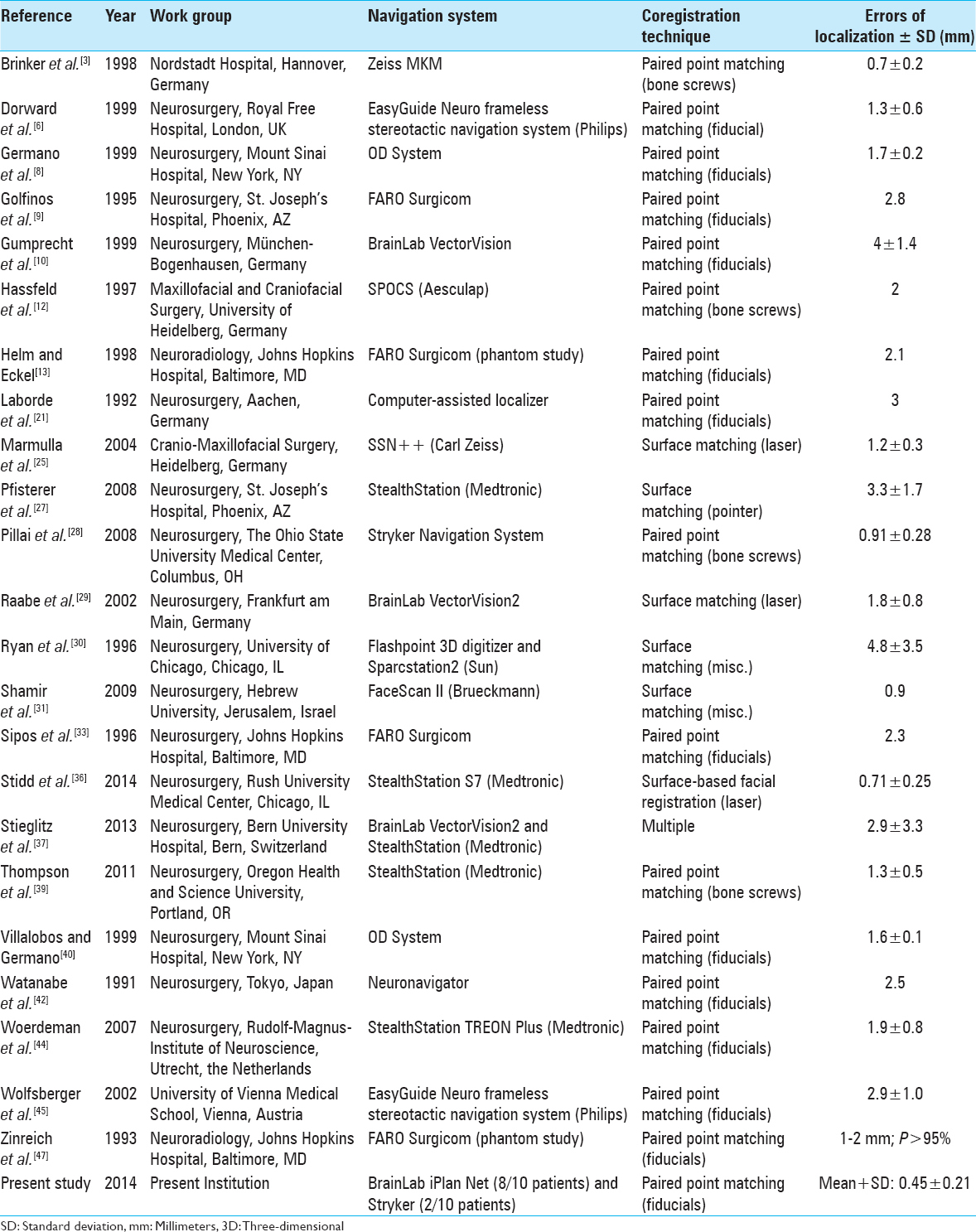
The mean ± SD of previously reported errors of localization in studies of frameless stereotactic neuronavigation (not including the present study) was 2.07 ± 1.16 mm [
DISCUSSION
Our preliminary data demonstrates that the image-based preoperative vascular and neural element segmentation, especially with 3D reconstruction, is highly informative for both preoperative planning and intraoperative navigation, particularly with complex tumors that invade and distort neurovascular and osseous structures. Image-guidance based neuronavigation will enhance the efficacy and safety of skull base surgery and can aid in directing the surgeon's attention to the nearby neurovascular structures. Neuronavigation is increasingly utilized in neurosurgery.[
Vessels and cranial nerves that are invaded, encased, or displaced by tumor may nevertheless be visualized on MRIs and displayed. Therefore, complex skull base tumors, in particular, benefit greatly from image-based preoperative vascular and neural element segmentation with 3D reconstruction. A number of important aspects of skull base surgery are discussed in the following paragraphs [Figures
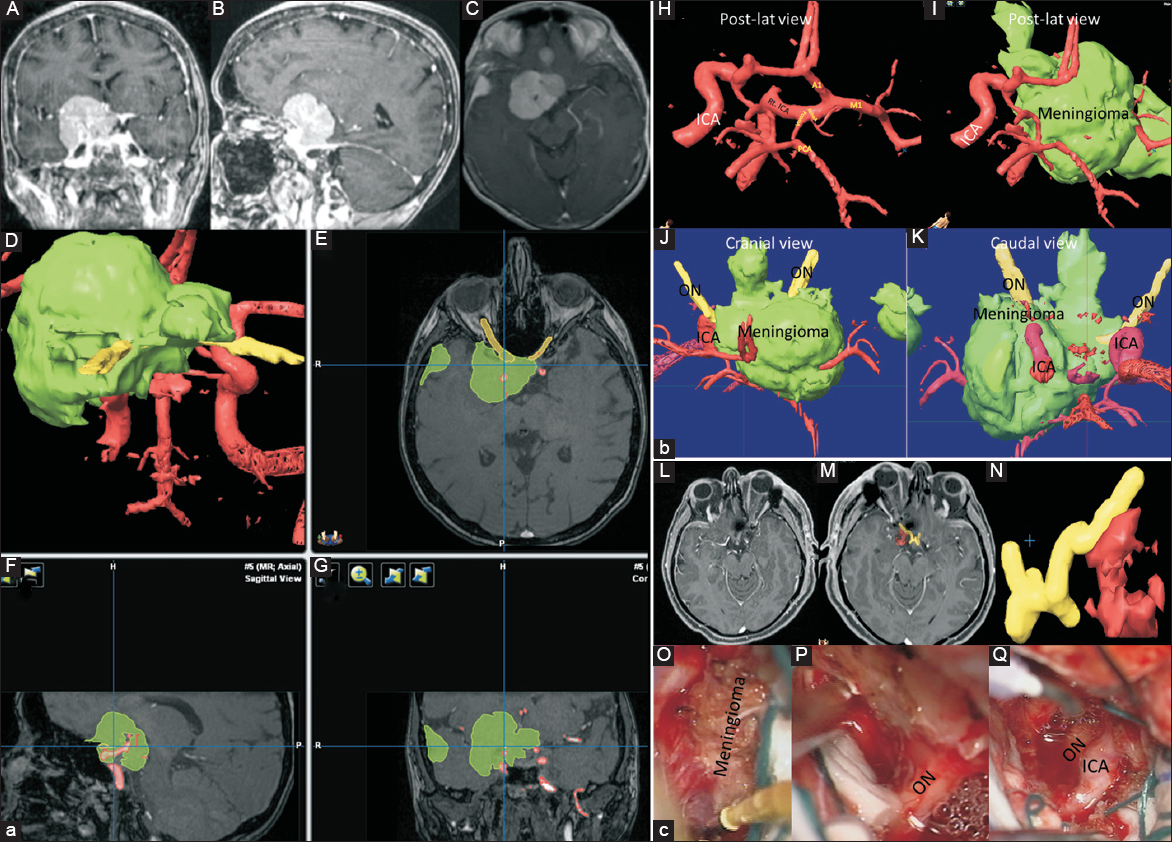
Figure 2
(a) A 55-year-old female presented with the progressive right visual loss. Panels A, B, and C show preoperative brain magnetic resonance imaging with gadolinium enhancement in coronal, sagittal, and axial views, respectively. Panels D, E, F, and G show preoperative segmentation of the tumor (green), optic and third nerves (yellow) and internal carotid artery and its branches using iPlan Net of BrainLAB system. (b) Panels H, I, J, and K show the preoperative segmentation of internal carotid artery and its branches (red), optic nerves (yellow), and tumor (green) of above–mentioned patient in detail. This patient underwent craniotomy and complete resection. (c) Panel L shows postoperative brain magnetic resonance imaging with gadolinium enhancement, Panels M (cranial view) and N (caudal view) show postoperative segmentation of optic nerve (yellow) and internal carotid artery (red), Panel O shows the meningioma encasing the right optic nerve and internal carotid artery and Panel P after decompression of the optic nerve and Panel Q, after decompressing of both optic nerve and internal carotid artery
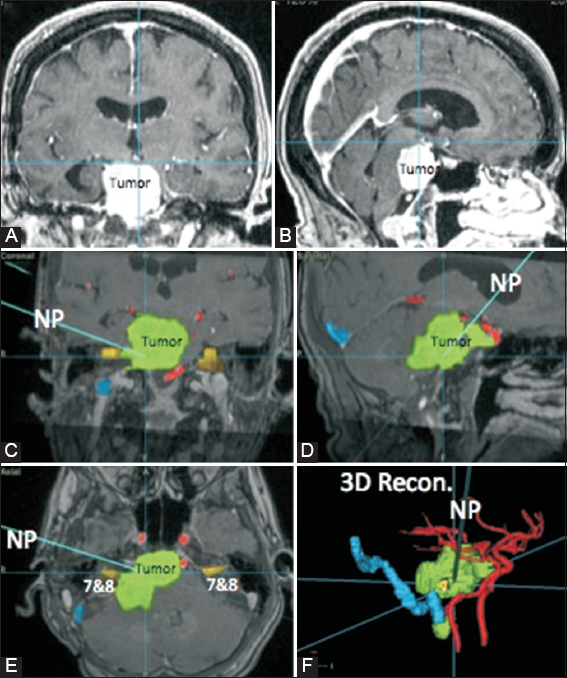
Figure 3
Intraoperative neuronavigation for a 70-year-old female [Case 9 of
Neuronavigation offers real time visualization of any region of interest, including tumors and anatomic landmarks. Such image guidance data are easily marked, stored, and reconstructed in a 3D mode or any plane. Accordingly, the surgeon can preoperatively define the landmarks. Preoperatively creating and planning the surgical vectors is critically important, helps minimize cortical trauma, and provides the ability to determine the required extent of the surgical exposure. Additionally, image-guided preoperative planning indicates the appropriateness of a particular skull base approach.
Although the significance of brain shift on preoperatively acquired image guidance and the subsequent need for intraoperative image updating is a controversial and unresolved issue, it has been known for some time that skull base lesions have a remarkably small extent of intraoperative shift due to their anchoring to the bony structures of the skull base. As the skull base tumors are firmly attached to the dura and bone, and the cranial nerves and the major blood vessels have relatively stable and protected position and course in this anatomic region, therefore, shift will not be a problem in neuronavigation in these cases. This feature indicates that image guidance offers a high degree of reliability for neuronavigation in the treatment of skull base lesions.[
Preliminary evaluations of the use of preoperative 3D image segmentation and patient registration have been reported for the surgery of aneurysms, meningioma, gliomas, pituitary tumors, cerebral cavernoma, and arteriovenous malformations.[
In our study, very good correspondence between image-based segmentation and the microscopic view was found at the surface of the tumor and tumor-normal brain interfaces in all cases [
In our experience, the prompt identification, and thus circumvention, of invaded, encased, or displaced vital neurovascular structures during tumor dissection is the most imperative advantage that is, offered by neuronavigation, especially when tumor encasing important neurovascular structure of the brain [Figures
The image-guided skull base surgery data demonstrates an improvement over comparable nonimage guided skull base surgery data. Several nonimage guided skull base surgery studies report gross total resection rates from 61% to 79%, new permanent cranial nerve deficits from 25% to 76%, and mortality rates from 0% to 11%,[
Therefore, we believe that the use of image-based preoperative vascular and neural element segmentation, especially with 3D reconstruction and intraoperative application of these data for navigation, as presented in our series might additionally help minimize the morbidity rate in patients suffering from extensive skull base lesions. Especially, in situations of complex anatomy or when the abnormality involves critical structures, navigational tools are likely to be beneficial, reducing operative time and increasing the safety of surgery. Image guidance can be very useful for enhancing the confidence of localizing vascular structures, tumor extension, and the osseous structures, particularly in less experienced neurosurgeons [Figures
Neuronavigation only provides surgical utility if the image guidance system data accurately corresponds to the perioperative patient anatomy. A previous retrospective review of the literature found that mean neuronavigation accuracies have ranged from 0.7 to 5 mm.[
Recently, one group has reported an accuracy similar to that obtained by paired point matching by bone screws, using the technique of preoperatively acquired 3D digital subtraction angiography (DSA) registered by the facial surface anatomy contained within the data set.[
Our group achieved a measurement accuracy of 0.45 ± 0.21 mm (mean ± SD), which is improved over previously reported accuracies ranging from 0.7 to 5 mm.[
For experienced surgeons, bony and neurovascular landmarks represent the best and most consistent navigational tools for avoiding neural and vascular compromise. However, using the currently described image-based vascular and cranial nerve segmentation technique confers several additional benefits, particularly for less experienced surgeons. First, the surgeon commits spending time for preoperative segmentation of the neurovascular elements in the course of access to the target tumor, which will aid in the understanding of these anatomical landmarks in advance. Second, these bony and neurovascular anatomical landmarks are not fixed in all cases. Therefore, preoperative segmentation will detect anatomical variations and warn the attending surgeon to start or continue the approach from a direction which has the least chance of causing vascular or cranial nerve injuries. Finally, the availability of a 3D segmented real time navigational tool is reassuring for less experienced neurosurgeons.
CONCLUSION
Image-based preoperative vascular and neural element segmentation, especially with 3D reconstruction, is highly informative for both preoperative planning and intraoperative navigation, particularly with complex skull base tumors that invade and distort neurovascular and osseous structures. Image-guidance based neuronavigation enhances the efficacy and safety of skull base surgery, in part by increasing the surgeon's vigilance, and helps to avoid vascular and neural injury during skull base surgeries. Additionally, the accuracy found in this study is superior to previously reported neuronavigation measurements, mostly because of the minimal brain shift encountered in skull base tumor surgeries. This novel preliminary study is encouraging for future prospective intraoperative validation with larger numbers of patients.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicst of interest.
References
1. Anxionnat R, Bracard S, Ducrocq X, Trousset Y, Launay L, Kerrien E. Intracranial aneurysms: Clinical value of 3D digital subtraction angiography in the therapeutic decision and endovascular treatment. Radiology. 2001. 218: 799-808
2. Bricolo AP, Turazzi S, Talacchi A, Cristofori L. Microsurgical removal of petroclival meningiomas: A report of 33 patients. Neurosurgery. 1992. 31: 813-28
3. Brinker T, Arango G, Kaminsky J, Samii A, Thorns U, Vorkapic P. An experimental approach to image guided skull base surgery employing a microscope-based neuronavigation system. Acta Neurochir (Wien). 1998. 140: 883-9
4. Cabrilo I, Bijlenga P, Schaller K. Augmented reality in the surgery of cerebral aneurysms: A technical report. Neurosurgery. 2014. 10: 252-60
5. Di Maio S, Rostomily R, Sekhar LN. Current surgical outcomes for cranial base chordomas: Cohort study of 95 patients. Neurosurgery. 2012. 70: 1355-60
6. Dorward NL, Alberti O, Palmer JD, Kitchen ND, Thomas DG. Accuracy of true frameless stereotaxy: In vivo measurement and laboratory phantom studies. Technical note. J Neurosurg. 1999. 90: 160-8
7. Dorward NL, Alberti O, Velani B, Gerritsen FA, Harkness WF, Kitchen ND. Postimaging brain distortion: Magnitude, correlates, and impact on neuronavigation. J Neurosurg. 1998. 88: 656-62
8. Germano IM, Villalobos H, Silvers A, Post KD. Clinical use of the optical digitizer for intracranial neuronavigation. Neurosurgery. 1999. 45: 261-9
9. Golfinos JG, Fitzpatrick BC, Smith LR, Spetzler RF. Clinical use of a frameless stereotactic arm: Results of 325 cases. J Neurosurg. 1995. 83: 197-205
10. Gumprecht HK, Widenka DC, Lumenta CB. BrainLab VectorVision Neuronavigation System: Technology and clinical experiences in 131 cases. Neurosurgery. 1999. 44: 97-104
11. Haerle SK, Daly MJ, Chan HH, Vescan A, Kucharczyk W, Irish JC. Virtual surgical planning in endoscopic skull base surgery. Laryngoscope. 2013. 123: 2935-9
12. Hassfeld S, Muehling J, Wirtz CR, Knauth M, Lutze T, Schulz HJ. Intraoperative guidance in maxillofacial and craniofacial surgery. Proc Inst Mech Eng H. 1997. 211: 277-83
13. Helm PA, Eckel TS. Accuracy of registration methods in frameless stereotaxis. Comput Aided Surg. 1998. 3: 51-6
14. Inoue D, Cho B, Mori M, Kikkawa Y, Amano T, Nakamizo A. Preliminary study on the clinical application of augmented reality neuronavigation. J Neurol Surg A Cent Eur Neurosurg. 2013. 74: 71-6
15. Kawamata T, Iseki H, Shibasaki T, Hori T. Endoscopic augmented reality navigation system for endonasal transsphenoidal surgery to treat pituitary tumors: Technical note. Neurosurgery. 2002. 50: 1393-7
16. Kersten-Oertel M, Chen SS, Drouin S, Sinclair DS, Collins DL. Augmented reality visualization for guidance in neurovascular surgery. Stud Health Technol Inform. 2012. 173: 225-9
17. King AP, Edwards PJ, Maurer CR, de Cunha DA, Hawkes DJ, Hill DL. A system for microscope-assisted guided interventions. Stereotact Funct Neurosurg. 1999. 72: 107-11
18. Kockro RA, Tsai YT, Ng I, Hwang P, Zhu C, Agusanto K. Dex-ray: Augmented reality neurosurgical navigation with a handheld video probe. Neurosurgery. 2009. 65: 795-807
19. Kurtsoy A, Menku A, Tucer B, Oktem IS, Akdemir H. Neuronavigation in skull base tumors. Minim Invasive Neurosurg. 2005. 48: 7-12
20. Kurtsoy A, Menku A, Tucer B, Suat Oktem I, Akdemir H, Kemal Koc R. Transbasal approaches: Surgical details, pitfalls and avoidances. Neurosurg Rev. 2004. 27: 267-73
21. Laborde G, Gilsbach J, Harders A, Klimek L, Moesges R, Krybus W. Computer assisted localizer for planning of surgery and intra-operative orientation. Acta Neurochir (Wien). 1992. 119: 166-70
22. Lang DA, Honeybul S, Neil-Dwyer G, Evans BT, Weller RO, Gill J. The extended transbasal approach: Clinical applications and complications. Acta Neurochir (Wien). 1999. 141: 579-85
23. Low D, Lee CK, Dip LL, Ng WH, Ang BT, Ng I. Augmented reality neurosurgical planning and navigation for surgical excision of parasagittal, falcine and convexity meningiomas. Br J Neurosurg. 2010. 24: 69-74
24. Maciunas RJ. Intraoperative cranial navigation. Clin Neurosurg. 1996. 43: 353-81
25. Marmulla R, Mühling J, Wirtz CR, Hassfeld S. High-resolution laser surface scanning for patient registration in cranial computer-assisted surgery. Minim Invasive Neurosurg. 2004. 47: 72-8
26. Oishi M, Fukuda M, Yajima N, Yoshida K, Takahashi M, Hiraishi T. Interactive presurgical simulation applying advanced 3D imaging and modeling techniques for skull base and deep tumors. J Neurosurg. 2013. 119: 94-105
27. Pfisterer WK, Papadopoulos S, Drumm DA, Smith K, Preul MC. Fiducial versus nonfiducial neuronavigation registration assessment and considerations of accuracy. Neurosurgery. 2008. 62: 201-7
28. Pillai P, Sammet S, Ammirati M. Application accuracy of computed tomography-based, image-guided navigation of temporal bone. Neurosurgery. 2008. 63: 326-32
29. Raabe A, Krishnan R, Wolff R, Hermann E, Zimmermann M, Seifert V. Laser surface scanning for patient registration in intracranial image-guided surgery. Neurosurgery. 2002. 50: 797-801
30. Ryan MJ, Erickson RK, Levin DN, Pelizzari CA, Macdonald RL, Dohrmann GJ. Frameless stereotaxy with real-time tracking of patient head movement and retrospective patient-image registration. J Neurosurg. 1996. 85: 287-92
31. Shamir RR, Freiman M, Joskowicz L, Spektor S, Shoshan Y. Surface-based facial scan registration in neuronavigation procedures: A clinical study. J Neurosurg. 2009. 111: 1201-6
32. Simpson D. The recurrence of intracranial meningiomas after surgical treatment. J Neurol Neurosurg Psychiatry. 1957. 20: 22-39
33. Sipos EP, Tebo SA, Zinreich SJ, Long DM, Brem H. In vivo accuracy testing and clinical experience with the ISG Viewing Wand. Neurosurgery. 1996. 39: 194-202
34. Stadie AT, Reisch R, Kockro RA, Fischer G, Schwandt E, Boor S. Minimally invasive cerebral cavernoma surgery using keyhole approaches-solutions for technique-related limitations. Minim Invasive Neurosurg. 2009. 52: 9-16
35. Steinmeier R, Fahlbusch R, Ganslandt O, Nimsky C, Buchfelder M, Kaus M. Intraoperative magnetic resonance imaging with the magnetom open scanner: Concepts, neurosurgical indications, and procedures: A preliminary report. Neurosurgery. 1998. 43: 739-47
36. Stidd DA, Wewel J, Ghods AJ, Munich S, Serici A, Keigher KM. Frameless neuronavigation based only on 3D digital subtraction angiography using surface-based facial registration. J Neurosurg. 2014. 121: 745-50
37. Stieglitz LH, Fichtner J, Andres R, Schucht P, Krähenbühl AK, Raabe A. The silent loss of neuronavigation accuracy: A systematic retrospective analysis of factors influencing the mismatch of frameless stereotactic systems in cranial neurosurgery. Neurosurgery. 2013. 72: 796-807
38. Sure U, Alberti O, Petermeyer M, Becker R, Bertalanffy H. Advanced image-guided skull base surgery. Surg Neurol. 2000. 53: 563-72
39. Thompson EM, Anderson GJ, Roberts CM, Hunt MA, Selden NR. Skull-fixated fiducial markers improve accuracy in staged frameless stereotactic epilepsy surgery in children. J Neurosurg Pediatr. 2011. 7: 116-9
40. Villalobos H, Germano IM. Clinical evaluation of multimodality registration in frameless stereotaxy. Comput Aided Surg. 1999. 4: 45-9
41. Watanabe E, Mayanagi Y, Kosugi Y, Manaka S, Takakura K. Open surgery assisted by the neuronavigator, a stereotactic, articulated, sensitive arm. Neurosurgery. 1991. 28: 792-9
42. Watanabe E, Watanabe T, Manaka S, Mayanagi Y, Takakura K. Three-dimensional digitizer (neuronavigator): New equipment for computed tomography-guided stereotaxic surgery. Surg Neurol. 1987. 27: 543-7
43. Wiltfang J, Rupprecht S, Ganslandt O, Nimsky C, Keßler P, Schultze-Mosgau S. Intraoperative image-guided surgery of the lateral and anterior skull base in patients with tumors or trauma. Skull Base. 2003. 13: 21-29
44. Woerdeman PA, Willems PW, Noordmans HJ, Tulleken CA, van der Sprenkel JW. Application accuracy in frameless image-guided neurosurgery: A comparison study of three patient-to-image registration methods. J Neurosurg. 2007. 106: 1012-6
45. Wolfsberger S, Rössler K, Regatschnig R, Ungersböck K. Anatomical landmarks for image registration in frameless stereotactic neuronavigation. Neurosurg Rev. 2002. 25: 68-72
46. Xu F, Karampelas I, Megerian CA, Selman WR, Bambakidis NC. Petroclival meningiomas: An update on surgical approaches, decision making, and treatment results. Neurosurg Focus. 2013. 35: E11-
47. Zinreich SJ, Tebo SA, Long DM, Brem H, Mattox DE, Loury ME. Frameless stereotaxic integration of CT imaging data: Accuracy and initial applications. Radiology. 1993. 188: 735-42